Determinants of mother-to-child transmission of hepatitis B virus among pregnant women in southwestern Nigeria
Musa Ayinde, Kehinde Olufemi-Aworinde, Victor Joel-Medewase, Medinat Aliu-Ayinde, Olufemi Aworinde, Adewale Adeyemi
Corresponding author: Olufemi Aworinde, Department of Obstetrics and Gynaecology, Faculty of Clinical Sciences, Ladoke Akintola University of Technology, Ogbomoso, Nigeria 
Received: 02 Sep 2023 - Accepted: 28 Jun 2024 - Published: 07 Aug 2024
Domain: Infectious diseases epidemiology,Obstetrics and gynecology
Keywords: HBsAg, HBeAg, HBV DNA, pregnant women, rate of vertical transmission of HBV
©Musa Ayinde et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Musa Ayinde et al. Determinants of mother-to-child transmission of hepatitis B virus among pregnant women in southwestern Nigeria. Pan African Medical Journal. 2024;48:161. [doi: 10.11604/pamj.2024.48.161.41605]
Available online at: https://www.panafrican-med-journal.com//content/article/48/161/full
Research 
Determinants of mother-to-child transmission of hepatitis B virus among pregnant women in southwestern Nigeria
Determinants of mother-to-child transmission of hepatitis B virus among pregnant women in southwestern Nigeria
Musa Ayinde1, Kehinde Olufemi-Aworinde2, Victor Joel-Medewase3, Medinat Aliu-Ayinde4, ![]() Olufemi Aworinde1,&, Adewale Adeyemi1
Olufemi Aworinde1,&, Adewale Adeyemi1
&Corresponding author
Introduction: hepatitis B virus (HBV) infection is a serious public health problem in sub-Saharan Africa and a common cause of liver disease globally. This study aimed to determine the seroprevalence and the rate of mother-to-child transmission of HBV after the age of viability.
Methods: the study was a cross-sectional study that involved 543 eligible consenting pregnant women and newborns of Hepatitis B surface antigen (HBsAg) positive mothers. A one-step rapid HBsAg strip was used to screen eligible patients for HBV infection. Venous blood sample (5mls) was taken from every HBsAg-positive woman for Hepatitis B e Antigen (HBeAg), Hepatitis B e antibody (HBeAb), Hepatitis B core Antibody (HBcAb) and Hepatitis B surface Antibody (HBsAb). In addition, 2mls of cord blood was taken to assay for HBsAg and HBV DNA. Data was analysed using SPSS version 22.
Results: of 543 pregnant women screened, 18 (3.3%) of them were HBsAg-positive with all of them testing negative for HBeAg. HBV DNA was detected in the cord blood of 4 (22.2%) new-borns delivered while 2 (11.1%) tested positive for HBsAg; the above finding indicated that only 4 of the neonates had detectable HBV DNA (>100copies/ml) in their cord blood.
Conclusion: findings from this study demonstrate a low prevalence of HBV infection among pregnant women after the age of viability in Ogbomoso. HBV DNA analysis rather than HBsAg was shown to be more sensitive and specific in determining the risk of intrauterine infections.
Hepatitis B virus (HBV) infection in pregnancy is an important public health concern, particularly in developing countries like Nigeria, HBV is a DNA virus that can be transmitted percutaneously, sexually, or perinatally. Hepatitis B e Antigen (HBeAg) was acknowledged later as a marker of infectivity [1]. Worldwide, about 2 billion people have been infected with HBV, and approximately 400 million people have Chronic Hepatitis B (CHB) infection [2]. The prevalence of HBV infection is relatively high in Africa, which has the second highest number of chronically HBV-infected individuals [3]. The majority of the world's chronic HBV infection carriers, especially children, acquire this infection via the perinatal route. If the infection occurs in the perinatal period, the risk of chronic carriage of HBV infection is about 95% [4].
Hepatitis B infection is endemic in Nigeria, with the prevalence ranging from 9.6% to 18.6% among pregnant women. Currently, about 18 million Nigerians are infected, of which most of them are unaware of their status. The effect of this is an increase in the incidence of complications such as chronic liver disease, cirrhosis, and hepatocellular carcinoma (HCC) [5]. Pregnant women with hepatitis B infection constitute a significant risk to their unborn children, partners, health care providers, and society at large. Mother-to-child transmission (MTCT) is the main route of transmission of hepatitis B virus in areas of high endemicity. MTCT of hepatitis B virus continues to occur despite the interventions of hepatitis B vaccination and immunoglobulin administration in settings where they are practiced. Infants most at risk are those whose mothers have high HBV DNA and produce the protein HBeAg [6]. Various studies in Nigeria have reported high HBV infection rates as well as HBeAg positivity among pregnant women which usually occur during delivery [7].
Detection of specific antibodies and/or antigens in the serum of patients is usually the basis for diagnosis. The most important laboratory test for diagnosis of HBV infection is the detection of HBsAg, which is the first antigen to appear. Antibodies to HBsAg (Anti-HBs) replace HBsAg once the acute infection resolves, and this indicates immunity in about 80% of cases [1]. Anti-HBs also appear after HBV vaccination, but these antibodies could be lost after acute HBV infection, and the patient may thus become susceptible to the disease [2]. Other investigations include the detection of HBeAg, which is an indicator of transmissibility and is replaced by HBeAb, whose presence indicates low transmissibility [2].
The reported prevalence rate of HBV infection among pregnant women in African countries ranged from 6% to 25% [8]. However, in developed countries, such as America and Europe, the prevalence was reported to be 0.1% among pregnant women [6]. The results of these studies confirm the conclusion of the World Health Organization (WHO) that African countries are endemic for HBV infection [8]. The age of viability is the gestational age at which a fetus has a 50% chance of survival in a particular environment. It varies from one country to the other [9]. The World Health Organization (WHO) takes 20 weeks as the age of fetal viability while in Nigeria, the age of viability is taken to be 28 weeks [10]. Since this study was carried out in southwestern Nigeria, 28 weeks was taken as the age of viability. The objectives of this study aimed to determine the seroprevalence and the rate of mother-to-child transmission (MTCT) of HBV during pregnancy after the age of viability in southwestern Nigeria and the factors that influence the rate of MTCT.
Study design: the study was a hospital-based cross-sectional study, conducted between 1st June 2020 to May 31st 2021 in pregnant women after the age of viability.
Setting: the study was carried out at the antenatal clinic and labour ward of the Department of Obstetrics and Gynaecology, Ladoke Akintola University of Technology Teaching Hospital (LTH), Ogbomoso, Oyo state, Nigeria.
Study participants and sampling procedure: a purposive non-probability sampling technique was used. All eligible and consented pregnant women were recruited into the study from the antenatal clinic and labour ward.
Research questions: what is the prevalence of HBsAg positivity among pregnant women after the age of viability? What is the rate of MTCT of hepatitis B virus infection among pregnant women after the age of viability? What are the factors influencing the rate of MTCT of hepatitis B infection among pregnant women after the age of viability?
Variables and data collection: participants who consented were interviewed. Pre-test counselling was done, and then the questionnaire was administered to obtain socio-demographic information and relevant history. Blood samples were obtained to screen eligible patients for HBV infection. HBsAg-positive mothers had post-test counselling. Their blood samples were further screened to detect HBeAg, HBeAb, HBcAb, and HBsAb. At delivery, cord blood of neonates of HBsAg-positive mothers was obtained and screened for HBsAg and HBV DNA. The HBsAg-positive mothers were managed according to the unit protocol. This involved delayed artificial rupture of membrane and low-pressure suctioning using an electrical suction machine after delivery. All babies delivered to HBsAg-positive mothers had hepatitis B immunoglobulin within 12 hours of delivery and hepatitis B vaccine on the contralateral thigh before discharge.
Study size: the sample size was derived using Fisher's formula [11]:
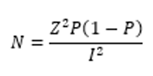
where N is the minimum sample size, z is the z-score that corresponds to the 95% confidence interval (1.96), and p is the expected proportion i.e. prevalence of 7.3% for the disease among pregnant women in Lagos was used [12], and l is the margin of error set at 5%. A sample size of five hundred and forty-three pregnant women was used.
Statistical methods: data were entered into the Statistical Product and Service Solutions (SPSS version 22) and analysed. Descriptive statistics was used to summarize the data. The proportions and the corresponding 95% confidence interval (CI) were presented. The frequency table was used for categorical variables, and the Chi-square test was used to test the association between variables. Bivariate analysis for each variable in the study was done and p-value <0.05 for strength of association. Specificity and sensitivity tests were done for both test methods. Multiple regression analysis of the maternal characteristics and neonatal HBV DNA was done.
Ethical approval: ethical clearance was obtained from the ethics committee of LAUTECH Teaching Hospital (LTH), Ogbomoso with protocol number LTH/OGB/EC/2020/210. Participant information was treated as strictly confidential and was not used for any unintended purposes.
Participants: five hundred and forty-three (543) subjects were recruited for the study as shown in Table 1. The majority of the respondents were within the age brackets of 20-29 years. More than half of the respondents were within the parity of 0-1 (64%). Nearly all were married with secondary and tertiary levels of education. Only eighteen (3.3%) of the subjects were HBsAg-positive.
Descriptive/outcome data: all the eighteen subjects who tested positive for HBV were negative for HBeAg. However, most of them (>70%) tested positive for Anti-HBsAg, Anti-HBeAg and anti-HBc antibodies (Table 2). The majority of the respondents delivered through spontaneous vaginal delivery. Most of the respondents (66.7%) had spontaneous rupture of membranes with most having less than 4 hours interval between membrane rupture and delivery. Most of the respondents (88.9%) delivered at term with labour duration of less than 12 hours (Table 2).
Main results: Table 3 shows that the rate of mother-to-child transmission of HBV infection among neonates of HBsAg-positive mothers delivered after the age of viability was 11.1% using HBsAg and 22.2% using HBV DNA (>100 copies/ml). It was found that 4 (22.2%) out of the 18 cord blood samples gotten from neonates of HBsAg-positive mothers were positive for HBV DNA while 2 (11.1%) were positive for HBsAg. This was statistically significant (p<0.05). The sensitivity and specificity of HBV DNA for detecting mother-to-child transmission of hepatitis B infection were 67.1% and 87.5% respectively while the sensitivity and specificity of HBsAg for detecting mother-to-child transmission of hepatitis B infection were 32.9% and 12.5% respectively (Table 3, Table 4, Table 5).
Other analysis: multiple regression analysis of the maternal characteristics and neonatal HBV DNA presence showed that an increase in the duration of labour and time interval between membrane rupture and delivery were associated with an increase in neonatal HBV DNA titre (p<0.005) (Table 3).
Key results: five hundred and forty-three women were recruited for this study. Most respondents were married young adults. This was similar to the findings of Aworinde et al. [13,14]. Eighteen of the women recruited tested positive for HBsAg, putting the prevalence of hepatitis B infection at 3.3%. This indicates a moderate prevalence of hepatitis B in our environment and does not support the inclusion of Nigeria by WHO as a high-endemic region with a prevalence greater than 8% [15]. The moderate prevalence in this study could be a result of the implementation of the national HBV infection prevention strategic plan. The strategic plan may have led to the reduction in the burden of endemicity of HBV infection and ultimately the rate of mother-to-child transmission. The national HBV infection prevention strategic plan includes routine screening of pregnant women and administration of passive immunoglobulin to all neonates of HBsAg-positive women in addition to active hepatitis B immunization to all newborns irrespective of the status of the mother. Another factor that might be responsible for the low prevalence may be the fact that the study was a hospital-based study involving only booked women whose pregnancy had reached the age of viability. Similar studies done in Osogbo and Makurdi reported low prevalence rates of 2.8% and 3.9% respectively, while higher values were recorded by other researchers [16-19].
This research tested for the presence of HBsAg and HBeAg in maternal blood as well as for both HBsAg and HBV DNA in the cord blood of neonates born to mothers who were HBsAg positive. The absence of HBeAg in all the respondents was similar to the findings in other African studies [4,14,20]. This could be due to the predominance of HBeAg-negative chronic HBV linked to HBV genotype D which is prevalent in Africa [21,22]. Maternal HBV DNA was not done in this study due to the negative HBeAg status of all the studied patients which indicates a low level of maternal viraemia and infectivity. However, other researchers such as Obi et al. [23] in Port Harcourt and Olaleye et al. in Ile-Ife noted a significant relationship between the maternal HBeAg, HBV DNA >107IU/ml and the risk of intrauterine infections [15,23]. These findings support a significant relationship between negative HBeAg, suspected low level of maternal viral load and lower risk of intra-uterine infection which was noted in our findings.
Multiple sexual partners, change of husband, previous history of blood transfusion, history of scarification and sharing of hair-parting needles were noted to be significantly associated with HBV infections among pregnant women recruited in our study. Similar findings were also noted by other researchers [23-27]. HBV is an organism that thrives in blood and other body fluids; hence, its transmission through the above routes is not surprising.
The incidence of mother-to-child transmission of HBV varies from 2.1 to 50% as reported by different researchers [28]. The vertical transmission risk of 22.2% was found in this study using HBV DNA and 11.1% using HBsAg with specificity of 87.5% and 12.5% respectively. Zhang and his colleagues found a lower incidence of 17.1% when using HBsAg compared to 41.5% on using HBV DNA [28]. Olaleye et al. reported an incidence of 24% using HBsAg and 72% using HBV DNA in Ile Ife [15]. The risk of intrauterine infection noticed in this study appeared lower when compared with the two studies above. The reason could be a result of the fact that most patients studied were in the reactivation phase with negative HBeAg which inferred a low level of maternal viral load which could account for the low risk of perinatal transmission as found in this study.
A negative maternal HBeAg was suspected to be the reason for the low risk of intrauterine infection reported in our study when compared with other studies, this is in line with the findings of Olaleye et al. and Xu et al. on a direct significant relationship between very high maternal HBV DNA levels and neonatal risk of intra-uterine infections [15,28]. There was a statistically significant association between the risk of MTCT of HBV DNA, prolonged labour and time interval between rupture of membrane and delivery greater than 4 hours. Olaleye et al. and Lavanchy et al. also had similar findings in their studies [15,29]. However a study done by Mimi and Cheung in Hong Kong found the mode of delivery to be significantly associated with the risk of MTCT of HBV DNA unlike in this study [30].
Limitations: none of the subjects studied was HBeAg positive so its association with HBV DNA core antigen in maternal-to-child transmission could not be assessed.
Findings from this study demonstrate a low prevalence of HBV infection among pregnant women after the age of viability in Ogbomoso. HBV DNA analysis rather than HBsAg was shown to be more sensitive and specific in determining the risk of intrauterine infections.
What is known about this topic
- Pregnant women with hepatitis B infection constitute a significant risk to their unborn children with infants most at risk being those whose mothers have high HBV DNA.
What this study adds
- The prevalence of hepatitis B in this study does not corroborate the previous inclusion of Nigeria by WHO as a high endemic region with a prevalence greater than 8%;
- The rate of MTCT is low in the absence of HBeAg.
The authors declare no competing interests.
The study was self-funded. It was sponsored by the authors with no external funding.
Musa Ayinde conceptualized the study, sought ethical approval, and collected data. Victor Joel-Medewase and Medinat Aliu-Ayinde analysed the data and drafted the manuscript. Kehinde Olufemi-Aworinde, Olufemi Aworinde and Adewale Adeyemi guided, supervised and revised the manuscript. All authors read and approved the final version of the manuscript.
We appreciate the contribution of the staff and students of the Department of Obstetrics and Gynaecology, Ladoke Akintola University of Technology Teaching Hospital, Ogbomoso.
Table 1: socio-demographic characteristics of the respondents (n=543)
Table 2: HBV serology profile and delivery history of HBV positive subjects (n=18)
Table 3: association between cord blood detection of HBV DNA and HBsAg positivity, its sensitivity and specificity
Table 4: relationship between delivery history and MTCT
Table 5: relationship between maternal characteristics and neonatal HBV DNA
- Dienstag JL. Hepatitis B virus infection. N Engl J Med. 2008;359(14):1486-500. PubMed
- Thio CL, Guo N, Xie C, Nelson KE, Ehrhardt S. Global elimination of mother-to-child transmission of hepatitis B: revisiting the current strategy. Lancet Infect Dis. 2015;15(8):981-5. PubMed | Google Scholar
- Mbaawuaga E, Enenebeaku M, Okopi J, Damen J. Hepatitis B Virus Infection among Pregnant Women in Makurdi, Nigeria. African Journal of Biomedical Research. 2008;11(2): 155-159. Google Scholar
- Jonas MM. Hepatitis B and pregnancy: an underestimated issue. Liver Int. 2009;29:133-9. PubMed | Google Scholar
- Musa BM, Bussell S, Borodo MM, Samaila AA, Femi OL. Prevalence of hepatitis B virus infection in Nigeria, 2000-2013: a systematic review and meta-analysis. Niger J Clin Pract. 2015; 18(2):163-72. PubMed | Google Scholar
- Lam NC, Gotsch PB, Langan RC. Caring for pregnant women and newborns with hepatitis B or C. Am Fam Physician. 2010;82(10):1225-9. PubMed | Google Scholar
- Okoth F, Mbuthia J, Gatheru Z, Murila F, Kanyingi F. Seroprevalence of hepatitis B markers in pregnant women in Kenya. East Afr Med J. 2006;83(9):485-93. PubMed | Google Scholar
- Dortey BA, Anaba EA, Lassey AT, Damale NKR, Maya ET. Seroprevalence of Hepatitis B virus infection and associated factors among pregnant women at Korle-Bu Teaching Hospital, Ghana. PLoS One. 2020;15(4):e023220. PubMed | Google Scholar
- Nadroo AM. ethical dilemmas in decision making at limits of viability. JIMA. 2011;43(3):188-191. PubMed | Google Scholar
- Ikechebelu JI, Eleje GU, Ugochukwu EF, Edokwe ES (2014) Should we Re-Define Age of Fetal Viability in Nigeria? A Case Report of Newborn Survival from Pre-Viable Pre-Labor Rupture of Membranes. J Womens Health, Issues Care 3:3. Google Scholar
- Adegbesan-Omilabu MA, Okunade KS, Gbadegesin A, Olowoselu OF, Oluwole AA, Omilabu SA. Seroprevalence of hepatitis B virus infection among pregnant women at the antenatal booking clinic of a Tertiary Hospital in Lagos Nigeria. Niger J Clin Pract. 2015;18(6):819-23. PubMed | Google Scholar
- Aworinde OO, Olufemi-Aworinde KJ, Ogunlaja OA, Olutogun TA, Akinola SE, Adeyemi AS. Optimal timing of post-operative haematocrit check in obstetric and gynaecological surgeries. Research Journal of Health Sciences. 2018; 6(1):35-42. Google Scholar
- Olufemi-Aworinde K, Aworinde O, Adejola T, Olayinka O, Abolarin A. Haematological parameters among pregnant women in Ogbomoso, Southwestern Nigeria. Integrated Journal of Medicine and Medical Sciences. 2021; 2(1):32-38.
- Olaleye OA, Kuti O, Makinde NO, Ujah AOI, Olaleye OA, Badejoko OO. Perinatal transmission of hepatitis B virus infection in Ile-Ife, South Western, Nigeria. J Neonatal Perinatal Med. 2013;6(3):231-6. PubMed | Google Scholar
- Adegbesan-Omilabu MA, Okunade KS, Gbadegesin A, Olowoselu OF, Oluwole AA, Omilabu SA et al. Sero-prevalence of hepatitis B virus infection among pregnant women at the antenatal booking clinic of a Tertiary Hospital in Lagos Nigeria. Niger J Clin Pract. 2015;18(6):819-23. PubMed | Google Scholar
- Ajayi AO, Ade-Ojo IP, Ajayi EA, Adegun PT, Ojo A et al. Sero-prevalence of hepatitis B infection in pregnant women at the Ekiti State University Teaching Hospital, Ado-Ekiti, Southwestern Nigeria. Afr J Intern Med. 2013;2(4):023-025. Google Scholar
- Kolawole OM, Wahab AA, Adekanle DA, Sibanda T, Okoh AI. Seroprevalence of hepatitis B surface antigenemia and its effects on hematological parameters in pregnant women in Osogbo, Nigeria. Virol J. 2012 Dec 27;9:317. PubMed | Google Scholar
- Aluor E, Oluma H, Ega R, Owolcho N. Sero-epidemiological survey and risk factors for Hepatitis B Virus (HBV) infection among pregnant women in Logo LGA, Benue State, Nigeria. AJCEM. 2015;17(1):66. Google Scholar
- Sangaré L, Sombié R, Combasséré AW, Kouanda A, Kania D, Zerbo O et al. Antenatal transmission of hepatitis B virus in an area of HIV moderate prevalence, Burkina Faso. Bull Soc Pathol Exot. 2009;102(4):226-9. PubMed | Google Scholar
- Ott JJ, Stevens GA, Groeger J, Wiersma ST. Global epidemiology of hepatitis B virus infection: New estimates of age-specific HBsAg sero-prevalence and endemicity. Vaccine. 2012; 30(12):2212- 19. PubMed | Google Scholar
- European Association For The Study Of The Liver. EASL Clinical Practice Guidelines: management of chronic hepatitis B. J Hepatol. 2009 Feb;50(2):227-42. PubMed | Google Scholar
- Obi RK, Emeh SC, Okurede OH, Iroagba II. Prevalence of hepatitis B virus among pregnant women in antenatal clinic in Port Harcourt, Nigeria. Afr J Clin Exp Microbiol, 2006;7:567-7. Google Scholar
- Alemu AA, Zeleke LB, Aynalem BY, Kassa GM. Hepatitis B Virus Infection and Its Determinants among Pregnant Women in Ethiopia: A Systematic Review and Meta-Analysis. Infect Dis Obstet Gynecol. 2020 Jun 11;2020:9418475. PubMed | Google Scholar
- Bruss V. Hepatitis B virus morphogenesis. World J Gastroenterol. 2007;13(1):65-73. PubMed | Google Scholar
- Akani CI, OjuleAC, Opurum AC, Ejielemele AA. Sero-prevalence of HBsAg in pregnant women in Port-Harcourt, Nigeria. Niger Postgrad Med J. 2005;12(4):266-70. PubMed
- Uyar Y, Cabar C, Balci A. Sero-prevalence of Hepatitis B virus among pregnant women Northern Turkey. Hepatitis Monthly. 2009;9(2):146-149. Google Scholar
- Xu DZ, Yan YP, Choi BC, Xu JQ, Men K, Zhang JX et al. Risk factors and mechanism of transplacental transmission of hepatitis B virus: a case-control study. J Med Virol. 2002;67(1):20-6. PubMed | Google Scholar
- Lavanchy D. Hepatitis B virus epidemiology, disease burden, treatment, and current and emerging prevention and control measures. J Viral Hepat. 2004;11(2):97-107. PubMed | Google Scholar
- Mimi TY, Cheung KW. Prevention of maternal to child transmission of hepatitis B infection: systemic review. Hong Kong J Gynaecol Obstet Midwifery 2021;21(1):49-52. Google Scholar











