Prevalence and associated factors of a positive Plasmodium falciparum antigen test among pregnant women at the Bamenda Regional Hospital, Cameroon: a cross-sectional analytical study
Dobgima Walters Pisoh, Achuo Ascensius Ambe Mforteh, William Ako Takang, Joseph Bakowe, Theodore Tameh, Merlin Boten, Audrey-Fidelia Eyere Mbi-Kobenge, Sama Julius Dohbit
Corresponding author: Achuo Ascensius Ambe Mforteh, Faculty of Health Sciences, University of Bamenda, Bamenda, Cameroon 
Received: 28 Jun 2023 - Accepted: 25 Feb 2024 - Published: 01 Mar 2024
Domain: Obstetrics and gynecology
Keywords: Plasmodium falciparum, pregnant women, malaria, prevalence, sub-Saharan Africa
©Dobgima Walters Pisoh et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Dobgima Walters Pisoh et al. Prevalence and associated factors of a positive Plasmodium falciparum antigen test among pregnant women at the Bamenda Regional Hospital, Cameroon: a cross-sectional analytical study. Pan African Medical Journal. 2024;47:99. [doi: 10.11604/pamj.2024.47.99.40899]
Available online at: https://www.panafrican-med-journal.com//content/article/47/99/full
Research 
Prevalence and associated factors of a positive Plasmodium falciparum antigen test among pregnant women at the Bamenda Regional Hospital, Cameroon: a cross-sectional analytical study
This article has been retracted
This article was retracted on the 9th August 2024. See PAMJ Retraction Policy
See retraction Pan African Medical Journal. 2024;48:168 | 10.11604/pamj.2024.48.168.40899
Prevalence and associated factors of a positive Plasmodium falciparum antigen test among pregnant women at the Bamenda Regional Hospital, Cameroon: a cross-sectional analytical study
Dobgima Walter Pisoh1,2, ![]() Achuo Ascensius Ambe Mforteh1,2,&, William Ako Takang1, Joseph Bakowe1, Theodore Yangsi Tameh1, Merlin Boten1, Audrey-Fidelia Eyere Mbi-Kobenge1, Sama Julius Dohbit 3
Achuo Ascensius Ambe Mforteh1,2,&, William Ako Takang1, Joseph Bakowe1, Theodore Yangsi Tameh1, Merlin Boten1, Audrey-Fidelia Eyere Mbi-Kobenge1, Sama Julius Dohbit 3
&Corresponding author
Introduction: approximately 25 million pregnant women in sub-Saharan Africa are at risk of malaria despite various preventive methods. This study aimed to determine the prevalence of Plasmodium falciparum parasite and its associated factors in pregnant women in Bamenda
Methods: a hospital-based cross-sectional study conducted in March and April 2021 including pregnant women attending ANC at the Bamenda Regional Hospital. A questionnaire was used to collect data. The positive Plasmodium falciparum test was determined using a malaria rapid diagnostic test. A frequency table was used to describe the data, and a Chi-square to determine the association between independent and dependent variables. Factors significantly associated with a positive test were included in a multivariable model.
Results: among the 380 participants, 70 had a positive rapid diagnostic test (prevalence: 18.4%). Living in rural area (OR: 4.93, 95% CI: 1.86-13.14), not using insecticide-treated nets (OR: 109.95, 95% CI: 49.43-271.73), not using indoor residual spraying (OR: 15.21, 95% CI: 3.25-271.35), first and second trimesters of gestation compared to third trimester (OR: 20.27, 95% CI: 7.33-61.29; OR: 2.75, 95% CI: 1.49-5.33 respectively) and presence of mosquito breeding site (OR = 99.75, 95% CI: 38.84-340) were associated with positive test on bivariate analysis. Following multivariate logistic regression, the presence of mosquito breeding sites (AOR: 21.89, 95% CI: 5.25-121.13), and not using insecticide-treated nets (AOR: 10.82, 95% CI: 3.13-40.37) remained significantly associated.
Conclusion: the prevalence of positive Plasmodium falciparum test is high among pregnant women. Numerous risk factors are associated with positive Plasmodium falciparum among these women.
Malaria is a life-threatening parasitic infection caused by protozoa of the genus Plasmodium and transmitted by female Anopheles mosquitoes. Five different human malaria species exist: P. falciparum, P. vivax, P. ovale, P. malariae, and P. knowlesi. In malaria endemic regions, individuals are repeatedly exposed to malaria parasites through bites from the aforementioned mosquitoes [1]. This frequent exposure leads to the development of an effective immunity against the disease, which prevents parasite loads from reaching life-threatening thresholds, and suppresses the proinflammatory responses that cause illness [2]. In 2016, an estimated 216 million cases of malaria and 445,000 deaths occurred worldwide [3]. About 90% of all malaria cases and 91 % of all malaria death in 2015 and 2016 were reported from the World Health Organization African Region [3,4]. Of the 91 countries reporting indigenous cases, around 80% of the total cases were from sub-Saharan African countries [3,4]. Malaria in pregnant women is caused mainly by the P. falciparum species, which is the most common species in Africa [1]. Two institution-based studies done among pregnant women attending antenatal care (ANC) in Nigeria showed the prevalence of malaria to be 41.6 % and 7.7% [5,6]. Another institution-based study in Eastern Sudan found that 13.7% of pregnant women were infected with P. falciparum [7]. Studies conducted in other African countries (Burkina Faso, and Malawi) also showed the prevalence to be 18.1% and 19% respectively [8,9]. The situation is similar in Cameroon as malaria still remains a major public health problem affecting children and pregnant women. In 2017, more than 1.191.257 cases of malaria were reported and this is most predominant among pregnant women and children below five years. The clinical morbidity rate is estimated at 41%, the mortality rate at 2.2% [10].
Pregnant women are three times more likely to suffer from severe morbidity from malarial infection compared with their non-pregnant counterparts, and have a high mortality rate that approaches 50% [11]. The principal impact of malaria infection is due to the presence of parasites in the placenta, which causes maternal anemia and low birth weight [12]. Numerous risks factors for malaria among pregnant women were identified by previous studies. These include low educational status [1,8], young maternal age [6], no or few Antenatal consultation visits [12], first trimester pregnancy [12], low parity [8,12], primigravida and no Insecticide treated-net utilization [13]. Diagnosis of malaria during pregnancy can be complicated by the absence of parasites in the peripheral blood or by parasite densities below the detection limit of microscopy caused by placental sequestration [14]. Accurate detection of parasite infection in the placenta requires histologic examination of sections of fixed placental tissue, which is the reference or gold standard for diagnosing placental malaria. Unfortunately, placental histologic analysis and microscopy of placental blood cannot be routinely performed before delivery. It is of prime importance both to mother and fetus that malaria in the peripheral blood be diagnosed earlier, and appropriate treatment given. It is therefore necessary to detect the placental infection with a marker that is present in peripheral blood. Currently, available methods for the diagnosis of malaria in peripheral blood are parasite detection by microscopy, DNA or RNA polymerase chain reaction (PCR), and rapid diagnostic tests (RDTs) to detect malarial antigens [14].
The sensitivity and specificity of malaria RDTs in non-pregnant subjects was estimated in one study at 80% and 93.8% respectively with microscopy as the reference test [15]. While many studies have not been done on sensitivity and specificity of RDTs in pregnancy, one study found sensitivity of both conventional RDTs and highly-sensitive RDTs to vary from 22.8 to 82.8% and 19.6 to 85.7% respectively, when compared with molecular testing [16]. They also found that RDTs could detect 15-30% of infections with parasitemia of 0-2 parasites per microliter which is far lower than the 5 parasites per microliter threshold for expert microscopists and 50-100parasites per microliter threshold for an average microscopist. The high burden of malaria especially in pregnant women warrants early detection and treatment of malaria among pregnant women in order to prevent complications. In a study by Elime et al. [17] in 2019, the prevalence of malaria among pregnant women in the Mamfe Health District, South West region of Cameroon was 39.6%. This study aims to determine the prevalence and associated risk factors of positive Plasmodium falciparum antigen among pregnant women in another geographical area of Cameroon, the North West region.
Study design: a hospital-based cross-sectional study was conducted to determine the prevalence and risk factors for positive Plasmodium falciparum amongst pregnant women in Bamenda.
Study setting and population: Bamenda is the capital city of the North West Region, one of the ten regions of Cameroon. It has an estimated population of 553,000 inhabitants. The Bamenda Regional Hospital is the main referral hospital of the region. It has three gynecologists/obstetricians and being a government owned facility, its services are relatively cheaper, hence it is more open to a cross-section of the population. The ante-natal consultation (ANC) clinic operates four days a week and receives about 30 women each ANC clinic day. About 200 deliveries are conducted monthly at the Bamenda Regional Hospital (BRH). Our study was piloted from February 22 to 24, 2021. Data for the study were collected from the 1st of March to the 30th of April 2021 at the antenatal clinic of the Bamenda Regional Hospital. Malaria remains a disease with high morbidity and mortality in Bamenda especially amongst pregnant women. Pregnant women routinely receive intermittent preventive treatment for malaria prevention with sulfadoxine-pyrimethamine. They also receive long-lasting insecticide treated mosquito nets. During ANC clinics, they are encouraged to keep their environment clean, and to clear bushes and standing water which may serve as breeding sites for mosquitos.
Inclusion and exclusion criteria: our parent population consisted of pregnant women attending ANC at the BRH. We included all pregnant women who gave an informed consent, and were not on daily cotrimoxazole. Cotrimoxazole has antiparasitic properties hence will act as bias to the study. Exclusion criteria was failure to provide complete data and sample for laboratory test.
Variables: socio-demographic variables consisted of maternal age (defined as the number of completed years from birth, measured in years, extracted from national identity card of participant), marital status (defined as presence of a marriage certificate, information obtained from the participant, classified as married or single), educational level (highest level of education attained by the participant, obtained from the participant), residence (place of residence, obtained from the participant classified as urban or rural) and employment status (classified as formal of participant has a formal job with a salary at month end, otherwise informal). Obstetric variables consisted of number of ANC (defined as the number of times the participant has been seen by a trained professional at the ANC clinic, obtained from ANC records), trimester (classified into 3: first trimester corresponds to up to 12 completed weeks, second trimester from 13 weeks to 27 weeks 6 days, and third trimester from 28 completed weeks to delivery, information calculated from last menstrual period), and number of intermittent preventive treatment with Sulfadoxine-pyrimethamine (SP) (number of times the participant has received SP, obtained from ANC records). Household variables consisted of use of ITNs (defined as sleeping under the ITN, information obtained from the participant), presence of mosquito breeding sites (presence of stagnant water, or slow flowing streams around in the neighborhood, information obtained from participant), and use of indoor residual spraying (define as use of IRS at least once during the current pregnancy). Clinical variables consisted of fever (axillary temperature ≥38.50c, measured using two separate mercury bulbs thermometer on both axillary region and an average taken), myalgia and or arthralgia (feeling of muscle pain, expressed by the participant), chills (sensation of shivering, expressed by the participant), abdominal pain (sensation of pain in the abdomen, expressed by the participant), and headache (pain on the head, expressed by the participant). All above variables were independent variables. Our dependent variable was the Plasmodium falciparum antigen test results (obtained using the malaria rapid diagnostic test). We considered that independent variables could act as possible confounders.
Data resource and management
Data collection tool: data were collected using a structured paper questionnaire. The questionnaire was designed by the investigators. It was pre-tested with 5% of our sample size during the pilot phase of the study by the investigators and a team of trained data collectors. Data obtained during this phase was not used in the final analysis. The data collectors consisted of final year medical students (students doing their research projects) after receiving an intensive training on data collection. Team members were trained on how to: present the study to prospective participants, select participants, request participation, administer informed consent and obtain assent, and administer the questionnaire. The Questionnaire consisted of five sections: sociodemographic data, obstetrical data, household data, clinical data, and paraclinical data from test results.
Data collection
Clinical data: the study was presented to all pregnant women in the ANC clinic. Pregnant women who were eligible and gave their consent were recruited into the study. A face-to-face interviewer-administered pre-tested questionnaire was used to collect data on independent variables by the team of trained data collectors. Collected data was entered into an Excel spreadsheet in a password protected computer. By interviewing the participants individually and assuring confidentiality, we hope that reported bias on sociodemographic and household variables were minimized.
Laboratory data: as concerns the dependent variable (Plasmodium falciparum antigen test result), blood samples were collected from all study participants by a trained laboratory technician in the ANC laboratory of the hospital, which was used to perform a malaria rapid diagnostic test. Blood was obtained from the third finger of each woman´s left hand. First, the tip of the finger was wiped with a piece of cotton wool lightly soaked in alcohol then piercing with a sterile lancet was done and blood was allowed to flow freely into the capillary tube without squeezing the finger. The blood collected was dropped into the round hole of the standard diagnostics (SD) Bioline Malaria Antigen (Ag) Plasmodium falciparum (Pf) Histidine Rich Protein 2 (HRP2) (05FK50) rapid diagnostics test (RDT) cassette and the assay diluent was dropped into the square hole of the cassette. Two tests were performed per participant to control for reliability of the test kit. Fifteen minutes later, results were read by two independent readers to ensure validity of the results. In the event where the two results were different, a third test was to be done and its result taken as the final result. No extra blood sample of participants was stored. Positive tests were obtained by visualizing two lines, while negative tests had just one line.
Sample size: the minimum sample size (n) to obtain a study power of 80% and 95% confidence interval was calculated using the Cochran´s formula;
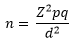
Data analysis: data were analyzed using the Statistical Package for Social Sciences (SPSS) version 20 software. Only data collected during the study proper was used. To obtain the prevalence of Plasmodium falciparum antigen in pregnant women at the BRH, we calculated the proportion of pregnant women with positive malaria RDT amongst all women in the study population. To obtain factors associated with positive Plasmodium falciparum antigen test, bivariate analysis with the Pearson Chi square test was done between each independent variable and the outcome variable, crude odd ratios were obtained, and factors significantly associated with the outcome variable were noted. Factors significant in the bivariate analysis were subsequently entered into a multivariable logistic regression model to obtain adjusted odd ratios and factors that remained significantly associated with a positive rapid diagnostic test. Statistical significance was set at p<0.05 and 95% confidence interval.
Ethical considerations: prior to the study, ethical clearance was obtained from the Bamenda University Ethical Review Board, while administrative clearance to conduct the study at the BRH was obtained from the administration of the hospital. Each participant was interviewed individually and in an enclosed room. Codes were used to replace names, and results of participants were given individually. Participants were informed that should they refuse to participate or consider withdrawing at any point, they shall continue to receive their routine care without any prejudice. Participants who tested positive for Plasmodium falciparum were referred to the obstetrician for management.
Characteristics of the study population
Socio-demographic characteristics: a total of 393 potential women were approached, of which 380 were enrolled and 13 not included (10 failed to give a consent and 3 others were on daily cotrimoxazole). The mean age of the participants was 27.69 ± 4.5 years. The most common age group was 25 - 29 years (n=121, 31.8%) with an age range of 15-49 years. Majority of the study participants were cohabiting (n=267, 70.3%), lived in urban residence (n=362, 95.3%) and had attained at least secondary school level of education (n=368, 96.84%). Also, more than half of the study participants were self-employed (211, 55.5%). Table 1 summarize the socio-demographic characteristics of the study population.
Obstetric and clinical characteristics: the Obstetric characteristics of our study participants are presented in Table 2 below. The mean gestational age was 25.79 ± 8.07 weeks. Most of the participants were either in the second or third trimester of gestation, 359 (94.5%). Three-quarter of the mothers had received IPT (n=288, 75.8%). Three hundred and one (79.2%) used an impregnated treated mosquito net. Unfortunately, up to 85% of women did not use IRS. The most frequent symptoms were fever (55, 14.5%) and chills (42, 11.1%) while headache was the least distributed symptom.
Prevalence of positive Plasmodium falciparum antigen: seventy out of the 380 women tested positive for Plasmodium falciparum giving a prevalence of 18.4% (95% CI:14.85-22.63).
Bivariate analysis: bivariate analysis to determine factors associated with a positive Plasmodium Falciparum test showed that pregnant women living in rural areas were 5 times more likely to have PPFA test than those living in urban areas (OR=4.93, CI: 1.86-13.14, p=< 0.001). Pregnant women living around mosquito breeding sites were 100 times more likely to have positive than those without breeding sites (OR =99.75, CI: 38.84-340.22, p= <0.001). Pregnant women who did not use ITN were 110 times more likely to have PPFA test compared to those that used (OR=109.95, 95% CI: 49.43-271.73, p <0.001). Also, pregnant women who did not use IRS were 15 times more likely to have PPFA test compared to those that used it (OR=15.21, 95% CI: 3.25-271.35, p = 0.008). No intake of IPT was associated with 21-fold increased odds of PPFA (OR= 21.66, CI: 11.64-42.18, p <0.001). Women in the first trimester and second trimesters compared to third trimester had higher odds of having a PPFA test (OR=20.27, 95% CI: 7.33-61.29, p <0.001; OR=2.75, 95% CI: 1.49-5.33, p <0.001 respectively). Positive HIV status was associated with 7-fold increased odds of PPFA (OR=7.18, CI=3.71-14.06 p=<0.001). Pregnant women who had fever were 4.66 times more likely to have a PPFA test (OR=4.66, 95% CI: 3.34-14.58, p =0.008), while those with myalgia were 10.88 times more likely to have a PPFA test (OR=10.88, 95% CI: 4.66-14.45, p <0.001), and those with chills were 7 times more likely to have a PPFA test (OR=7.22, 95% CI: 5.44-8.66, p =0.004).
Multivariate analysis: we further sought to study the association between positive Plasmodium falciparum test and independent variables for those that had a significant association by controlling for potential confounders. After multivariate logistic regression, presence of mosquito breeding site (AOR = 21.89, 95% CI: 5.25-121.13), and no utilization of ITN (AOR = 10.82, 95% CI: 3.13-40.37) remained positively associated with positive Plasmodium falciparum test. Table 3 summarizes the results of multivariate logistic regression.
Summary of findings: in this cross-sectional analytical study, we set out to determine the prevalence and associated risk factors for positive Plasmodium falciparum test by rapid diagnostic testing in pregnant women in the North West region of Cameroon. We obtained a prevalence of 18.4% with positively associated factors being: living in a rural area, not using insecticide-treated nets, not using indoor residual spraying, mothers in the first and second trimesters of gestation compared to third trimester, and the presence of a mosquito breeding site.
Prevalence rate of Plasmodium falciparum amongst pregnant women: we found a high prevalence of positive Plasmodium falciparum antigen amongst pregnant women attending ANC at the Bamenda Regional Hospital. This is an alarm that malaria is still a major public health problem, especially amongst pregnant women attending ANC at the BRH. Our finding is similar to that of studies conducted in Some parts of Cameroon (21% and 19.3%) [18,19], and other countries including Burkina Faso (18.1%) [8], and Malawi (19.6 %) [9]. The similarities could be due to similar study populations and study design. However, some studies have reported prevalence that are lower than ours such as in Rwanda (13.6%) [20], Ghana (5%) [21], Laos (8.3%) [22], Sudan (13.7%) [7], Nigeria (7.7%) [6] and India (5.4%) [23]. This difference could be attributed to the variations in urbanization, and time-periods during which the studies were carried-out. For instance, the low prevalence rate in the study in India may be due to better urbanization and low distribution of the Plasmodium falciparum parasite. This discrepancy might also result from the fact that, in our study, we used RDT which can detect the presence of the antigen several days after parasite clearance and therefore cannot be used to distinguish current from previous infection. Also, we included both symptomatic and asymptomatic pregnant women unlike most other studies. On the other hand, the prevalence in our study was found to be lower than that of a study conducted in Mamfe Health District of Cameroon (39.4%) [17], and three studies in Nigeria (41.6%; 58%; 59.9%) [5,24,25]. This discrepancy could be attributed to the fact that in our study, two thirds of the mothers had received IPT and 3 out of 4 mothers owned an insecticide impregnated treated mosquito net. Another reason for the low prevalence in our study could be the fact that our study was done during the low malaria transmission season.
Factors associated with positive Plasmodium falciparum antigen: the current study established that living in a rural area was associated with a higher risk of positive Plasmodium falciparum antigen. This is similar to findings from the study by Deressa Wet al. [26]. This finding however does not coincide with what was found in the study done by Erhabo et al. [27]. Their study was conducted in a specialized hospital and therefore their study population was different and could explain the discrepancy. According to our study, pregnant women who were in the first and second trimester of pregnancy were at a higher risk of developing malaria infection compared to pregnant women in the third trimester. This is similar to research done by Gontie et al. [28]. This is explained by the fact that pregnant women in first and second trimester lack the specific immunity to placental malaria that is acquired from exposure to malaria parasites during pregnancy. Pregnant women living near mosquito sites were found to be over 20 times more likely to have a positive Plasmodium falciparum test compared to those living in areas without mosquito breeding sites after adjusting for possible confounders. Similar observations have been reported by other researchers such as Elime FA et al. [17] in Cameroon and Patricia M Graves et al. [29] in Ethiopia. This finding strongly supports the need for environmental sanitation, especially amongst pregnant women. Our study showed that not using vector targeting strategies such as ITN and IRS was associated with increased odds of having a positive Plasmodium falciparum antigen test. Findings from previous studies such as a study in Malawi [9], Nigeria [1] and Cameroon [17] support the increased odds of positive test without use of ITN. A study by Gontie et al.[28] supported the association of positive test with the absence of use of IRS. Vector targeting strategies such as use of ITN and IRS are major public health actions to combat malaria infection as they effectively reduce human-mosquito contact and hence prevent infective bites from the mosquito. Surprisingly, our study did not show any statistically significant association between use of IPT and positive Plasmodium falciparum test. This could probably be explained by the fact that in our study we grouped all those who had taken one dose or two doses or three doses of IPT into the same category and did not do subgroup analyses to check for association with increasing IPT doses.
Limitation and strengths of our study: our study is limited in that it is a cross-sectional study and temporality cannot be ascertained. To assure the internal validity of our study, we minimized reported bias by using antenatal clinic cards to confirm participants� information, and we also ran two test per participant to ensure reliability of our test results. Also, the choice of the Bamenda regional hospital which is a public hospital and thus open to persons of different works of life in Bamenda adds strength to the internal validity, hence our study findings could be generalized to the entire North West region of Cameroon.
The prevalence of positive Plasmodium falciparum antigen is high amongst pregnant women in the BRH with a prevalence rate of 18.4%. Numerous risk factors are associated with positive Plasmodium falciparum amongst pregnant women including: the presence of mosquito breeding sites, no usage of ITN, no usage of IRS, living in rural area, and pregnant women in the first and second trimester of pregnancy. Clinicians working in ANC should therefore reinforce preventive strategies against malaria such as IRS, ITN, and environmental hygiene.
What is known about this topic
- Malaria is endemic in Cameroon;
- The prevalence of malaria in pregnant women is high.
What this study adds
- We obtained the prevalence rate of Plasmodium falciparum among pregnant women which despite continuous efforts to fight malaria infection, still remains high;
- Our study adds evidence to the growing literature to support malaria preventive strategies amongst pregnant women.
The authors declare no competing interest.
Conception and design: Dobgima Walter Pisoh, Joseph Bakowe, Sama Julius Dohbit. Acquisition of data: Dobgima Walter Pisoh, Joseph Bakowe, Achuo Ascensius Ambe Mforteh. Analysis and interpretation of data: all authors. Drafting the manuscript: Dobgima Walters Pisoh, Achuo Ascensius Ambe Mforteh, William Ako Takang. Revising the manuscript critically for important intellectual content: all authors. Final approval of the version to be published: Dobgima Walter Pisoh, Achuo Ascensius Ambe Mforteh,William Ako Takang, Sama Julius Dohbit. All authors have read and agreed to the final manuscript.
We are grateful to all the pregnant women who took part in this study. We also thank the midwives and nurses of the BRH antenatal clinic for their support during the data collection period.
Table 1: characteristics of the study population
Table 2: association between factors and positive Plasmodium falciparum test (bivariate analysis)
Table 3: multivariate logistic regression of factors associated with positive Plasmodium falciparum test
- Fana SA, Bunza MD, Anka SA, Imam AU, Nataala SU. Prevalence and risk factors associated with malaria infection among pregnant women in a semi-urban community of Northwestern Nigeria. Infect Dis Poverty. 2015 Apr 24:4:24. PubMed | Google Scholar
- Riley EM, Hviid L, Theander TG. Malaria. In: Kierszenbaum F, editor. Parasitic Infections and the Immune System. 1st ed. New York: Academic Press; 1994. P.119-43.
- World Health Organization. World malaria report 2017. Geneva: WHO. 2017 Nov:19- 196p.
- World Health Organization. World malaria report 2016. Geneva: WHO. 2016 NOV:186p.
- Kagu M, Kawuwa M, Gadzama. Anaemia in pregnancy: a cross-sectional study of pregnant women in a Sahelian tertiary hospital in northeastern Nigeria. J Obstet Gynaecol. 2007 Oct;27(7):676-9. PubMed | Google Scholar
- Agomo CO, Oyibo WA, Anorlu RI, Agomo PU. Prevalence of malaria in pregnant women in Lagos, South-West Nigeria. Korean J Parasitol. 2009 Jun;47(2):179-83. PubMed | Google Scholar
- Adam I, Khamis AH, Elbashir MI. Prevalence and risk factors for plasmodium falciparum malaria in pregnant women of eastern Sudan. Malar J. 2005 Apr 13:4:18. PubMed | Google Scholar
- Cisse M, Sangare I, Longue G, Bamba S, Bayane D, Guigemde RT. Prevalence and risk factors for plasmodium malaria in pregnant women attending antenatal clinic in Bobo- Dioulasso (Burkina Faso). BMC Infect Dis. 2014 Nov 19:14:631. PubMed | Google Scholar
- Boudova S, Divala T, Mawindo P, Cohee L, Kalilani-Phiri L, Thesing P et al. The Prevalence of malaria at first antenatal visit in Blantyre, Malawi declined following a universal bed net campaign. Malar J. 2015 Oct 29:14:422. PubMed | Google Scholar
- Jäckle MJ, Blumentrath CG, Zoleko RM, Akery-Diop D, Mackanga JR, Adegnika AA et al. Malaria in pregnancy in rural Gabon: a cross-sectional survey on the impact of seasonality in high-risk groups. Malar J. 2013 Nov 13:12:412. PubMed | Google Scholar
- World Health Organization. Guidelines for the Treatment of malaria. Geneva: WHO. 2015 Aug 13. PubMed | Google Scholar
- Rogerson SJ, Boeuf P. New approaches to malaria in pregnancy. Parasitology. 2007;134(Pt 13):1883-93. PubMed | Google Scholar
- Asmamaw T, Alemu A, Alemu A, Unakal C. Prevalence of malaria and HIV among pregnant women attending antenatal clinics at felege hiwot referral hospital and Addis zemen health center. Int J Life Sci Biotechnol pharma Res. 2013; 2:1-3. Google Scholar
- Fried M, Muehlenbachs A, Duffy PE. Diagnosing malaria in pregnancy: an update. Expert Rev Anti Infect Ther. 2012 Oct;10(10):1177-87. PubMed | Google Scholar
- Ezeudu CE, Ebenebe JC, Ugochukwu EF, Chukwuka JO, Amilo GI, Okorie OI. The performance of an Histidine rich protein-2 rapid diagnostic test (RDT) against the standard microscopy in the diagnosis of malaria parasitaemia among febrile under five children at Nnewi. Niger J Paed. 2015;42(1):59-63. Google Scholar
- Ding, XC, Incardona S, Serra-Casas E, Charnaud SC, Slater HC, Domingo GJ et al. Malaria in pregnancy (MiP) studies assessing the clinical performance of highly sensitive rapid diagnostic tests (HS-RDT) for Plasmodium falciparum detection. Malar J. 2023 Feb 20;22(1):60. PubMed | Google Scholar
- Elime FA, Nkenyi NR, Ako-Egbe L, Njunda A, Nsagha D. Malaria in pregnancy: prevalence and risk factors in the Mamfe Health District, Cameroon. JAMMR. 2019 Jul 6;30(1):1-1.
- Nkuo-Akenji T, Ntonifor NN, Ndukum MB, Abongwa EL, Nkwescheu A, Anong DN et al. Environmental factors affecting malaria parasite prevalence in rural Bolifamba, South West Cameroon. Afri J Health Sci. 2006;13(1-2)40-6. PubMed | Google Scholar
- Sumo L, Mbah NE, Nana-Djeunga CH. Malaria in pregnancy in the Ndop Health District (North West Region, Cameroon): results from retrospective and prospective surveys. J Parasitol vector Bio. 2015 Oct 31;7(9):177-81. Google Scholar
- Van Geertruyden JP, Ntakirutimana D, Erhart A, Rwagacondo C, Kabano A, D'Alessandro U. Malaria infection among pregnant women attending antenatal clinics in six Rwandan districts. Trop Med Int Health. 2005 Jul;10(7):681-8. PubMed | Google Scholar
- Steffen R, Debernardis C, Baños A. Travel epidemiology: a global perspective. Int J Antimicrob Agents. 2003 Feb;21(2):89-95. PubMed | Google Scholar
- Briand V, Le Hesran JY, Mayxay M, Newton PN, Bertin G, Houze S et al. Prevalence of malaria in pregnancy in southern Laos: a cross-sectional survey. Malar J. 2016 Aug 26;15(1):436. PubMed | Google Scholar
- Sohail M, Shakeel S, Kumari S, Bharti A, Zahid F, Anwar S et al. Prevalence of malaria infection and risk factors associated with Anaemia among pregnant women in Semiurban Community of Hazaribag, Jharkhand, India. Biomed Res Int. 2015:2015:740512. PubMed | Google Scholar
- Ifeanyi U, Onyebuchi V, Ukamaka T, Ugochukwu B. Asymptomatic Plasmodium parasitaemia in pregnant Nigerian women: almost a decade after Roll Back malaria. Trans R Soc Trop Med Hyg. 2009 Jan;103(1):16-20 PubMed | Google Scholar
- Ogbodo SO, Nwagha UI, Okaka AN, Ogenyi SC, Okoko RO, Nwagha, T. Malaria parasitaemia among pregnant women in a rural community of eastern Nigeria; need for combined measures. Niger J Physiol Sci. 2009 Dec;24(2):95-100. PubMed | Google Scholar
- Deressa W, Ali A, Hailemariam D. Malaria-related health-seeking behaviour and challenges for care providers in rural Ethiopia: implications for control. J Biosoc Sci. 2008 Jan;40(1):115-35. PubMed | Google Scholar
- Osaro E, Abdullahi A, Tosan E, Adias TC. Risk factors associated with malaria infection among pregnant women of African Descent in Specialist Hospital Sokoto, Nigeria. Obstet Gynecol Int J. 2019;10(4):274-280. Google Scholar
- Gontie GB, Wolde HF, Baraki AG. Prevalence and associated factors of malaria among pregnant women in Sherkole District, Benishangul Gumuz Regional State, West Ethiopia. BMC Infect Dis. 2020 Aug 5;20(1):573. PubMed | Google Scholar
- Graves PM, Richards FO, Ngondi J, Emerson PM, Shargie EB, Endeshaw T et al. Individual, household and environmental risk factors for malaria infection in Amhara, Oromia and SNNP regions of Ethiopia. Trans R Soc Trop Med Hyg. 2009 Dec;103(12):1211-20. PubMed | Google Scholar











