Foot-at-risk among adult outpatients with diabetes mellitus in Bowen University Teaching Hospital, Ogbomoso, Nigeria
Adeola Ebenezer Idowu, Isaac Olusayo Amole, Adewumi Ojeniyi Durodola, Stephen Adesope Adesina, Olatayo Araade Idowu, Adepeju Olatayo Adegoke, Philip Bamigboye, Olufemi Timothy Awotunde, Akintayo David Ola Olorun
Corresponding author: Adeola Ebenezer Idowu, Department of Family Medicine, Bowen University Teaching Hospital, P.O. Box 15, Ogbomoso, Nigeria 
Received: 04 Mar 2023 - Accepted: 09 Nov 2023 - Published: 14 Dec 2023
Domain: Diabetes care,Family Medicine
Keywords: Foot-at-risk, neuropathy, peripheral arterial disease, glycemic control
©Adeola Ebenezer Idowu et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Adeola Ebenezer Idowu et al. Foot-at-risk among adult outpatients with diabetes mellitus in Bowen University Teaching Hospital, Ogbomoso, Nigeria. Pan African Medical Journal. 2023;46:106. [doi: 10.11604/pamj.2023.46.106.39397]
Available online at: https://www.panafrican-med-journal.com//content/article/46/106/full
Research 
Foot-at-risk among adult outpatients with diabetes mellitus in Bowen University Teaching Hospital, Ogbomoso, Nigeria
Foot-at-risk among adult outpatients with diabetes mellitus in Bowen University Teaching Hospital, Ogbomoso, Nigeria
![]() Adeola Ebenezer Idowu1,&,
Adeola Ebenezer Idowu1,&, ![]() Isaac Olusayo Amole2,
Isaac Olusayo Amole2, ![]() Adewumi Ojeniyi Durodola2,
Adewumi Ojeniyi Durodola2, ![]() Stephen Adesope Adesina2,
Stephen Adesope Adesina2, ![]() Olatayo Araade Idowu1,
Olatayo Araade Idowu1, ![]() Adepeju Olatayo Adegoke2, Philip Bamigboye1,
Adepeju Olatayo Adegoke2, Philip Bamigboye1, ![]() Olufemi Timothy Awotunde2,
Olufemi Timothy Awotunde2, ![]() Akintayo David Ola Olorun2
Akintayo David Ola Olorun2
&Corresponding author
Introduction: the rising prevalence of diabetes mellitus (DM) around the world has dramatically increased the number of people bearing the complications of this potentially incapacitating disease. One of these complications is foot ulcers that may result in amputation. This study sets out to determine the profiles of the “foot-at-risk” for ulceration and the associated socio-medical factors in DM patients.
Methods: this study was conducted at Bowen University Teaching Hospital, Ogbomoso, Southwest, Nigeria. This was a descriptive cross-sectional study comprising 299 outpatient adults aged 18 years and above with diabetes mellitus of at least 6 months in duration. Comprehensive Foot Examination and Risk Assessment tool was used to identify the foot-at-risk categories of the participants. Data analysis was done using Statistical Package for Social Sciences (SPSS) version 20.
Results: the prevalence of foot-at-risk among the participants was 64.9% (194). Among the 194 participants with foot-at-risk, 35.1% (105) belonged to the foot-at-risk categories 0, 37.8% (113) in category 1, 16.1% (48) in category 2, and 11.0% (33) in category 3. Other factors that had a statistically significant association with foot-at-risk included; age, religion, level of education, duration of diabetes, history of smoking, and glycemic control.
Conclusion: foot-at-risk was found to have an alarming prevalence among the participants. In addition, the level of glycemic control in this group was unacceptably poor. Clinicians need to intensify preventive measures like foot screening and health education to prevent foot ulcerations, which may result in limb amputation in DM patients.
Diabetes mellitus (DM) is fast assuming pandemic with alarming increase in its prevalence rate [1]. The rising prevalence of diabetes around the world has dramatically increased the number of people bearing its complications. One of such complications is the diabetic foot disease (DFD) [2]. It is defined as a foot affected by ulceration that is associated with neuropathy and/or peripheral arterial disease of the lower limb in a patient with diabetes. The burden of DFD is so intense that one person undergoes amputation every 30 seconds around the globe [3].
Mortality rates after diabetic foot ulceration and amputation are high, with up to 70% of people dying within 5 years of having an amputation and around 50% dying within 5 years of developing a diabetic foot ulcer [4]. Up to 85% of these amputations can be prevented with adequate measures [5]. It is therefore necessary that screening of patients with DM to identify foot-at-risk of ulceration and associated risk factors could be instituted to reduce the risk of foot ulceration and foot amputation [4]. Risk identification is fundamental for effective preventive management of the foot in people with DM [6]. This involves identifying foot-at-risk of ulceration as well as assessment of their glycaemic control. For the purpose of this study, “foot-at-risk” refers to the foot in DM patient with intact skin which may have bony or soft tissue deformities, features of neuropathy and/or vasculopathy, and a history of previous ulceration and/or amputation [6]. It is important to note that when appropriate actions are instituted after identification of foot-at-risk of ulceration, the rate of DFD will significantly reduce.
Unfortunately, according to our research, there has not been any recent report on the foot-at-risk in the Southwestern part of Nigeria. Hence, this study was conducted to assess the profile of foot-at-risk and associated socio-medical factors in diabetic patients accessing outpatient care in Bowen University Teaching Hospital, Ogbomoso, South-west Nigeria.
Setting: the study was conducted in Bowen University Teaching Hospital (BUTH), Ogbomoso, South-west Nigeria. It was formerly called Baptist Medical Centre and was established in 1907 and upgraded to a teaching hospital in 2009. It has over 400 beds and the hospital sees over 50,000 outpatients and 10,000 in-patients per year.
Study design: this was a cross-sectional study conducted between March and June 2018. Systematic sampling technique was used for the selection of participants and 299 participants were recruited for the study.
Study size determination: the sample size for the study was calculated using the statistical formula [7].
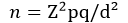
Where: n = desired sample size, Z = Two standard deviations usually set at 1.96 which corresponds to 95% confidence level. p = the proportion in the target population estimated to have a particular characteristic. q = the proportion of the population not involved in the study i.e. 1-p. d = the degree of accuracy desired usually set at 0.05. Using the prevalence of diabetic foot-at-risk of 41.5% in South-west Nigeria [6]. Therefore, n = 373.1 approximated to 373.
However, the population of patients with diabetes mellitus that was seen in the last one year from the records department was 1200. Since the study population is < 10,000, the sample size was adjusted using the formula; [7].
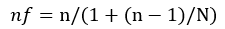
Where: nf = Desired sample size when population is less than 10,000. n = Desired sample size when population is greater than 10,000. N = Estimate of the population size = 1200. Therefore, nf = 373/ (1+ (373-1)/1200) � 285. An allowance of 5% (14.3) was given for poorly completed questionnaire and missing test results. This was added to the desired sample size to give a total of 299.
Study participants and eligibility criteria: consenting patients aged 18 years and above with diabetes mellitus that have been on treatment for at least 6 months were recruited for the study. Pregnant patients, cognitively impaired patients and patients with active foot ulcers were excluded from the study.
Variables: for each of the recruited subjects, data collected were: socio-demographic and medical characteristics which included age, gender, marital status, religion, level of education, social class, ethnicity, residence, smoking history, duration of DM and comorbid medical illness; physical examination using comprehensive foot examination and risk assessment used to identify and categorize foot at risk; venous blood sample was taken for HbA1C measurement (glycaemic control) using A1CNow®+ system.
Data sources/measurement: the comprehensive foot examination and risk assessment was used for identification and categorization of foot-at-risk. It is a validated tool for categorizing foot-at-risk in patient with DM [8]. It included three (3) criteria which were; Foot inspection - for identifying foot deformities, Neurologic assessment - for identifying loss of protective sensation (LOPS) and Vascular assessment - for identifying peripheral arterial disease (PAD).
Foot inspection: the feet of participants were inspected under bright light for the presence of foot deformities and callous.
Neurologic assessment: a 10-g Semmes-Weinstein monofilament test and 128-Hz tuning fork test were used to identify LOPS as recommended by ADA [9]. At least one abnormal test suggested LOPS. The 10-g Semmes-Weinstein monofilament (manufactured by Atlas Biomechanics, Scottsdale, USA) was used to test for pressure perception. This was done in four sites (1st, 3rd, and 5th metatarsal heads and plantar surface of the distal hallux) each on both feet. With patient´s eyes opened, the device was placed perpendicular to the skin of the palm with pressure applied until the monofilament buckles (it was held in place for ~1 s and then released) after the sensation of pressure has been recognized by patient, the sensation in the foot was tested with patient´s eyes closed. Patient was expected to respond “yes” when the monofilament was applied to a particular site and also to identify the correct site of placement. The vibration test was done by using a 128-Hz tuning fork. Vibration perception was tested over the bony prominence situated at the dorsum of the foot corresponding to the head of the first metatarsal. With the patient lying supine and eyes closed, the stylus of the tuning fork was placed over the dorsal hallux. This process was initially demonstrated on the upper limb for the patient to appreciate the buzzing sensation.
Vascular assessment: the ankle-brachial index (ABI) measurement was performed by measuring the systolic blood pressure from both brachial arteries and from both posterior tibial arteries after the patient has been at rest in the supine position for 10 minutes. The systolic pressures were recorded with a hand-held 8-mHz Vascular Doppler (True Sonotrax Vascular Doppler manufactured by Edan Instrument Inc, China) and Accoson® mercury sphygmomanometer manufactured by Dekamet Accoson, England with appropriate cuff size. In measuring the brachial pressure, the patient was placed in supine position and blood pressure cuff placed around the arm. Ultrasound gel was applied to the antecubital fossa over the brachial pulsation. The transducer of the vascular Doppler was placed on the gel and its position was adjusted to the area with maximum signal intensity. The cuff of the sphygmomanometer was inflated to about 20mmHg above the expected systolic blood pressure of the patient. Then, the cuff was slowly deflated at approximately 2mmHg/sec until the Doppler signal reappeared. The pressure at which the Doppler signal reappeared was recorded as the brachial systolic pressure. Same procedure was repeated for the second arm and the highest brachial systolic pressure was used for ABI calculation [10].
In measuring the ankle pressure, the cuff was placed mid-way between the calf muscle and the malleolus. Ultrasound gel was applied to the posterior aspect of the medial malleolus. The transducer of the vascular Doppler was placed on the gel and its position was adjusted to the area with maximum signal intensity. The cuff of the sphygmomanometer was inflated to about 20mmHg above the expected systolic blood pressure of the patient. Then, the cuff was slowly deflated at approximately 2mmHg/sec until the Doppler signal reappears. The pressure at which the Doppler signal reappears was recorded as the ankle systolic pressure. Same procedure was repeated for the second ankle and the highest ankle systolic pressure was used for ABI calculation [10].
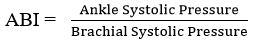 . Calculated ABI value was recorded to two decimal places.
. Calculated ABI value was recorded to two decimal places.
Data analysis: data analysis was done using Statistical Package for Social Sciences (SPSS) for Windows, version 20. Descriptive statistics was used to generate frequency table to determine the distribution of categories of foot-at-risk. The associations of foot-at-risk with the categorical variables were tested using either chi-square test or fisher's exact test. P-value was set at 0.05 (P-value = 0.05 was regarded as statistically significant).
Operational definitions
Glycaemic control: good glycaemic control implied glycosylated haemoglobin lesser than 7% while a value of = 7% was considered poor glycaemic control [11].
Loss of protective sensation (LOPS) was considered to be present when a patient has at least one abnormal test from the 10-g monofilament test and vibration test. The 10-g monofilament test was considered abnormal if total sensation from both feet is < 7/8. Vibration test was considered abnormal if there was absent sensation in at least one foot.
Definition of PAD was made by using ABI. It was considered to be present if ABI is either < 0.91 or > 1.30 [12]. Foot was considered to be deformed if there was presence of any of these; prominent metatarsals, callus, claw toes, hallux valgus, hammer toes and high arched feet. Participants were categorized into four categories as illustrated in Table 1.
Ethical consideration: the study protocol was reviewed by the ethical committee of Bowen University Teaching Hospital, Ogbomoso and ethical approval was obtained before the study commenced. The study participants were required to sign the informed consent after the objectives and the procedure of the study were explained to them. All the consent forms and the questionnaires were kept confidential and used only for the study. This study protocol complied with the Declaration of Helsinki.
Socio-demographic and medical characteristics of the participants: the age group with the highest proportion of participants was age above 60 years with 41.8% (125) of the participants while the group below 30 years was the lowest with a frequency of 1.3% (4). Only 39.1% (117) of the participants were males and 60.9% (182) were females, giving a male to female ratio of 0.6:1. Majority of the participants (274) were married. Christianity was the more practiced religion with a frequency of 76.3% (228). The largest proportion of the participants (285) belonged to Yoruba ethnic group. About 67.9% (203) of the respondents were in class 2 and majority of the participants (208) were urban dwellers. About 67.2% (201) of the participants had duration of diabetes less than 5 years. Among the participants, 51.5% (154) had co-existing medical illness. Of the co-existing medical conditions in the participants, hypertension ranked the highest with a prevalence of 43.5% (130). Other conditions reported included; osteoarthritis (5.7% (17)), asthma (1.0% (3)) and visual impairment (1.3% (4))Table 2.
Category of foot-at-risk in the participants: majority of the participants (37.8% (113)) belonged to category 1 of foot-at-risk while the minimum number fell into category 3 with a frequency of 11.0% (33). Therefore, the prevalence of foot-at-risk in the study population was 64.9% (194) (Table 3).
Prevalence of the implicated risk factors: the prevalence of foot deformity, peripheral neuropathy and peripheral arterial disease were 30.8% (92), 37.8% (113), and 16.1% (48) respectively. Callus accounted for the highest form of foot deformity in participants (16.4% (49)). Most of the participants had a poor glycaemic control with a frequency of 83.6% (250). The mean level of glycosylated haemoglobin (HbA1C) in this study was 9.2 ± 2.5%. The values of HbA1C of participants in this study ranged between 4.6 to 21% (Table 4).
Factors associated with foot-at-risk: Table 5 showed the associations of foot-at-risk with age group, religion, level of education, duration of care, smoking history and glycaemic control were found to be statistically significant.
The age group above 60 years had the highest proportion of respondents, which was 41.8% while the age group below or equal to 30 years was the lowest, which was 1.3%. The minimum age in the participants was 30 years while the maximum was 85 years and the mean age was 59.3 ± 11.2 years. The age distribution of the respondents in this study illustrated that majority of the participants with DM are usually in the older age group since the risk of Type 2 DM increases with age. The age distribution finding in this study aligned with other related studies [6,13-16].
Majority of the respondents were females with a prevalence of 60.9% and a male-to-female ratio of 0.6:1. The larger proportion of female in the study reflected the positive attitude of females to seeking health care. However, the report by Ogbera et al. [6]. revealed that males were more than females (1:0.97). This may be attributed to unhealthy lifestyle lived by urban male dwellers because the study was conducted in Lagos. In this study, 76.3% of the respondents were Christians. Ogbomoso is generally known to be a Christian community that might have accounted for the above finding. However, previous similar studies did not emphasize this and this may be important in foot-at-risk and foot care practice. It can be seen that 51.5% of the respondents had co-existing medical conditions. The identified medical conditions included; hypertension, asthma, visual impairment and osteoarthritis. However, the condition with the highest proportion was hypertension which was seen in 43.5% of the participants. In this study, 67.2% of the respondents had duration of DM treatment less than 5 years while a smaller percentage (16.7%) had duration of treatment over 10 years. The shorter duration of treatment seen in the respondents may be due to late presentation to the hospital for diagnosis and treatment. The late presentation may also be attributed to the cultural perception of patients and the low level of education reported earlier. However, Ogbera et al. [6] reported that 40.6% of the respondents had medium term DM duration (5-9 years). This may be due to higher level of education seen in urban dwellers. More than half of the participants had foot-at-risk with a prevalence of 64.9%. This prevalence was distributed across the different categories with the highest in category 1. The distribution included; 35.1% in category 0, 37.8% in category 1, 16.1% in category 2, and 11% in category 3. In this study, a high prevalence of foot-at-risk may be due to the increase in the prevalence of diabetes with resultant increase in its complications. Conversely, Ogbera et al. [6] in a study done in Lagos to assess the level of foot-at-risk, reported the prevalence of foot-at-risk as 41.5%. However, the foot-at-risk was only identified but not categorized. This may be because the study had earlier been done before the comprehensive foot examination and categorization was released in 2008. Other similar studies reported a prevalence of 41.4% with category 3 being the highest (17.3%) [15]; 52% majority of the participants belonged to category 0 (48%) [17]; 15.3% with majority in category 1 [13].
As earlier highlighted, the common risk triad in the development of foot ulceration in DM patients is peripheral neuropathy, peripheral arterial disease and foot deformity [18]. Peripheral neuropathy was seen in 37.8% of the participants. The level of peripheral neuropathy seen in the study population may be because the majority of the participants belonged to age range over 60 years. A similar prevalence was reported by Ogbera et al. [12]. In addition, the level of peripheral neuropathy reported in this study and others may be due to poor glycaemic control as well as coexisting conditions like hypertension. Peripheral arterial disease was assessed with a vascular Doppler in this study. The prevalence of PAD in this study was 16.1%. However, Ogbera et al. [12] reported PAD prevalence as 40%. A vascular Doppler with frequency of 12mHz was used to assess PAD contrary to the conventional 8mHz used for this study. So, this may be responsible for higher prevalence of PAD reported. Kishore et al. [17] reported a prevalence of 10% for PAD. The varying levels of PAD in the reported studies may be related to the poor metabolic control of the participants and other factors like, coexisting conditions, assessment tools, assessors' expertise and so on. The prevalence of foot deformity in this study was 30.8%. Among the study participants, callus accounted for 16.4%, hammer toe accounted for 3.7%, claw toes accounted for 2.0%, hallux valgus was seen in 7.7% and 1.0% had high-arched feet. The higher prevalence of callus seen may be due to the advanced age of the participants, the geographical location of the study area and the social class of the participants.
There was an unacceptable poor glycaemic control in participants of as low as 16.4% good control level. The mean glycaemic control was 9.2%. A larger portion of participants with good glycaemic control were located in category 1 of foot-at-risk with a percentage of 44.9%. In addition, the group with worst glycaemic control was category 3 with none of the participants with good control. In other words, there was a trend between the increasing severity of the category of foot-at-risk and glycaemic control. The association between foot-at-risk and glycaemic control was statistically significant (P-value = 0.001). As earlier noted, the prevalence of foot-at-risk in this study was 64.9%, the poor glycaemic control among participants might have accounted for the high prevalence of foot-at-risk. Consistent with the present study, several studies have reported similar outcome [6,12,19]. The proportion of foot-at-risk in all categories increased with age. Majority of those with foot-at-risk belonged to the age group above 60 years. The association of foot-at-risk with age was statistically significant (P-value = 0.047). In other words, increase in age has the tendency to increase the risk of foot ulcer. The above finding may be due to the risk of development of both neuropathy and arterial disease with advancing age. This assumption was supported by Ogbera et al. [12] that reported that older age of > 60 years and poor glycaemic control were potential predictors of neuropathy.
Majority of the study participants (67.2%) had duration of diabetes mellitus less than 5 years. Among the participants with diabetes less than 5 years, 61.8% of them had foot-at-risk. The association of foot-at-risk with duration of diabetes was found to be statistically significant (P-value = 0.001). Similarly, Shahbazian et al. [20] in Iran reported that there was statistically significant association between higher foot-at-risk and longer diabetes duration (P-values = 0.001). Alam et al. [21] reported that longer duration of diabetes greater than 10 years was a significant risk factor in the development of diabetic foot complications. Other factors that were found to have statistically significant association with foot-at-risk included; religion (P-value = 0.001), level of education (P-value = 0.002), and history of smoking (P-value = 0.029). Few Muslims had foot-at-risk from this study. This may be associated with the good foot care activities practiced by the religion. The level of education of the patients may affect both the understanding of foot care education, foot care practice, and their glycaemic control activities.
Study limitation: since this study was hospital-based and not multicenter study, with a low response from other ethnic groups in the country the result may not be representative of what obtains in the general population.
This study confirmed the alarming burden of foot-at-risk in patients with diabetes mellitus attending the outpatient clinics in Bowen University Teaching Hospital, Ogbomoso. Majority of the patients in this study population are at considerable risk of developing foot ulcers. In view of this, foot screening should be done by all clinicians routinely for DM patients. In addition, patients identified should be managed and followed up appropriately [9]. One of the important factors in the genesis of DFD is poor glycaemic control. Unfortunately, the long-term glycaemic control of the participants was unacceptably poor. This finding showed that there is need to intensify efforts targeting good glycaemic control in the patients with DM in order to prevent complications from the disease. The socio-medical factors associated with foot-at-risk in this study were age, religion, level of education, duration of diabetes, and history of smoking.
What is known about this topic
- Foot-at-risk has been known to be the predecessor of diabetic foot disease, which may progress to foot amputation; as shown in different classification of diabetic foot;
- The burden of foot-at-risk is high in South-west, Nigeria as reported by Ogbera et al [6].
What this study adds
- Even though foot-at-risk is known to precede diabetic foot, researchers have only concentrated on DM foot studies. In addition, there is paucity of data on foot-at-risk in South-west, Nigeria, the few available studies are old and standardized tools were not used for assessment of foot-at-risk;
- This study reviewed the current profile of foot-at-risk in DM patients in the South-western, Nigeria using a standardized tool;
- This study emphasized on the need for proactive actions by both clinician and DM patients towards prevention of DM foot.
The authors declare no competing interest.
This work was carried out in collaboration among all authors. Adeola Ebenezer Idowu designed the study and wrote the protocol. Isaac Olusayo Amole, Adewumi Ojeniyi Durodola, Stephen Adesope Adesina, Adepeju Olatayo Adegoke, Philip Bamigboye collected the data. Isaac Olusayo Amole, Olufemi Timothy Awotunde and Akintayo David Ola Olorun analyzed and interpreted the data. Adeola Ebenezer Idowu, Isaac Olusayo Amole, Adewumi Ojeniyi Durodola, Stephen Adesope Adesina, Olatayo Araade Idowu, Adepeju Olatayo Adegoke, Philip Bamigboye, Olufemi Timothy Awotunde, Akintayo David Ola Olorun, Philip Bamigboye, and Olatayo Araade Idowu wrote the first draft of the manuscript. Stephen Adesope Adesina, Adewumi Ojeniyi Durodola, Adepeju Olatayo Adegoke, Olufemi Timothy Awotunde and Akintayo David Ola Olorun revised the manuscript. All authors read and approved the final manuscript.
Table 1: categories of foot-at-risk
Table 2: socio-demographic and medical characteristics of the participants (N = 299)
Table 3: category of foot-at-risk in the participants
Table 4: foot-at-risk factors in the participants (N = 299)
Table 5: socio-medical factors associated with foot-at-risk in the participants (N = 299)
- Dahiru T, Aliyu AA, Shehu AU. A review of population-based studies on diabetes mellitus in Nigeria. Sub-Saharan Afr J Med. 2016;3(2):59-64. Google Scholar
- Parisi MC, Moura Neto A, Menezes FH, Gomes MB, Teixeira RM, de Oliveira JE et al. Baseline characteristics and risk factors for ulcer, amputation and severe neuropathy in diabetic foot at risk: the BRAZUPA study. Diabetol Metab Syndr. 2016;8(1):25-35. PubMed | Google Scholar
- Desalu OO, Salawu FK, Jimoh AK, Adekoya AO, Busari OA, Olokoba AB. Diabetic foot care: self reported knowledge and practice among patients attending three tertiary hospital in Nigeria. Ghana Med J. 2011;45(2):60-65. PubMed | Google Scholar
- NICE. Diabetic foot problems: prevention and management. NICE Guidel 2015. Cited 2018 Jun 2.
- Ugwu E, Adeleye O, Gezawa I, Okpe I, Enamino M, Ezeani I. Burden of diabetic foot ulcer in Nigeria: current evidence from the multicenter evaluation of diabetic foot ulcer in Nigeria. World J Diabetes. 2019;10(3):200-11. PubMed | Google Scholar
- Ogbera AO, Adedokun A, Fasanmade OA, Ohwovoriole AE, Ajani M. The foot at risk in Nigerians with diabetes mellitus-the Nigerian scenario. Int J Endocrinol Metab. 2005;4(1):165-73. PubMed | Google Scholar
- Araoye MO. Research methodology with statistics for health and social sciences. Ilorin: Nathadex Publishers. 2004.
- Monteiro-Soares M, Vaz-Carneiro A, Sampaio S, Dinis-Ribeiro M. Validation and comparison of currently available stratification systems for patients with diabetes by risk of foot ulcer development. Eur J Endocrinol. 2012;167(3):401-7. PubMed | Google Scholar
- Boulton AJ, Armstrong DG, Albert SF, Frykberg RG, Hellman R, Kirkman MS et al. Comprehensive foot examination and risk assessment. Diabetes Care. 2008;31(8):1679-85. PubMed | Google Scholar
- Aboyans V, Lacroix P. Teaching and evaluating measurement of the ankle - brachial index. Vasc Med. 2018;23(2):114-5. PubMed | Google Scholar
- American Diabetes Association. Standards of medical care in diabetes - 2017. J Clin Appl Res Educ. 2017;40(suppl.1):1-142. Google Scholar
- Ogbera AO, Adeleye O, Solagberu B, Azenabor A. Screening for peripheral neuropathy and peripheral arterial disease in persons with diabetes mellitus in a Nigerian university teaching hospital. BMC Res Notes. 2015;8(1):533-9. PubMed | Google Scholar
- Tshitenge S, Ganiyu A, Mbuka D, Shama JM. The diabetic foot risks profile in Selebi Phikwe Government Hospital, Botswana. African J Prim Heal Care Fam Med. 2014;6(1):6-10. PubMed | Google Scholar
- Wu L, Hou Q, Zhou Q, Peng F. Prevalence of risk factors for diabetic foot complications in a Chinese tertiary hospital. Int J Clin Exp Med. 2015;8(3):3785-92. PubMed | Google Scholar
- Leye A, Diack ND, Leye YM, Ndiaye N, Bahati A, Dieng A et al. Assessment of the podiatric risk on diabetics in Dakar hospital area: cross-sectional study in regard to 142 patients. J Diabetes Mellit. 2018;08(01):1-8. Google Scholar
- Ogbogu CJ, Opara CH, Anarado AN, Anetekhai WI. High-risk diabetic foot among adults with diabetes in Enugu State, Nigeria. WCET Journal. 2018;38(3):10-20. Google Scholar
- Kishore S, Upadhyay AD, Jyotsna VP. Categories of foot at risk in patients of diabetes at a tertiary care center: insights into need for foot care. Indian J Endocrinol Metab. 2015;19(3):405-10. PubMed | Google Scholar
- Pendsey SP. Understanding diabetic foot. Int J Diabetes Dev Ctries. 2010;30(2):75-9. PubMed | Google Scholar
- Din SA, Mekkawy MM, Besely WN, Azer SZ. Prevalence of risk factors for Egyptian diabetic foot ulceration. IOSR J Nurs Heal Sci. 2016;5(2):45-57. Google Scholar
- Shahbazian H, Yazdanpanah L, Latifi SM. Risk assessment of patients with diabetes for foot ulcers according to risk classification consensus of International Working Group on Diabetic Foot (IWGDF). Pak J Med Sci. 2013;29(3):730-4. PubMed | Google Scholar
- Alam M, Butt SJ, Hayyat K, Arshad A, Mubarak S. Study of risk factor cotributary in the development of diabetic foot ulceration. Pakistan J Med Heal Sci. 2017;11(4):1282-6. PubMed | Google Scholar











