Drug storage, polypharmacy and frailty syndrome in older people: an observational study
Maturin Tabué-Téguo, Roxane Villeneuve, Jeannie Helene-Pelage, Matteo Cesari, Jordane Chovino, Axelle Boire, Moustapha Dramé, Nadine Simo-Tabué, Denis Boucaud-Maitre
Corresponding author: Denis Boucaud-Maitre, Centre Hospitalo-Universitaire de Guadeloupe, Pointe-à-Pitre, Guadeloupe, France 
Received: 30 Nov 2021 - Accepted: 17 Oct 2022 - Published: 11 May 2023
Domain: Geriatrics,Drug delivery systems
Keywords: Frailty, polypharmacy, drug storage, elderly
©Maturin Tabué-Téguo et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Maturin Tabué-Téguo et al. Drug storage, polypharmacy and frailty syndrome in older people: an observational study. Pan African Medical Journal. 2023;45:29. [doi: 10.11604/pamj.2023.45.29.32636]
Available online at: https://www.panafrican-med-journal.com//content/article/45/29/full
Drug storage, polypharmacy and frailty syndrome in older people: an observational study
![]() Maturin Tabué-Téguo1,2,3,
Maturin Tabué-Téguo1,2,3, ![]() Roxane Villeneuve1,3, Jeannie Helene-Pelage4, Matteo Cesari5, Jordane Chovino1,
Roxane Villeneuve1,3, Jeannie Helene-Pelage4, Matteo Cesari5, Jordane Chovino1, ![]() Axelle Boire1, Moustapha Dramé5, Nadine Simo-Tabué1,
Axelle Boire1, Moustapha Dramé5, Nadine Simo-Tabué1, ![]() Denis Boucaud-Maitre1,2,&
Denis Boucaud-Maitre1,2,&
&Corresponding author
Introduction: the increasing prevalence of polypharmacy in the older population could lead to inappropriate storage of medicines at home. Since polypharmacy is associated with frailty, the main objective of the Karukera Study of Aging - Drug Storage (KASADS) study was to investigate the association between drug storage and frailty. If such an association exists, drug storage could be a simple tool for the identification of medication vulnerability by non-medical staff in the elderly.
Methods: observational, cross-sectional study in community-dwelling older adults (>65 years old). Drug storage was defined as any drug in excess compared to a medical prescription, any unused and/or expired drug, or any drug without a medical prescription. Frailty was measured with the Study of Osteoporotic Fractures (SOF) scale, and polypharmacy was defined as a prescription of at least 5 drugs. Bivariate and multivariate analyses were performed to study the associations between drugs storage, frailty, and polypharmacy.
Results: during the study period (01/10/2019 to 15/03/2020), 115 elderly people were interviewed in their own homes. The average age was 76.0 ± 7.8 years old. Seventy-two percent of the participants met the criteria for polypharmacy and 30.4% were prefrail/frail. They stored an average of 14.7 ± 18.2 boxes. Drug storage was associated with polypharmacy (17.5 boxes versus 10.0; p=0.031) but not with frailty (15.6 versus 14.3; p=0.724). In multivariate analysis, drug storage was associated with not having a school degree (OR: 1.78; 95%CI: 1.13-2.79), suffering from dyslipidemia (OR: 2.00; 95% CI:1.28-3.17) and suffering from cognitive disorders evaluated by the Mini Mental State Examination (MMSE) score (OR: 1.10; 95%CI:1.02-1.17).
Conclusion: drug storage was not significantly associated with frailty. Nevertheless, it was associated with polypharmacy and other medical outcomes, and could therefore represent a new area for research in geriatrics and pharmacy.
For several years now, in France, public authorities have been launching mass media campaigns encouraging users to return unused medicines to pharmacies. These messages were aimed at tackling self-medication, and especially iatrogenesis and its deleterious effects among the older population [1], partly stemming from inappropriate storage of medicines at home. However, drug storage has received little attention in the scientific community and has no consensual definition. In an Australian study [2], conducted in 2018 in 166 households, the authors found 1429 “unused” drugs. Storage was defined as "any medicine not used daily" (unused medicines and those with an expiry date that had passed). In another study in Pakistan, assessing the causes of drug storage and the different uses of the stored medicines [3], drug storage was not clearly defined. However, according to the authors, 100% of households "stocked" drugs: current treatment represented 20.4% of the stored medicine; 70.9% were drugs kept for future use, and 8.7% were leftover drugs from previous treatments. Over the last decades, the number of drugs per prescription has increased [4], and this could be one of the main reasons for the inappropriate storage of medicines at home. However, the link between polypharmacy and drug storage has seldom been studied.
Polypharmacy is defined by the World Health Organization (WHO) as "the administration of many drugs at the same time or the administration of an excessive number of drugs" [5-7]. As is the case for a majority of geriatric syndromes, there is as yet no consensus on its definition. A threshold of 5 or more drugs is commonly reported [8,9]. The prevalence of polypharmacy increases with age, from 55.9% in people in their forties to 88.9% in octogenarians. It then decreases in centenarians (26.3%). It is therefore a public health issue for older people. Moreover, the relationship between polypharmacy and the risk of negative health events in older people has been clearly demonstrated, such as falls, dependency, nursing home admission, death, etc., and particularly frailty [10].
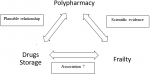
Drug storage, polypharmacy, and frailty are all parameters associated with aging and could be interrelated. The association between frailty and drug storage needs to be explored, as drug storage could serve as a simple tool (requiring no medical expertise) for the identification of frailty by non-medical staff in the older population (Figure 1). However, to our knowledge, no study has assessed the relationship between frailty and drug storage, and there are relatively few reports on the link between polypharmacy and drug storage. The main objective of the KASADS study was therefore to investigate an association between drug storage and frailty syndrome in the elderly (Figure 1).
Study design: this prospective, observational, cross-sectional study was conducted in Guadeloupe (French West Indies) between October 1, 2019, and March 15, 2020. The study was approved by the Ethics Committee of the University Hospital of Guadeloupe (Ref: A6_19_10_01_KASADS).
Setting and participants: the participants of the study were recruited from the patient base of 15 volunteer GPs in the territory. Within the same week, each physician had to systematically recommend the study to older people consulting at their medical practice, until the quota of 10 older people per physician was reached. The inclusion criteria were as follows: man or woman aged at least 65 years old, affiliated to a social security plan, with a medical prescription at the end of the visit. The criteria for non-inclusion were refusal to participate in the study, severe or unstable general pathology, being under guardianship, or living in an institution.
The selected patients were contacted by phone by two medical school students, and after obtaining a verbal agreement, the same two medical school students went to their homes to collect data, using a standard case report form.
Variables: the following variables were collected: age, gender, medical history, level of education, current and previous prescriptions, frailty, undernutrition, cognitive status, and dependence assessment scales. All the drugs that were stored, in the bathroom or in another room of the house, were recorded.
Drug storage was defined as: any drug in excess of the medical prescription prescribed by the various doctors, including those used on a daily basis; not used, and/or expired; as well as any non-prescription drug. Storage was analyzed according to the total number of boxes (main analysis), the total number of boxes under the list, and the total number of boxes over the counter. When the storage concerned the same drug, all the boxes were counted. Frailty was assessed using the SOF scale [11,12] based on the following 3 questions: (1) unintentional weight loss in the last year of more than 5 kilos; (2) ability to get up from a chair 5 times without using the arms; (3) lack of energy in the last week. Each item was scored as "1" or "0", a score of 0 corresponding to a robust older person, a score of 1 or more was considered frail.
Polypharmacy was defined as a prescription for at least 5 medications [8]. Participants were asked whether they were diagnosed with hypertension, dyslipidemia, and diabetes. Disability for the basic activities of daily living [13] (ADL) and instrumental (IADL) was assessed based on the Katz index and Lawton and Brody scale [14] respectively. The Katz index is designed to assess the subject's capacity to perform ADL such as taking a bath, getting dressed, and using the toilet among others. On the other hand, the Lawton and Brody scale evaluates the subject's ability to perform IADLs such as using the phone, managing money, using transportation, and shopping, among others. Other scales were used to assess nutritional status Mini Nutritional Assessment Short-Form (MNA-SF) [15], and cognitive status (MMSE) [16].
Study size: no sample size was calculated, due to the lack of literature on drug storage. Nevertheless, the intended sample size was 150 patients in this exploratory study.
Statistical methods: quantitative variables were expressed as mean ± standard deviation, categorical variables as percentages. Bivariable analyses were conducted to compare 1. The clinical characteristics and the amount of medicine packages in storage between patients with frailty or no frailty syndrome 2. The association between the amount of medicine packages in storage and polypharmacy. For the multivariable analysis, a quasi-Poisson model was used to study the relationship between the amount of medicine packages in storage and the variables of interest (age, sex, education, hypertension, diabetes, dyslipidemia, frailty, MMSE, and IADL scores). P-values, a threshold of 0.05, and 95% confidence intervals were reported. All analyses were performed using R v.3.0.2.
Participants: a total of 115 older people were included out of the 150 originally planned. The inclusion of patients was stopped on March 15, 2020 due to the COVID-19 crisis and the ensuing lockdown in France [17]. These participants were 76.0 ± 7.8 years old on average, were predominantly female (67.8%), and had a BMI of 26.8 ± 5.3 kg/m2. In our sample, 43.5% suffered from diabetes, 87.0% from hypertension, 58.3% from obesity, and 52% from dyslipidemia. Polypharmacy was observed in 63% of the participants (Table 1). The prevalence of drug storage was 82.6% (95 out of 115 people were storing drugs). A feature of prefrailty or frailty was observed in 35 (30.4%) older people, and these frail people were older than the robust (79.5 years versus 74.5, p=0.001) and more often on polypharmacy (77.1% versus 56.6%) (Table 1).
Main results
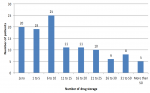
Associations between drug storage, frailty and polypharmacy: a total of 1,696 boxes of medicine were registered, with an average of 14.7 ± 18.2 boxes per older person. Only 17.4% of older people did not stock any medication. The distribution for storage is shown in Figure 2. No difference was observed between frail and robust older people on the total number of packages (15.6 versus 14.3; p=0.724), the total number of packages under prescription (8.3 versus 7; p=0.566), or the total number of packages over the counter (7.3 versus 7.4; p=0.945) (Table 2).
On the other hand, polypharmacy was associated with storage with respect to the total number of boxes (17.5 versus 10.0; p=0.031) and the number of prescription boxes (9.2 versus 4.3; p=0.021) (Table 2).
Other analyses
Determinants of drug storage: in a multivariate analysis (Table 3), overall drug storage was associated with not having a degree (OR: 1.78; 95%CI: 1.13-2.79), being dyslipidemic (OR: 2.00; 95% CI:1.28-3.17) and MMSE score (OR: 1.10; 95%CI:1.02-1.17).
Prescription drug storage was associated with male sex (OR: 2.02; 95%CI: 1.18-3.39), being dyslipidemic (OR: 2.67; 95%CI: 1.57-4.78), being on polypharmacy (1.98; 95%CI: 1.10-3.75) and MMSE score (OR: 1.10; 95%CI: 1.01-1.19). Finally, storage of over-the-counter drugs was only associated with the MMSE score (OR: 1.11; 95%CI: 1.03-1.19) and with being dyslipidemic (OR: 1.63; 95%CI: 1.02-2.64) (Table 3).
In this study, we did not find any significant association between drug storage and frailty in older people, for both prescription and over-the-counter drugs. Storage is therefore not a potential tool for detecting frailty, unlike, for example, reduced walking speed, which seems to be a reliable tool for identifying the risk of dependence, institutionalization, falls or mortality [18,19]. Nevertheless, this tool is not necessarily applied in routine care, and remains poorly accessible to untrained people.
From a clinical point of view, this lack of association between drug storage and frailty remains intriguing. The expected results were indeed in favor of a strong association, in relation to multiple prescriptions, polypharmacy, or non-systematic taking of treatments, particularly in people with low MMSE scores. However, this is not the result that appears in this study.
Nevertheless, our study suggests that drug storage may be associated with other factors. As expected, drug storage was associated with polypharmacy, both for the total number of drugs and the sub-sample of prescription drugs. This large amount of medicines in storage, and the high proportion of people stockpiling drugs, is worthy of further investigation. Beyond the issue of non-prescription drugs, whose storage is facilitated by their accessibility, other hypotheses can be put forward, such as the irregular taking of drugs but their regular renewals or changes in treatment - following hospitalization, changes in health status, discontinuation of a given drug (for example, prescribed by both the general practitioner and a specialist).
One hypothesis that should be explored is the specificity of the drug delivery system in France. Indeed, while in some countries the exact dose of capsules or tablets is delivered to the patient, in France, medicines are only available in packages with a predefined number of tablets [20]. Although the number of tablets is generally calculated to correspond to a weekly or monthly standard treatment plan, it is not uncommon for the number of tablets needed for treatment to be too large. Leftover treatment may be stored in the medicine cabinet, "just in case" instead of being taken back to the pharmacy. This accumulation of unused medicines also has a significant cost for social security, as the quantities charged are higher than the quantities prescribed. For example, Megerlin [21,22], and Lhoste [23,24] estimated the cost of medicines prescribed, but not used, in nursing homes at €60,409,310.40 per year (for 574,670 residents). Moreover, the French healthcare system facilitates access to medicines (free of charge for the patient in a majority of cases). This situation favors stockpiling for both frail and non-frail people. This may explain the lack of significant difference.
Since drug storage and polypharmacy are associated, further studies would be needed to determine whether drug storage is associated with other health events related to polypharmacy, including non-adherence to treatment, iatrogenesis falls [25], hospitalization, and mortality [26,27].
Limitations: the positive association between drug storage and MMSE score may seem counter-intuitive at first glance. However, the lower the MMSE score, the more likely it is that older people are no longer responsible for refilling prescriptions at the pharmacy. In people with cognitive impairment, if medication management is overseen by an informal caregiver or healthcare professional, risks of buying unnecessary non-prescription drugs are lessened, and proper renewal and intake of medication altogether are more likely. Nevertheless, no information is available in our database regarding family and social support, such as the presence of a nurse or a relative to help with medication management. In addition, we cannot rule out that for people with cognitive impairment who do not live alone, some non-prescription medications could be intended for others in the same household. We also found an association between drug storage and dyslipidemia. This result, quite surprising, could be explained in part by the impact of cardiovascular risk factors (diabetes, hypertension, obesity...) in polypharmacy in older persons and those the due to the association with other chronic diseases and/or complications.
Drug storage is an understudied topic, even though it could provide a simple tool for detecting negative health events, in order to implement preventive action. In our study, drug storage was not associated with frailty, but larger studies would be necessary to confirm or infirm our results. Drug storage was nevertheless associated with polypharmacy, which is itself associated with negative health events (iatrogenicity, hospitalization, etc.) that could be worth exploring in the context of future research.
What is known about this topic
- Polypharmacy in the older population could lead to inappropriate storage of medicines at home;
- Since polypharmacy is associated with frailty, the main objective of this study was to investigate the association between drug storage and frailty.
What this study adds
- Drug storage was not associated with frailty but associated with other clinical outcomes;
- Drug storage is an understudied topic, even though it could provide a simple tool for detecting negative health events, in order to implement preventive actions;
- Drug storage could therefore represent a new area for research in geriatrics and pharmacy.
The authors declare no competing interests.
Maturin Tabué-Téguo, Denis Boucaud-Maitre, Nadine Simo-Tabué, and Jeannie Helene-Pelage, Axelle Boire, Jordane Chovino have contributed to the conception and design of the study, acquisition, analysis, and interpretation of data; Matteo Cesari, Moustapha Dramé, and Roxane Villeneuve were involved in the analysis and interpretation of data, drafting the manuscript, revising it critically and gave final approval of the version to be submitted. All the authors read and approved the final version of this manuscript.
We are grateful to Ms. Lydia Foucan, for her assistance with the statistical analysis of the data, and to the staff of the KASADS study for their collaboration and assistance with the collection of data.
Table 1: characteristics of participants by frailty profile
Table 2: relationship between drug storage, frailty and polypharmacy
Table 3: determinants of drug storage
Figure 1: main hypothesis of the study
Figure 2: number of drug storage
- Cyclamed. Cyclamed renouvelle sa campagne d´affichage dans le métro et RER francilien. 2021. Accessed February 19, 2021.
- Kelly F, McMillan S, Spinks J, Bettington E, Wheeler AJ. 'You don´t throw these things out:´ an exploration of medicines retention and disposal practices in Australian homes. BMC Public Health. 2018;18(1):1026. PubMed | Google Scholar
- Hussain R, Rashidian A, Hafeez A. A Survey On Household Storage of Medicines In Punjab, Pakistan. J Ayub Med Coll Abbottabad. 2019;31(1):90-97. PubMed | Google Scholar
- Barat I, Andreasen F, Damsgaard EM. The consumption of drugs by 75-year-old individuals living in their own homes. Eur J Clin Pharmacol. 2000 Sep;56(6-7):501-9. PubMed | Google Scholar
- Kim J, Parish AL. Polypharmacy and Medication Management in Older Adults. Nurs Clin North Am. 2017 Sep;52(3):457-468. PubMed | Google Scholar
- Lai SW, Liao KF. Optimal cutoff of polypharmacy and outcomes. J Clin Epidemiol. 2013;66(4):465. PubMed | Google Scholar
- Herr M, Robine J-M, Pinot J, Arvieu JJ, Ankri J. Polypharmacy and frailty: prevalence, relationship, and impact on mortality in a French sample of 2350 old people. Pharmacoepidemiol Drug Saf. 2015;24(6):637-646. PubMed | Google Scholar
- Gnjidic D, Hilmer SN, Blyth FM, Naganathan V, Waite L, Seibel MJ et al. Polypharmacy cutoff and outcomes: five or more medicines were used to identify community-dwelling older men at risk of different adverse outcomes. J Clin Epidemiol. 2012;65(9):989-995. PubMed | Google Scholar
- Tabue-Teguo M, Grasset L, Avila-Funes JA, Genuer R, Proust-Lima C, Péres K et al. Prevalence and Co-Occurrence of Geriatric Syndromes in People Aged 75 Years and Older in France: Results From the Bordeaux Three-city Study. J Gerontol A Biol Sci Med Sci. 2017;73(1):109-116. PubMed | Google Scholar
- Saum K-U, Schöttker B, Meid AD, Holleczek B, Haefeli WE, Hauer K et al. Is Polypharmacy Associated with Frailty in Older People? Results From the ESTHER Cohort Study. J Am Geriatr Soc. 2017;65(2):e27-e32. PubMed | Google Scholar
- Ensrud KE, Ewing SK, Taylor BC, Fink HA, Cawthon PM, Stone KL et al. Comparison of 2 frailty indexes for prediction of falls, disability, fractures, and death in older women. Arch Intern Med. 2008;168(4):382-389. PubMed | Google Scholar
- Kiely DK, Cupples LA, Lipsitz LA. Validation and comparison of two frailty indexes: The MOBILIZE Boston Study. J Am Geriatr Soc. 2009;57(9):1532-1539. PubMed | Google Scholar
- KATZ S, FORD AB, MOSKOWITZ RW, JACKSON BA, JAFFE MW. STUDIES OF ILLNESS IN THE AGED. THE INDEX OF ADL: A STANDARDIZED MEASURE OF BIOLOGICAL AND PSYCHOSOCIAL FUNCTION. JAMA. 1963 Sep 21;185:914-9. PubMed | Google Scholar
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9(3):179-186. PubMed | Google Scholar
- Rubenstein LZ, Harker JO, Salvà A, Guigoz Y, Vellas B. Screening for undernutrition in geriatric practice: developing the short-form mini-nutritional assessment (MNA-SF). J Gerontol A Biol Sci Med Sci. 2001;56(6):M366-372. PubMed | Google Scholar
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975 Nov;12(3):189-98. PubMed | Google Scholar
- Peretti-Watel P, Verger P, Launay O, COCONEL Study Group. The French general population´s attitudes toward lockdown against COVID-19: a fragile consensus. BMC Public Health. 2020;20(1):1920. PubMed | Google Scholar
- Abellan van Kan G, Rolland Y, Andrieu S, Bauer J, Beauchet O, Bonnefoy M et al. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J Nutr Health Aging. 2009;13(10):881-889. PubMed | Google Scholar
- Studenski S, Perera S, Patel K, Rosano C, Faulkner K, Inzitari M et al. Gait speed and survival in older adults. JAMA. 2011 Jan 5;305(1):50-8. PubMed | Google Scholar
- Huon J-F, Lenain E, LeGuen J, Chatellier G, Sabatier B, Saint-Jean O. How Drug Use by French Elderly Patients Has Changed During the Last Decade. Drugs Real World Outcomes. 2015;2(4):327-333. PubMed | Google Scholar
- Megerlin F. [Conditional pricing for innovative medicines in France: stop telling about risk-sharing!]. Ann Pharm Fr. 2013;71(5):291-301. PubMed | Google Scholar
- Megerlin F, Lopert R, Taymor K, Trouvin J-H. Biosimilars and the European experience: implications for the United States. Health Aff (Millwood). 2013;32(10):1803-1810. PubMed | Google Scholar
- Lhoste F. [8th Descartes-Berkeley interuniversity symposium on health economics, May 2013, Paris, France: an overview]. Ann Pharm Fr. 2013;71(5):277-278. PubMed | Google Scholar
- Lhoste F. [Trust-based economics with medicine outcome-based pricing]. Ann Pharm Fr. 2013;71(5):279-284. PubMed | Google Scholar
- Huang Y, Wang L, Zhong C, Huang S. Factors influencing the attention to home storage of medicines in China. BMC Public Health. 2019;19(1):833. PubMed | Google Scholar
- Abushanab AS, Sweileh WM, Wazaify M. Storage and wastage of drug products in Jordanian households: a cross-sectional survey. Int J Pharm Pract. 2013;21(3):185-191. PubMed | Google Scholar
- Ocan M, Bbosa GS, Waako P, Ogwal-Okeng J, Obua C. Factors predicting home storage of medicines in Northern Uganda. BMC Public Health. 2014;14:650. PubMed | Google Scholar