Ultra sonographic indices of the carotid artery in healthy adult population of southwest, Nigeria: a cross-sectional study
Abiola Omobonike Adekoya, Ayodeji Anike Olatunji, Racheal Adeyanju Akinola, Olatunde Odusan, Adesola Olubunmi Adekoya, Mojisola Adejoke Olusola-Bello, Olatunbosun Oladipupo Olawale
Corresponding author: Abiola Omobonike Adekoya, Department of Radiology, Olabisi Onabanjo University Teaching Hospital, Sagamu, Nigeria 
Received: 16 Jun 2021 - Accepted: 14 Feb 2023 - Published: 20 Feb 2023
Domain: Radiology, Non-Communicable diseases epidemiology, Health information system management
Keywords: Atherosclerosis, carotid flow velocities, subclinical atherosclerosis
©Abiola Omobonike Adekoya et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Abiola Omobonike Adekoya et al. Ultra sonographic indices of the carotid artery in healthy adult population of southwest, Nigeria: a cross-sectional study. Pan African Medical Journal. 2023;44:97. [doi: 10.11604/pamj.2023.44.97.30386]
Available online at: https://www.panafrican-med-journal.com//content/article/44/97/full
Research 
Ultra sonographic indices of the carotid artery in healthy adult population of southwest, Nigeria: a cross-sectional study
Ultra sonographic indices of the carotid artery in healthy adult population of southwest, Nigeria: a cross-sectional study
![]() Abiola Omobonike Adekoya1,&, Ayodeji Anike Olatunji1, Racheal Adeyanju Akinola2, Olatunde Odusan3,
Abiola Omobonike Adekoya1,&, Ayodeji Anike Olatunji1, Racheal Adeyanju Akinola2, Olatunde Odusan3, ![]() Adesola Olubunmi Adekoya4, Mojisola Adejoke Olusola-Bello1,
Adesola Olubunmi Adekoya4, Mojisola Adejoke Olusola-Bello1, ![]() Olatunbosun Oladipupo Olawale5
Olatunbosun Oladipupo Olawale5
&Corresponding author
Introduction: atherosclerosis develops insidiously, offering time and opportunities for early detection. Screening for subclinical atherosclerosis via structural wall changes and flow velocities among apparently healthy adults using carotid ultrasonography may help its early detection, offer timely intervention and reduce morbidity and mortality.
Methods: a cross-sectional study of 100 participants with a mean age of 56.1 ± 6.9 years, were enrolled from a community population. Both carotid arteries were examined for plaques, carotid intima-media thickness (CIMT), and flow velocities - peak systolic velocity (PSV), end-diastolic velocity (EDV), pulsatility index (PI), and resistive index (RI) using 4-12MHz linear array transducer. Visceral obesity, serum lipids, and blood glucose were also evaluated and correlated with ultrasound findings.
Results: the mean CIMT was 0.07 ± 0.02cm and 15% of the participants had increased CIMT. Statistically significance but weak correlations were observed between CIMT and FBG (r = 0.199, p = 0.047), EDV (r =0.204, p= 0.041), PI (r = -0.287, p = 0.004) and RI (r = -0.268, p =0.007). Statistically significance with modest correlations were observed between EDV and PSV (r = 0.48, p = 0.000), PI (r = -0.635, p = 0.000) and RI (r = -0.637, p = 0.000). The PI and RI showed strong correlation with statistical significance (r= 0.972, p = 0.000).
Conclusion: statistical significances in the flow velocities, derived flow indices and increased CIMT may be an early indication of subclinical atherosclerosis. Therefore, ultrasonography may facilitate its early detection and possible prevention of complications.
Atherosclerosis is a chronic immune-inflammatory fibro-proliferative disease of large and medium-sized arteries of varying etiologies [1]. It is the leading cause of morbidity and mortality in westernized society and the primary cause of cardiovascular diseases (CVD), resulting in myocardial infarction, stroke, and peripheral arterial disease [2-4]. Known risk factors for developing atherosclerotic cardiovascular disease (ASCVD) include age, smoking, diabetes mellitus (DM), hypertension (HTN), sedentary lifestyle, obesity, and deranged lipids [5,6]. These, however, do not accurately predict ASCVD development [6]. Early atherosclerosis is usually asymptomatic and clinically dormant as subclinical atherosclerosis (SCA) [7]. The early structural arterial changes involve smooth muscle cell proliferation, subendothelial macrophage migration, and fatty streak formations with the eventual development of matured vulnerable plaques [7]. This asymptomatic slow disease build-up offers time for screening and detection of these changes in the vascular structures [8].
Ultrasonography (US) evaluation of the carotid artery for SCA has been validated while various other non-invasive methods for its clinical risk assessment exists [9,10]. Coronary artery calcium score (CACS), CIMT, and plaque on carotid artery ultrasound (CU) and ankle-brachial index (ABI) have proven to be useful [11,12]. Computed tomography (CT) CACS better predicts future CVD and coronary events, however, the relatively poor mobility of CT scanners with ionizing radiation and late identification of atherosclerotic lesions are some drawbacks [13,14]. Measurement of ABI, though safe and available, is dependent on the presence of advanced stenotic disease with reduced ABI reported in advanced atherosclerosis [10]. Carotid artery US is a safe, patient-friendly, reproducible, and relatively affordable imaging technique that has been validated for generalized atherosclerosis burden and vascular disease risk [15-17]. Increased CIMT is reportedly associated with future CVS events, while ultrasound-derived carotid plaque burden (CPB) has a similar predictive value as CACS for future CVD event development [18]. Carotid artery structural changes and flow velocities using CU amongst high-risk individuals have been widely reported, however, there is a paucity of such studies on individuals with no known cardiovascular disease risk factors (CVRFs).
Objectives: we aimed to determine the prevalence of atherosclerosis via structural wall changes and blood flow velocities of the carotid arteries of apparently healthy adults in our environment as this may facilitate their early detection of SCA. We hypothesized that SCA may be absent in them as they have no known CVRFs.
Study design and setting: this was a hospital-based cross-sectional study approved by our hospital´s Health Research and Ethics Committee (NHREC/08/10/2012). One hundred non-hypertensive, non-diabetic adults aged 30 - 70 years were purposively enrolled for a year (December 2017 - November 2018) from a community population attending the tertiary hospital. All the participants gave informed/written consent before participating in the study.
Study participants: all participants were selected from the Medical and General out-patient clinics where they had presented for ailments with no known CVRFs, except age. Excluded from the study were patients with HTN, DM, chronic kidney disease (CKD), past or present history of heart failure, myocardial infarction (MI), coronary heart disease (CHD), cerebrovascular accident, and cancer. Pregnant and lactating women, patients on tracheostomy tubes, and central lines, and those with anatomical constraints like short muscular necks, high bifurcation, and tortuous arteries were exempted from the study.
Methodology and bias removal: using a pre-tested interviewer-administered structured questionnaire, information on socio-demography, past medical history, current medications, alcohol use, and smoking were obtained as well as anthropometric, clinical, and laboratory parameters. The same ultrasound machine [4-12MHz linear array (PHILIPS® CLEAR VUE 550, Phillips Healthcare, 2014)] was used for all the participants to exclude errors due to inter-equipment variation. Inter-observer variability errors were minimized by calibration and recalibration of the same equipment before use. Strict hygienic measures such as cleaning equipment before and after each use, washing of hands, and use of latex gloves were adhered to, according to standard operating procedures.
Study size: this was by convenience sampling.
Anthropometry and clinical variables: body weight (kg) was assessed using a standing weighing scale, (Seca® 755, Hamburg, Germany), and placed on an even, horizontal hard surface. Height (m) was measured standing using a stadiometer, (Seca® 755, Hamburg, Germany) with the head positioned in the Frankfort plane [19]. The body mass index (BMI) in kg/m2 was calculated with participants classified as underweight (BMI< 17.8), normal (BMI = 17.8-23.6), overweight (BMI = 23.7 - 26.8), and obese (BMI ≥26.9) in males and underweight (BMI <17.8), normal (BMI = 17.8 - 25.6), overweight (BMI = 25.7 - 28.7) and obese (BMI > 28.8) in females [20]. The waist circumference (WC) was measured following the WHO STEPS protocol [21] and classified as normal (WC <82.99cm), overweight (WC = 83-94.99cm) and obese (WC >95cm) in women, and normal (WC <83.99cm), overweight (WC = 84-95.99cm) and obese (WC >96cm) in men [22]. An average of two brachial blood pressure measurements [systolic blood pressure (SBP) and diastolic blood pressure (DBP)] by auscultation using Accuson® mercury sphygmomanometer with a suitable cuff size in a sitting position, after 5 minutes rest was recorded.
Laboratory procedure and definitions: participants' venous blood samples (10mls) were collected aseptically between 07:00 and 08:00 (GMT + 1), after an overnight fast, and assayed for fasting blood glucose (FBG) and serum lipids. Two milliliters (mL) and 3mL of blood were dispensed into fluoride oxalate and plain sample bottles for FBG estimation and lipid assay respectively. Plasma samples for FBG were analyzed immediately on the day of collection, while samples for lipid assay were centrifuged (swing-bucket) within two hours of collection at 3000 revolutions per minute (RPM) for five minutes. The separated serum samples in plain plastic screw-capped containers were stored frozen (-20°C) and assayed for serum lipids within one week of collection. Colorimetric kits manufactured by Randox Laboratories Ltd, United Kingdom, were used for the assay of FBG, Total Cholesterol (TC), Triglyceride (TG), and high-density lipoprotein cholesterol (HDL-C). Low-density cholesterol (LDL-C) was calculated using the Friedewald formula. Laboratory evaluation included FBG (normal 70-110mg/dl) [23] and normal fasting serum lipid (TG<200mg/dl, TC <200mg/dl, LDL-C <160mg/dl and HDL-C >40mg/dl) [24]. All blood samples were analyzed in our hospital´s Research Laboratory by a Consultant Chemical Pathologist.
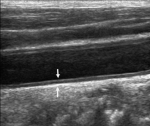
Ultrasound evaluation and definition: the CIMT is the distance between the leading edge of the first hyper-echogenic line (lumen-intima interface) to the leading edge of the second hypo-echogenic line (media-adventitia interface) of the posterior wall of the carotid artery [25]. Using the Mannheim criteria, carotid plaques (CP) are the focal echogenic structures encroaching into the arterial lumen with a thickness of more than 1.4mm when measured from the media-adventitia interface to the intima interface (Figure 1) [25]. Three radiologists with 5-years of experience in CU concurrently performed the US of the participants, each blinded to the result of the other. The participants were placed in a supine position with jewelry and clothing removed from the study area. The chin was raised and slightly extended and the head rotated about 45° away from the examined side after applying the warm water-based ultrasonic gel.
Carotid B-mode, color, and spectral Doppler US in the longitudinal plane of the common carotid artery (CCA), carotid bulb (CB), and internal carotid artery (ICA) on the right and left sides were examined (Figure 1). Three measurements of CIMT, measured with in-built electronic calipers after freezing the images, at the far wall were obtained at the CB, 1cm below and above the bulb for CCA and ICA respectively. Left and right CIMT measurements were averaged and an overall CIMT was taken as a mean of both. Values between 0.9-1.4 mm were considered as increased CIMT and >1.4mm indicated atheromatous plaque [26]. The near and far walls of carotid arterial segments were evaluated for CP bilaterally. The echogenicity of CP was classified as isoechoic, hypoechoic, hyperechoic, or calcified, and the surface was expressed as smooth, irregular, or ulcerated. Location, mobility, and the number of arterial segments with plaques were also noted. Spectral waveforms of PSV and EDV were obtained from continuous-wave Doppler examination within one cardiac cycle at an angle of 45 - 600 in the distal CCA and proximal ICA, about 2cm beyond the bifurcation bilaterally. The highest and lowest velocities during systole and diastole were taken as PSV and EDV, respectively. Three measurements taken bilaterally were averaged and overall PSV and EDV were taken as a mean of the CCA and ICA. In cases of atrial fibrillation, average values of five consecutive velocity rhythms were used. Resistive and pulsatility indices which are dependent on the blood-shape waveform and independent of the insolation angle were derived [27]. The pulsatility index (PI), known as the Gosling index, is the difference between PSV and EDV divided by mean flow velocity [28] and RI (Pourcelot index) is the difference between PSV and EDV divided by PSV [27]. The average scan time was 30 minutes per patient. After the procedure, the gel was wiped off the participant´s skin with soft tissue paper, and the neck returned to its normal position.
Data management and statistical methods: the generated data were analyzed using the Statistical Package for the Social Sciences (SPSS) for Windows IBM compatible version 23.0. Continuous variables were expressed as mean± standard deviation and categorical variables as proportions and frequencies (percentage). Values were compared for differences using the Student´s t-test and Chi-square tests. Analysis of Variance (ANOVA) and Pearson correlation coefficient (r) were used to determine the relationship between continuous variables with statistical significance set at p< 0.05.
One hundred volunteered eligible participants completed the study and were analyzed. Sixty-two females and 38 males (male-to-female ratio of 0.6:1) with a mean age of 56.1 ± 6.9 years participated in the study. There were 21 (21%), 48 (48%), and 31 (31%) in the < 50 years, 51-60 years, and 61-70 years age groups respectively. The socio-demographic parameters of study participants are shown in Table 1. Their mean FBG was 79.9 ± 13.1mg/dl with mean SBP and DBP at 119.6 ± 11.5mmHg and 77.5 ± 8.7mmHg, respectively. There were no statistical significances in the mean WC (t = 0.038, p = 0.970), BMI (t = 0.046, p = 0.963), FBG (t = -1.211, p = 0.229), LDL -C (t = -0.705, p = 0.482), HDL -C (t = -0.944, p = 0.348), TG (t = 1.811, p = 0.073), and TC (t = -0.926, p = 0.357) along gender divide (Table 2).
The mean value of CIMT and flow velocities with their derived values are shown in Table 3. The mean CIMT was 0.07 ± 0.02cm in both gender and 0.07 ± 0.02cm for the age groups <50, 51-60, and 61-70 years with no statistical significance (p = 0.576). The carotid bulb had the highest CIMT bilaterally (0.08 ± 0.03cm). Fifteen percent of the study participants had increased CIMT distributed as 29.5%, 57.4%, and 13.1% at the CCA, bulb, and ICA respectively. The increased CIMT showed no statistically significant differences with almost equal gender distribution (46.7% males and 53.3% females). The mean CIMT had positive weak correlations with statistical significances with FBG (r = 0.199, p = 0.047) and SBP (r = 0.224, p = 0.025). There were no statistically significant differences with BMI (p = 0.987), WC (p = 0.406), TC (p = 0.193), TG (p = 0.532), HDL-C (p = 0.429), and LDL-C (p = 0.126). Carotid plaque seen in 2% of the population (females in the 5th and 6th decades), had equal distribution at the bulbs. They were non-mobile, echogenic, and heterogenic on the right and left respectively with smooth outlines.
The mean flow velocity values were higher on the left and showed statistical significance with moderate positive correlations (r =0.639, p < 0.005). A higher PSV and EDV value was observed in males and females respectively, however, gender comparison showed no statistically significant difference (Table 3). Statistically, significances were observed between CCA-PSV and BMI (p = 0.008), WC (p = 0.017), SBP (p = 0.0008), and DBP (p = 0.019); ICA-PSV and BMI (p =0.0010, WC (p= 0.009), SBP (p = 0.023) and DBP (p= 0.009); CCA-EDV and CIMT (p =0.047), TC (p = 0.004) and LDL-C (p = 0.006) and ICA-EDV and FBG (p = 0.032). The mean PSV showed a weak negative association with BMI, WC, SBP, and DBP with statistically significant differences (p < 0.004) (Table 4). The mean PI and RI were higher in the males with no statistically significant difference (p = 0.138 and 0.093 for PI and RI respectively). Moderate and positive correlations were observed between the right and left PI and RI with statistical significance (p < 0.05). However, a strong positive correlation was observed between PI and RI with statistical significance (r = 0.972, p = 0.000). Details of correlations of CIMT, PSV, EDV, PI, and RI with other parameters of the participants are shown in Table 4.
This study presents carotid artery ultrasonography (CU) findings in otherwise healthy adults, with previously unreported CVRFs. The CVRFs among the study participants were age, with 79% of the participants older than 50 years, central obesity (31%), generalized obesity (25%), and dyslipidemia (58%). These findings were like earlier reports amongst Nigerian adults in cross-sectional hospital-based studies which reported obesity at 34.2% and 44.7%, dyslipidemia at 68.5% and 44.7%, and hypertension at 49.4% and 40.8% respectively [29,30].
Carotid intima-media thickness and plaque: the mean CIMT in this study was similarly reported by Baba et al. [31]. However, its value is higher than that reported in Turkey [32] and lower than the controls of Koc et al. [33]. The differences could be due to ethnic or racial differences, anatomical selection sites and the instrument used. The CIMT at the CB being significantly higher than the CCA and ICA as similarly reported by Baba et al. [31] may be due to anatomy at carotid artery bifurcation and resultant hemodynamic changes [34]. The increased CIMT observed in our study participants was lower than 53.7% and 36.5% reported by Omisore et al. [29] and Okeahialam et al. [35] respectively. They used varying cutoffs with the presence of CVRFs in their study population. In our study, 2% of the participants had CP which contradicts other studies [29,36] reporting a higher prevalence of CP with statistical significance and a strong positive correlation with serum lipids although known CVRFs were also present [29,36]. A study in China reported significant carotid atherosclerotic plaque formation, independent of other vascular risk factors in pre-hypertensives with the same severity as in hypertensives [37].
Carotid flow velocities: there are reports associating carotid flow velocities and CVD in high-risk individuals [38-40], although, its clinical application for vascular complication prevention in people with no known risk factor is limited while flow changes may be present in them. Non-invasive tracking of changes in flow velocity using ultrasonography may provide additional markers for SCA leading to early detection and prevention of atherosclerosis. The PSV and EDV are independently associated with CVD development [38]. The EDV with the capacity of intracranial circulation blood provision at diastole [38], improves future prediction of CVD more than PSV [39] and for ischemic stroke than RI and PSV [38]. In this study, the PSV and EDV values were similar to the normotensives reported by Park et al. [40], although they reported higher values in the pre-hypertensive and controls, a difference which could be attributed to co-morbid factors in their study population [40,41]. An earlier report from a case-control study observed an association of high ischemic stroke risk with low CCA EDV while low EDV in hypertensive resulted in decreased arterial dispensability and low diastolic perfusion pressure, leading to low shear stress, hence, atherosclerosis [42,43]. In our study, a moderate correlation was observed between the EDV and PSV with a statistically significant difference.
Derived carotid flow indices: pulsatility and resistive indices assess the arterial wall shear stress, an important predictor of cardiovascular risk and atherosclerosis development [44]. The PI reflects the transmission of pulsatile energy into the CVS and end-organ microcirculation while the RI mirrors the vascular flow resistance in the end-organ vessels [40,44]. High carotid flow PI and RI are associated with CVRFs and suggest arterial wall changes [39,41]. In contrast to other studies [38-41], lower PI and RI were observed in this study with a strong positive correlation between the PI and RI. A high PI (>1.60) reported amongst the elderly with high FBG, large WC, and low HDL-C was associated with a higher incidence of stroke with a high hazard stroke ratio [41]. In Taiwan, RI > 0.76 was positively associated with CVD than a lower value [39]. Therefore, the US evaluation of these indices may be helpful in distal arterial vessel resistance and elasticity assessment where they are not easily accessible with ultrasonography.
Carotid flow velocities, carotid intima-media thickness, and metabolic disorder: hypertension, hyperglycemia, and dyslipidemia are risk factors for atherosclerotic vasculopathy with a known association with visceral adiposity [36]. A greater association of WC with metabolic syndrome than BMI or body fat percentage (BFP) has been reported. Visceral adiposity via WC is reportedly a better index for CVD development than regional or generalized obesity [45-47]. High WC independently contributes to high levels of TG and low plasma HDL-C concentrations in the body [48]. In this study, significant differences were noted between WC and TC and TG, a finding consistent with Ishihara et al. [49]. Weak associations observed between CIMT and WC, BMI, FBG, SBP, DBP, and serum lipids in this study, showed statistical significance only with FBG and SBP. The PSV also showed a weak association with SBP, DBP, WC, and BMI with statistical significance, indicating a relationship between flow velocity and atherosclerotic risk factors.
Limitations: a major limitation of this study is that it was hospital-based. This may prevent the actual representation of the findings among the general population. In addition, the carotid wall diameter and echocardiographic parameters of study participants were beyond the scope of this study and hence not evaluated. However, the statistically significant differences in participants with higher CIMT and strong positive correlations between PI and RI are important.
The statistically significant differences observed in CIMT, flow velocities, and the derived flow indices of participants with no known CVDRFs are essential. Therefore, the combination of structural arterial wall changes and velocity flow rates could be predictive of early diagnosis of SCA and promptly institute preventive measures for atherosclerosis.
What is known about this topic
- There are widely reported values of CIMT in various individuals with co-morbid disease conditions;
- A CIMT value greater than 0.09mm reportedly connotes atherosclerosis in the co-morbid states;
- Atherosclerotic vessels lose their elasticity and recoil, therefore, increasing their blood flow velocities.
What this study adds
- Despite various reported carotid US evaluations of different individuals, there is no information on apparently healthy individuals with no known CVDRs which may be a pointer to early detection of non-symptomatic atherosclerosis;
- Statistically significant differences seen between the CIMT, and flow velocities and the derived PI and RI of this study population are reflective of ongoing SCA; in addition, this study provides reference values of CIMT, PSV, EDV, PI, and RI in individuals in our environment with no known CVDRFs;
- The absence of known CVDRFs in people does not exclude them from developing atherosclerosis; therefore, screening of the carotid arteries via US may provide an avenue for early detection of SCA and prompt intervention, reducing morbidity and mortality.
The authors declare no competing interests.
Abiola Omobonike Adekoya contributed to the conception and design, data acquisition, data analysis, and interpretation with critical revision of the final article for an intellectual concept. Ayodeji Anike Olatunji contributed to the design and data acquisition and analysis. Racheal Adeyanju Akinola contributed to the manuscript conception and design, interpreted the data, and reviewed the manuscript. Olatunde Odusan contributed immensely to the data conception, design, data analysis, and interpretation, and revised the final manuscript thoroughly with intellectual concept input. Adesola Olubunmi Adekoya contributed to the conception, design, data analysis, and interpretation by thoroughly revising the work for important intellectual content. Mojisola Adejoke Olusola-Bello designed the manuscript, worked with data collection, and analysis, and critically revised the final manuscript. Olatunbosun Oladipupo Olawale contributed to the data analysis and thoroughly carried out all laboratory analyses. All the authors have read and agreed to the final manuscript.
The authors acknowledge Dr. Fatade Oluwatosin for his immense contribution to the data collection of this article.
Table 1: socio-demographics of the study population
Table 2: mean and proportional distribution of clinical and lab parameters along the gender divide
Table 3: mean value of ultrasonographic parameters of the study population along the gender line
Table 4: correlation coefficients between traditional risk factors and carotid intima-media thickness, peak systolic velocity, end-diastolic velocity, pulsatility index, and resistance index of participants
Figure 1: B-mode, longitudinal ultrasound of the carotid artery showing the intima-media thickness in between the white arrows
- World Health Organization. Global Health Observatory Data Repository-Mortality and burden of disease-WHO regions. Geneva: World Health Organization; 2008.
- Mendis S, Puska P, Norrving BE. Global atlas on cardiovascular disease prevention and control. Geneva: World Health Organization; 2011. Google Scholar
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M et al. Heart Disease and Stroke Statistics - 2016 update: a report from the American Heart Association Circulation. 2016 Jan 26;133(4):e38-360. PubMed | Google Scholar
- Dichgans M, Pulit SL, Rosand J. Stroke genetics: discovery, biology, and clinical applications. Lancet Neurol. 2019;18(6):587-599. PubMed | Google Scholar
- McGill HC Jr, McMahan CA, Gidding SS. Preventing heart disease in the 21st century: implications of the Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Study. Circulation. 2008 Mar 4;117(9):1216-27. PubMed | Google Scholar
- Ware JH. The limitations of risk factors as prognostic tools. N Engl J Med. 2006 Dec 21;355(25):2615-7. PubMed | Google Scholar
- Salonen R, Salonen JT. Progression of carotid atherosclerosis and its determinants: A population-based ultrasonography study. Atherosclerosis. 1990 Feb;81(1):33-40. PubMed | Google Scholar
- Sillesen H, Falk E. Why not screen for subclinical atherosclerosis? Lancet. 2011 Aug 20;378(9792):645-6. PubMed | Google Scholar
- Fernandez-Friera L, Penalvo JL, Fernandez-Ortiz A, Ibanez B, Lopez-Melgar B, Laclaustra M et al. Prevalence, vascular distribution, and multi-territorial extent of subclinical atherosclerosis in a middle-aged cohort the PESA (Progression of Early Subclinical Atherosclerosis) Study. Circulation. 2015 Jun 16;131(24):2104-13. PubMed | Google Scholar
- Sillesen H, Muntendam P, Adourian A, Entrekin R, Garcia M, Falk E et al. Carotid plaque burden as a measure of subclinical atherosclerosis: comparison with other tests for the subclinical arterial disease in the high-risk plaque bio-image study. JACC Cardiovasc Imaging. 2012 Jul;5(7):681-9. PubMed | Google Scholar
- Holewijn S, den Heijer M, Stalenhoef AF, De Graaf J. Non-invasive measurements of atherosclerosis (NIMA): current evidence and future perspectives. Neth J Med. 2010 Dec;68(12):388-99. PubMed | Google Scholar
- Greenland P, Alpert JS, Beller GA, Benjamin EJ, Budoff MJ, Fayad ZA et al. 2010 ACCF/AHA Guideline for Assessment of Cardiovascular Risk in Asymptomatic Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2010 Dec 14;56(25):e50-103. PubMed | Google Scholar
- van der Meer IM, Bots ML, Hofman A, del Sol AI, van der Kuip DA, Witteman JC. Predictive value of non-invasive measures of atherosclerosis for incident myocardial infarction: the Rotterdam Study. Circulation. 2004 Mar 9;109(9):1089-94. PubMed | Google Scholar
- Otsuka F, Sakakura K, Yahagi K, Joner M, Virmani R. Has our understanding of calcification in human coronary atherosclerosis progressed? Arterioscler Thromb Vasc Biol. 2014 Apr;34(4):724-36. PubMed | Google Scholar
- Polak JF, O´Leary DH. Carotid Intima-Media Thickness as Surrogate for and Predictor of CVD. Global Heart. 2016 Sep;11(3):295-312.e3. PubMed | Google Scholar
- Komorovsky R, Desideri A. Carotid ultrasound assessment of patients with coronary artery disease: a useful index for risk stratification. Vasc Health Risk Manag. 2005;1(2):131-6. PubMed | Google Scholar
- Sillesen H, Sartori S, Sandholt B, Baber U, Mehran R, Fuster V. Carotid plaque thickness and carotid plaque burden predict future cardiovascular events in asymptomatic adult Americans. Eur Heart J Cardiovasc Imaging. 2018 Sep 1;19(9):1042-1050. PubMed | Google Scholar
- Baber U, Mehran R, Sartori S, Schoos MM, Sillesen H, Muntendam P et al. Prevalence, impact, and predictive value of detecting subclinical coronary and carotid atherosclerosis in asymptomatic adults. J Am Coll Cardiol. 2015 Mar 24;65(11):1065-74. PubMed | Google Scholar
- Public Health Intelligence, with assistance from Professor Mike Marfell-Jones, UCOL Ministry of Health. Protocols for collecting Height, Weight, and Waist Measurements in New Zealand Health Monitor (NZHM) surveys. Wellington: Ministry of Health. 2008;2-4. Accessed 18th September, 2020.
- Ogunlade O, Adalumo OA, Asafa MA. Challenges of body mass index classification: New criteria for young adult Nigerians. Niger J Health Sci. 2015;15(2):71-74. Google Scholar
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation. Geneva, 8-11. 2008;5-7. Accessed on 18th September, 2020.
- Okafor CI, Raimi TH, Gezawa ID, Sabir AA, Enang O, Puepet F et al. Performance of waist circumference and proposed cut-off levels for defining overweight and obesity in Nigerians. Ann Afr Med. 2016 Oct-Dec;15(4):185-193. PubMed | Google Scholar
- American Diabetes Association. Diagnosis, and Classification of Diabetes Mellitus. Diabetes Care. 2010 Jan;33 Suppl 1(Suppl 1):S62-9. PubMed | Google Scholar
- Healthline. The recommended cholesterol levels by age. Accessed on 8th September, 2020.
- Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N et al. Mannheim carotid intima-media thickness and plaque consensus (2004-2006- 2011). An update on behalf of the advisory board of the 3rd, 4th, and 5th watching the risk symposia, at the 13th, 15th, and 20th European Stroke Conferences, Mannheim, Germany, 2004, Brussels, Belgium, 2006, and Hamburg, Germany, 2011. Cerebrovasc Dis. 2012;34(4):290-6. PubMed | Google Scholar
- Stein JH, Kocarz CE, Hurst RT, Lonn E, Christopher BK. Use of Carotid Ultrasound to Identify Subclinical Vascular Disease and Evaluate Cardiovascular Disease Risk: A Consensus Statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. J Am Soc Echocardiogr. 2008 Feb;21(2):93-111; quiz 189-90. PubMed | Google Scholar
- Pourcelot L. Applications cliniques de l´examen Doppler transcutan. Velocimetrie Ultrasonore Doppler. 1975:213-240. Google Scholar
- Gosling RD, King DH. Arterial assessment by Doppler-shift Ultrasound. Proc R Soc Med. 1974 Jun;67(6 Pt 1):447-9. PubMed | Google Scholar
- Omisore AD, Famurewa OC, Komolafe MA, Asaleye CM, Fawale MB, Afolabi BI. Association of traditional cardiovascular risk factors with carotid atherosclerosis among adults at a teaching hospital in south-western Nigeria. Cardiovasc J Afr. 2018;29(3):183-188. PubMed | Google Scholar
- Akintunde AA, Salawu AA, Opadijo OG. Prevalence of traditional cardiovascular risk factors among the staff of Ladoke Akintola University of Technology, Ogbomoso, Nigeria. Niger J Clin Pract. 2014 Nov-Dec;17(6):750-5. PubMed | Google Scholar
- Baba MM, Talle MA, Ibinaiye PO, Abdul H, Buba F. Carotid Intima-Media Thickness in Patients with Diabetes Mellitus Attending Tertiary Care Hospital in Nigeria. Angiol. 2018;6(210):2. Google Scholar
- Bulut A, Avci B. Carotid intima-media thickness values are significantly higher in patients with prediabetes compared to normal glucose metabolism. Medicine (Baltimore). 2019 Nov;98(44):e17805. PubMed | Google Scholar
- Koc AS, Sumbul HE. Increased aortic intima-media thickness may be used to detect macrovascular complications in adult type II diabetes mellitus patients. Cardiovasc Ultrasound. 2018 Jun 12;16(1):8. PubMed | Google Scholar
- Ku DN, Giddens DP, Zarins CK, Glagov S. Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arteriosclerosis. 1985 May-Jun;5(3):293-302. PubMed | Google Scholar
- Okeahialam BN, Alonge BA, Pam SD, Puepet FH. Carotid intima-media thickness as a measure of cardiovascular disease burden in Nigerian Africans with hypertension and diabetes mellitus. Int J Vasc Med. 2011;2011:327171. PubMed | Google Scholar
- Ibinaiye PO, Kolade-Yunusa HO, Abdulkadir A, Yunusa T. Relationship of carotid artery intima-media thickness to blood pressure, age, and body mass index of hypertensive adult patients. Arch Int Surg. 2015 Apr 1;5(2):63. Google Scholar
- Hong H, Wang H, Liao H. Prehypertension is associated with an increased carotid atherosclerotic plaque in the community population of Southern China. BMC Cardiovascular Disorders. 2013 Mar 19;13:20. PubMed | Google Scholar
- Chuang SY, Bai CH, Chen JR, Yeh WT, Chen HJ, Chiu HC et al. Common carotid end-diastolic velocity and intima-media thickness jointly predict ischemic stroke in Taiwan. Stroke. 2011 May;42(5):1338-44. PubMed | Google Scholar
- Chuang SY, Bai CH, Cheng HM, Chen JR, Yeh WT, Hsu PF et al. Common carotid artery end-diastolic velocity is independently associated with future cardiovascular events. Eur J Prev Cardiol. 2016 Jan;23(2):116-24. PubMed | Google Scholar
- Park J, Na Y, Jang Y, Park SY, Park H. Correlation of pre-hypertension with carotid artery damage in middle-aged and older adults. Int J Environ Res Public Health. 2020 Oct 21;17(20):7686. PubMed | Google Scholar
- Chuang SY; Cheng HM, Bai CH, Yeh WT, Chen JR, Pan WH. Blood Pressure, Carotid flow pulsatility, and the risk of stroke - a community-based study. Stroke. 2016 Sep;47(9):2262-8. PubMed | Google Scholar
- Bai CH, Chen JR, Chiu HC, Pan WH. Lower blood flow velocity, higher resistance index, and larger diameter of extracranial carotid arteries are associated with ischemic stroke independently of carotid atherosclerosis and cardiovascular risk factors. J Clin Ultrasound. 2007 Jul-Aug;35(6):322-30. PubMed | Google Scholar
- Jiang YN, Kohara K, Hiwada K. Alteration of carotid circulation in essential hypertensive patients with left ventricular hypertrophy. J Hum Hypertens. 1998 Mar;12(3):173-9. PubMed | Google Scholar
- Wielicka M, Neubauer-Geryk J, Kozera G, Bieniaszewski L. Clinical application of pulsatility index. Med Res J. 2020;5(3):201-210. Google Scholar
- Shen W. Punyanitya M, Chen J, Gallagher D, Albu J, Pi-Sunyer X et al. Waist circumference correlates with metabolic syndrome indicators better than percentage fat. Obesity (Silver Spring). 2006 Apr;14(4):727-36. PubMed | Google Scholar
- Carr DB, Utzschneider KM, Hull RL, Kodama K, Retzlaff BM, Brunzell JD et al. Intra-abdominal fat is a major determinant of the National Cholesterol Education Program Adult Treatment Panel III criteria for the metabolic syndrome. Diabetes. 2004 Aug;53(8):2087-94. PubMed | Google Scholar
- Zhang A, Yao Y, Xue Z, Guo X, Dou J, Lv Y et al. A Study on the factors influencing triglyceride levels among adults in Northeast China. Sci Rep. 2018 Apr 23;8(1):6388. PubMed | Google Scholar
- Rydén M, Arner P. Subcutaneous adipocyte lipolysis contributes to circulating lipid levels. Arterioscler Thromb Vasc Biol. 2017 Sep;37(9):1782-1787. PubMed | Google Scholar
- Ishihara T, Watanabe M, Tanaka K, Nakamura K. Association of visceral adiposity with hypertension, dyslipidemia, and type 2 diabetes: a cross-sectional study among Japanese men and women. Kitasato Med J. 2011;41:10-18. Google Scholar