Characterization of Hashimoto´s thyroiditis in Sudanese children: a cross-sectional study at Gaafar Ibnauf Hospital, Khartoum
Ghassan Faisal Fadlalbari, Salwa Abdelbagi Musa, Samar Sabir Hassan, Areej Ahmed Ibrahim, Mohamed Ahmed Abdullah
Corresponding author: Ghassan Faisal Fadlalbari, Gaafar Ibnauf Pediatric Tertiary Hospital, Pediatric Endocrinology Unit, Khartoum, Sudan 
Received: 28 May 2022 - Accepted: 04 Nov 2022 - Published: 14 Feb 2023
Domain: Pediatric endocrinology
Keywords: Children, clinical features, Hashimoto' thyroiditis, outcome, Sudan
©Ghassan Faisal Fadlalbari et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Ghassan Faisal Fadlalbari et al. Characterization of Hashimoto´s thyroiditis in Sudanese children: a cross-sectional study at Gaafar Ibnauf Hospital, Khartoum. Pan African Medical Journal. 2023;44:86. [doi: 10.11604/pamj.2023.44.86.35649]
Available online at: https://www.panafrican-med-journal.com//content/article/44/86/full
Research 
Characterization of Hashimoto´s thyroiditis in Sudanese children: a cross-sectional study at Gaafar Ibnauf Hospital, Khartoum
Characterization of Hashimoto´s thyroiditis in Sudanese children: a cross-sectional study at Gaafar Ibnauf Hospital, Khartoum
![]() Ghassan Faisal Fadlalbari1,&, Salwa Abdelbagi Musa1,
Ghassan Faisal Fadlalbari1,&, Salwa Abdelbagi Musa1, ![]() Samar Sabir Hassan1, Areej Ahmed Ibrahim1, Mohamed Ahmed Abdullah1,2
Samar Sabir Hassan1, Areej Ahmed Ibrahim1, Mohamed Ahmed Abdullah1,2
&Corresponding author
Introduction: literature on Hashimoto´s thyroiditis, the common thyroid illness in the young populations, in Sudan and Africa is scarce. We aimed to study its clinical profile and outcome among Sudanese children and adolescents.
Methods: records of 73 patients were reviewed. Data related to demographics, presenting features, family history and coexistence of autoimmune diseases, physical examination findings, and biochemical progression over time were obtained.
Results: patients´ mean age at the diagnosis was 10.6 ± 2.9 years, 80.8% (n = 59) of them were female and 83.6% (n = 61) were residing in iodine-sufficient areas. The commonest presenting features were thyromegaly and fatigability (79.5%, n = 58 and 43.8%, n = 32, respectively) after an illness duration of 0.5-48 months. Autoimmune comorbidities were documented in 8.2% (n = 6) of our series and more than half (53.4%, n = 39) of them were pre-pubertal at the diagnosis. Sixty point three percent (60.3%) (n = 44), 20.5% (n = 15), 13.7% (n = 10) and 5.5% (n = 4) of patients presented with overt hypothyroidism, sub-clinical hypothyroidism, euthyroidism and hyperthyroidism respectively, and there were no significant differences in the clinical profile between them. In patients´ continued follow-up, 94.1% (n = 32/34) of those presented with overt hypothyroidism required levothyroxine therapy to maintain euthyroidism for 0.5-13 years, while 85.7% (n = 6/7) of those with euthyroidism remained so for 0.5-6 years. Remission was reported in all hyperthyroid patients and in only 5.9% (n = 2/34) of those with overt hypothyroidism at diagnosis. The majority of our patients with subclinical hypothyroidism were treated with levothyroxine and continued to be euthyroid for 10 months to 13 years.
Conclusion: goiter was the commonest presenting feature of Hashimoto´s thyroiditis. The majority of patients had overt or subclinical hypothyroidism and almost all of them required long-term levothyroxine therapy.
Hashimoto´s thyroiditis is a common acquired thyroid disorder, particularly in iodine-sufficient areas (ISA), with a prevalence of 1-3% in children [1-5]. Recently, it showed a rising incidence which has been referred to the advancement in thyroid autoantibody testing technology and availability of fine needle aspiration facilities [2].
Hashimoto´s thyroiditis is diagnosed more frequently in females in early or middle adolescence with a (females:males) F:M ratio of 2-5.1:1 [4,6,7]. It results from cellular-mediated progressive destruction and fibrous replacement of the thyroid which might lead to thyromegaly or atrophy [8-11]. Furthermore, autoantibodies are formed against several thyroid-specific antigens, commonly thyroid peroxidase and thyroglobulin [12]. Hashimoto´s thyroiditis tends to be co-morbid with other non-thyroidal autoimmune diseases (NTADs) or to run in families [6]. The genetic predisposition of Hashimoto´s thyroiditis is emphasized by its aggregation in certain families and several genes are believed to be involved [6,8]. Thyroid enlargement and fatigue are the commonest presenting features, however thyroid status at onset and in long-term shows variation between different studies [3,4,6,7,13,14].
The authors aimed to study the clinical presentation, associated autoimmune diseases (ADs), and progression of Hashimoto´s thyroiditis in the Sudanese young population. No such data were previously published from Sudan.
Study design and setting: this was a retrospective records review of patients who were diagnosed to have Hashimoto´s thyroiditis and presented to the Pediatric Endocrinology Unit at Gaafar Ibnauf Hospital in the period from January 2006 to November 2020. It is the only tertiary pediatric endocrinology center in the country and referrals are received from all over Sudan.
Study population: all records of patients less than 18-year-old at presentation, had thyroid enlargement and/or symptoms of hypo- or hyperthyroidism (with no characters suggestive of Graves´ disease) with biochemical and/or thyroid ultrasonography features of Hashimoto´s thyroiditis were collected. Those with an incomplete record, who were less than four years at presentation, had unexplained learning disabilities, no documentation for anti-thyroperoxidase (anti-TPO) and/or anti-thyroglobulin (anti-TG) testing, or coming from iodine-deficient areas (IDAs) with no biochemical or ultrasound evidence of Hashimoto´s thyroiditis were excluded.
Data collection: data related to demographic characters, presenting clinical and biochemical features, the coexistence of NTADs, family history of thyroid or other ADs, anthropometric measures, physical findings and workup results were collected using a predesigned data collection sheet. The thyroid gland was graded according to World Health Organization (WHO) criteria to G0 if not palpable and not visible, G1 if palpable but not visible, and G2 if visible. In addition, its consistency and surface characters on palpation were described. Patients were classified into pre-pubertal or pubertal according to their puberty status at presentation. Delayed puberty was defined as having no secondary sexual characteristics by the age of 13 in girls and 14 in boys. Thyroid function test (TFT), anti-TPO, and anti-TG were done on a commercial basis and results were considered abnormal in comparison to the laboratory reference ranges (LRR). Missing data and the last TFT were collected by contacting patients and/or their caregivers.
Definitions: study participants were divided into four functional subgroups according to their initial TFT. Subgroups were defined as follows: euthyroidism if thyroid-stimulating hormone (TSH) was 0.2-5 mU/L and free thyroxine (FT4) was within the LRR, sub-clinical hypothyroidism if TSH was >5 mU/L and FT4 was within the LRR, overt hypothyroidism if TSH was >10 mU/L and FT4 was below the LRR and hyperthyroidism if TSH was <0.1 mU/L and FT4 was above the LRR. The evolution of the thyroid function was assessed in those who were consistent with follow-up for at least six months.
Statistical analysis: the Statistical Package for the Social Sciences (SPSS) version 25, a computer-based software, was used to enter and analyze data. Results were shown in mean/median or range for numeric variables and frequencies and percentages for categorical data. The comparison between subgroup variables was carried out using analysis of variance (ANOVA) for normally distributed data, Kruskal-Wallis for skewed continuous data and Chi-square for categorical data. Significance was considered at a p-value of <0.05.
Ethical considerations: the study protocol was approved by the endocrine institutional review board at Sudan Childhood Diabetes Center (approval reference: ENDO-SCDC-IRB-21117).
Demographic and presenting features: one-hundred and twelve patients were provisionally diagnosed to have Hashimoto´s thyroiditis. Those who (n=39) were diagnosed clinically and/or based on ultrasonography findings without testing for thyroid autoantibodies and/or having incomplete records were excluded. A record of 73 patients met the inclusion criteria and were furtherly studied. The mean (range) age at presentation was 10.6±2.9 (4-17) years with a female to male ratio of 4.2:1. Table 1 shows the descriptive data of the studied population at the diagnosis. The majority of the patients were residing in ISAs. Neck swelling and goiter were the commonest presenting complaint and rational for referral to our clinic. The median (range) duration of the illness before the presentation was five (0.5-48) months. Short stature, wasting and obesity were detected in 17.8% (n=13), 12.3% (n=9) and 11% (n=8), respectively. More than half of the patients (n=39) were pre-pubertal at presentation, two of them were having delayed puberty.
The thyroid gland was diffusely enlarged in the majority (82.2%, n=60) of the study population, however, it was neither visible nor palpable in 13 (17.8%) patients (Table 1). Among those with goiter (n=60), the thyroid consistency was firm in 32 (53.3%), soft in 28 (46.7%) and the thyroid gland surface was smooth in 52 (86.7%), nodular in three (5%) and multi-nodular goiter were reported in five (8.3%) of them. At presentation, 60.3% (n=44) of patients had overt hypothyroidism, 20.5% (n=15) had subclinical hypothyroidism, 13.7% (n=10) were euthyroid and 5.5% (n=4) presented with the thyrotoxicosis (Table 1). Anti-TPO was the only tested autoantibody in the majority of the study participants (72.6%, n=53). Only 27.3% (n=20) of our patients were able to test for both anti-TPO and anti-TG, 15 had elevated both anti-TPO and anti-TG and 5 had elevated anti-TG only. The most common finding on thyroid ultrasonography scan, among those who were able to do it (n=34/73), was diffuse goiter with multiple hypoechoic areas (Table 1).
There was no significant difference between the functional subgroups of the study population in the demographic features, the coexistence of NTAD, family history of autoimmune thyroid diseases (AITD), anthropometry, goiter grade or pubertal status (Table 2).
Coexistence and family history of ADs: coexistence of NTADs were documented in 8.2% (n=6) of our cohort which included type 1 diabetes (T1D) (n=1), celiac disease (CD) (n=1), systemic lupus erythematosus (SLE) (n=1), autoimmune myopathy (n=1), myasthenia gravis (n=1), and one patient had T1D, SLE and Addison´ disease. While in two-third (n=4/6) of them, the diagnosis of Hashimoto´s thyroiditis preceded the onset of the NTAD by a median (range) of 30 (10-42) months, the other two patients were diagnosed to have Hashimoto´s thyroiditis with SLE and Hashimoto´s thyroiditis with myasthenia gravis simultaneously at presentation. ADs were reported in 29 (39.7%) families of our patients, including Hashimoto´s thyroiditis (n=22), Graves´ disease (n=4), SLE (n=1), Hashimoto´s thyroiditis/SLE (n=1), and Hashimoto´s thyroiditis/CD (n=1).
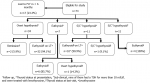
Thyroid function evolution over time: twenty-three point three percent (23.3%, n=17) of our patients dropped out of the follow-up in less than 6 months and have been excluded from the assessment of the outcome (Figure 1). All patients with overt hypothyroidism at onset (n=34) were treated with levothyroxine, 94.1% (n=32) of them remained euthyroid while on levothyroxine for 0.5-13 years and 5.9% (n=2) stopped the therapy and had complete remission. On the other side, those who presented with hyperthyroidism (n=4) went into euthyroidism in 3.5 ± 1.1 (2-5) months with propranolol only. The majority of patients with euthyroidism at onset (n=6/7) continued to be so for 0.5-6 years duration of the follow-up and only one patient developed overt hypothyroidism five years after the diagnosis. Out of the 11 patients who presented with subclinical hypothyroidism, ten patients had a TSH of >10 mU/L and were treated with levothyroxine with an average dose of 1.31 ± 0.38 mcg/kg/d. All of them became euthyroid in three to ten months while on levothyroxine therapy and remained euthyroid for 10 months to 13 years. The remaining patient with subclinical hypothyroidism at onset had a TSH of 7.6 mU/L, was not treated with levothyroxine, and remained to have subclinical hypothyroidism during the one-year follow-up duration.
Hashimoto´s thyroiditis is a common thyroid disorder that we encounter in our clinic. Studying the mode of its presentation and evolution in our population will be valuable in the short and long-term management planning as well as counseling of patients and/or their caregivers. This study showed that Hashiomto´s thyroiditis is more prevalent among prepubertal females and those living in ISAs. Moreover, thyromegaly was the commonest presenting feature and the majority had impaired thyroid function. To the best of our knowledge, this is among the first studies in Africa that characterized its clinical profile among children [15].
Hashimoto´s thyroiditis, in our cohort, was significantly more prevalent in females (80%) which is consistent with different international and regional reports [3,8,13,15]. The ADs predilection towards females is well known, Hashimoto´s thyroiditis is not an exception, and that may be related to the biased X-chromosome inactivation, female sex hormone or immune system response [16-18]. The mean age of our group at the diagnosis was 10.6 ± 2.9 years which is in the line with different studies [13-15]. Sudan is a big country, many regions are known to be IDAs, therefore it was interesting to explore whether Hashimoto´s thyroiditis is more prevalent among those living in ISAs or IDAs. Eighty-three point six percent of our patients were residing in ISAs that agreed with some epidemiological studies [19,20].
Thyroid enlargement was the commonest consultation in our clinic similar to many published reports [4,15,21,22]. However, goiter was not detected in 17.8% of our cohort, non-goitrous form of Hashimoto´s thyroiditis has been described in children, consistent to a recent report from Egypt (16.8%) [15]. A significant proportion of our patients (43.8%) had fatigability and this was the commonest presenting complaint of 122 pediatric Hashimoto´s thyroiditis patients in Ibili et al. study [13]. It might be explained by the thyroid dysfunction, however, the autoimmune process itself is a possible contributing factor [23]. The prevalence of short stature among our cohort was 17.8% which is close to that reported in the Egyptian study (18.7%) [15].
In contrast to the literature, that most of the patients are diagnosed at their mid or late puberty, more than half of this study participants (n=37) were pre-pubertal excluding those who had delayed puberty (n=2) [8,13,24]. Furthermore, 60.3% of our series presented with overt hypothyroidism which is comparable to the Egyptian study findings (67.8%) [15]. However, many studies from developed world claimed that euthyroidism is the commonest mode of presentation [3,4,25,26]. These regional variations suggest that other factors, like environmental factors or endocrine disruptor exposure, may modulate the presentation mode of Hashimoto´s thyroiditis among children in our community [27]. Autoimmune diseases (ADs) are characterized by their tendencies to aggregate on the same patient and family. In this research, the prevalence of NTAD was 8.2% in comparison to 6.5% by Ibili et al. [13]. However, a higher prevalence (18.8%) has been reported in a larger young population with Hashimoto´s thyroiditis (n=553) [28]. Although some authors suggest screening for NTAD in children with AITD, the argument probably would be against it in limited-resource settings due to testing costs [3]. The high prevalence of ADs including AITDs in antecedents of Hashimoto´s thyroiditis patients in this research (39.7%), similar to 40% and 54.4% reported by Admoni and Calcaterra et al. respectively, emphasizes the implication of genetic factors on its pathogenesis [3,8].
This study didn´t detect significant differences between functional subgroups in demographics, presenting clinical and biochemical features. That is in agreement with Özsu et al. and Dündar et al. studies which failed to show significant differences in ages, gender, and absence of goiter between different subgroups among 106 and 78 pediatric patients with Hashimoto´s thyroiditis, respectively [22,29]. Nonetheless, later research revealed that goiter was more prevalent in those who were euthyroid at onset [29]. In addition, de Vries et al. study didn´t show a difference in height or body mass index (BMI) of their 112 Hashimoto´s thyroiditis patients in relation to their biochemical presentation [21]. However, contrary to our findings and those of de Vries et al. those with overt hypothyroidism at presentation were younger than their peers on some reports [3,4,21,30]. Furthermore, the ratio of pubertal to pre-pubertal participants was higher amongst those who were euthyroid at diagnosis [4,30]. Different sample sizes and definitions of the functional subgroups may explain the variations between these studies.
Few studies have addressed the long-term progress of childhood Hashimoto´s thyroiditis [31]. In sense of the urgent need for hormonal replacement of thyroid failure, all our patients with overt hypothyroidism at diagnosis (n=34) were treated with levothyroxine. Most of them (94.1%) required the therapy to remain euthyroid during the follow-up period which is comparable to the Gopalakrishnan et al. and Admoni et al. findings [2,3]. However, the higher rate of remission in the latter study (16%), even higher in other studies, suggests that levothyroxine is not necessary to be a lifelong therapy in hypothyroid Hashimoto´s thyroiditis patients, and it is worth reassessing the TFT while off the levothyroxine later in the course of the illness [3,32,33].
It is rare for Hashimoto´s thyroiditis to present with hyperthyroidism, 5.5% in this research compared to 3.5% on other reports, that almost always remit spontaneously in a short time with no subsequent relapse [3,30,31]. Therefore, increasing awareness about the condition among health practitioners is essential to avoid unnecessary carbimazole initiation. Most of our patients with euthyroidism at onset continued to have so for up to 6 years, however due to the risk of developing overt hypothyroidism, 14.3% in our series compared to 12.3-26% in the literature, long-term TFT monitoring is needed [3,34]. Ninety point nine percent of our patients with subclinical hypothyroidism at onset had a TSH >10 mIU/L, they were considered to have a compensated hypothyroidism and all of them required the levothyroxine to maintain euthyroidism for up to 13 years. Although there is no consensus guideline on the management of childhood and juvenile subclinical hypothyroidism so far, Crisafulli et al. have shown that the long-term outcome may be unfavorable in those with a TSH >10 mIU/L [35]. Moreover, an elevated TSH was found to have a possible role in the development of thyroid malignancy in patients with Hashimoto´s thyroiditis [36].
The retrospective nature of this research and the discrepancy in the follow-up duration between the participants are among the limitations. Moreover, around one-third of our cohort has been excluded due to the challenges of confirming the diagnosis. Barriers were including the cost and periodic unavailability of thyroid autoantibodies testing (around 50 US dollars in Sudan). Furthermore, in countries like Sudan where the consanguineous marriage rate is high, late-onset thyroid dyshormonogenesis cannot be ruled out without testing for thyroid autoantibodies [37].
This study showed that goiter is the commonest presenting feature of Hashimoto´s thyroiditis. Therefore, testing for thyroid autoantibodies is paramount in children who present with thyroid enlargement. Unlike reports from developed countries where most of the patients are euthyroid at diagnosis, the majority of our cases had either overt or subclinical hypothyroidism requiring levothyroxine to remain euthyroid for the entire follow-up period.
What is known about this topic
- Hashimoto´s thyroiditis is the commonest acquired thyroid disorder in iodine-sufficient areas and more prevalent in females at mid or late puberty;
- Goiter and fatigability are the commonest presenting features and most of the patients in developed countries are euthyroid at the diagnosis;
- Some reports revealed a younger age and more prevalent goiter in those presented with overt hypothyroidism and euthyroidism, respectively.
What this study adds
- More than half of our cohort were pre-pubertal;
- The majority of our patients presented with thyroid impairment either overt or sub-clinical hypothyroidism requiring levothyroxine to remain euthyroid during the follow-up period;
- Our study failed to show any significant differences in the patients´ clinical profile in relation to their presenting thyroid function status.
The authors declare no competing interests.
Conception and study design: Ghassan Faisal Fadlalbari, Salwa Abdelbagi Musa, Samar Sabir Hassan, Areej Ahmed Ibrahim and Mohamed Ahmed Abdullah; data collection: Ghassan Faisal Fadlalbari and Salwa Abdelbagi Musa; data analysis and interpretation, and manuscript drafting: Ghassan Faisal Fadlalbari; manuscript revision: Salwa Abdelbagi Musa, Samar Sabir Hassan, Areej Ahmed Ibrahim and Mohamed Ahmed Abdullah; guarantor of the study: Ghassan Faisal Fadlalbari. All the authors read and approved the final version of this manuscript.
We would like to expand our gratitude to medical records officers at Gaafar Ibnauf Hospital, Mr. Mutassim Fadol and our colleagues at pediatric endocrinology department for their great help in collecting data of this research.
Table 1: clinical characteristics at presentation (n=73)
Table 2: comparison between functional subgroups at presentation in relation to demographic data, history and physical findings (n=73)
Figure 1: long term outcome of the study participants
- Zdraveska N, Kocova M. Hashimoto thyroiditis in childhood-review of the epidemiology, genetic susceptibility and clinical aspects of the disease. Maced J Med Sci. 2012;5(3):336-45. Google Scholar
- Gopalakrishnan S, Chugh PK, Chhillar M, Ambardar VK, Sahoo M, Sankar R. Goitrous autoimmune thyroiditis in a pediatric population: a longitudinal study. Pediatrics. 2008;122(3):e670-4. PubMed | Google Scholar
- Admoni O, Rath S, Almagor T, Elias-Assad G, Tenenbaum-Rakover Y. Long-Term Follow-Up and Outcomes of Autoimmune Thyroiditis in Childhood. Front Endocrinol (Lausanne). 2020;11:309. PubMed | Google Scholar
- Tuhan H, Isik S, Abaci A, Simsek E, Anik A, Anal Ö et al. Celiac disease in children and adolescents with Hashimoto Thyroiditis. Turk Pediatri Ars. 2016;51(2):100-5. PubMed | Google Scholar
- Penta L, Cofini M, Lanciotti L, Leonardi A, Principi N, Esposito S. Hashimoto´s Disease and Thyroid Cancer in Children: Are They Associated? Front Endocrinol. 2018;9:565. PubMed | Google Scholar
- Pomahacova R, Zamboryova J, Paterova P, Fiklik K, Cerna Z, Lad V et al. Autoimmune disease, familial clustering and thyroid carcinoma coexistent with autoimmune thyroiditis in children and adolescence: a cross-sectional study from the Czech Republic. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2018. PubMed | Google Scholar
- Baruah MP, Duttachoudhury S, Saikia M, Saikia UK, Bhuyan SB, Bhowmick A et al. Guwahati thyroid epidemiology study: High prevalence of primary hypothyroidism among the adult population of Guwahati city. Thyroid Research and Practice. 2019;16(1):12-9. Google Scholar
- Calcaterra V, Nappi RE, Regalbuto C, De Silvestri A, Incardona A, Amariti R et al. Gender Differences at the Onset of Autoimmune Thyroid Diseases in Children and Adolescents. Front Endocrinol (Lausanne). 2020 Apr 17;11:229. PubMed | Google Scholar
- Won JH, Lee JY, Hong HS, Jeong SH. Thyroid nodules and cancer in children and adolescents affected by Hashimoto´s thyroiditis. Br J Radiol. 2018;91(1087):20180014. PubMed | Google Scholar
- Farghaly HS, Metwalley KA, Ahmed FA, Raafat DM, El-Asheer O, Ali AM et al. Visfatin level in children and adolescents with autoimmune thyroiditis. Ther Adv Endocrinol Metab. 2017;8(8):119-25. PubMed | Google Scholar
- Jeong SH, Hong HS, Lee JY. The association between thyroid echogenicity and thyroid function in pediatric and adolescent Hashimoto´s thyroiditis. Medicine (Baltimore). 2019;98(14):e15055. PubMed | Google Scholar
- Brown RS. Autoimmune thyroiditis in childhood. J Clin Res Pediatr Endocrinol. 2013;5 Suppl 1(Suppl 1):45-9. PubMed | Google Scholar
- Ibili ABP, Selver Eklioglu B, Atabek ME. General properties of autoimmune thyroid diseases and associated morbidities. J Pediatr Endocrinol Metab. 2020;33(4):509-15. PubMed | Google Scholar
- De Luca F, Santucci S, Corica D, Pitrolo E, Romeo M, Aversa T. Hashimoto´s thyroiditis in childhood: presentation modes and evolution over time. Ital J Pediatr. 2013;39:8. PubMed | Google Scholar
- Metwalley KA, Metwalley MKA. Pattern of autoimmune thyroiditis in Eygptian children and adolescence. Thyroid Research and Practice. 2020;17(1):7-13. Google Scholar
- Ozcelik T, Uz E, Akyerli CB, Bagislar S, Mustafa CA, Gursoy A et al. Evidence from autoimmune thyroiditis of skewed X-chromosome inactivation in female predisposition to autoimmunity. Eur J Hum Genet. 2006 Jun;14(6):791-7. PubMed | Google Scholar
- Simmonds MJ, Kavvoura FK, Brand OJ, Newby PR, Jackson LE, Hargreaves CE et al. Skewed X chromosome inactivation and female preponderance in autoimmune thyroid disease: an association study and meta-analysis. J Clin Endocrinol Metab. 2014;99(1):E127-31. PubMed | Google Scholar
- Ngo ST, Steyn FJ, McCombe PA. Gender differences in autoimmune disease. Front Neuroendocrinol. 2014;35(3):347-69. PubMed | Google Scholar
- Ragusa F, Fallahi P, Elia G, Gonnella D, Paparo SR, Giusti C et al. Hashimotos' thyroiditis: Epidemiology, pathogenesis, clinic and therapy. Best Pract Res Clin Endocrinol Metab. 2019 Dec;33(6):101367. PubMed | Google Scholar
- Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan CM et al. Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol. 2018;14(5):301-16. PubMed | Google Scholar
- de Vries L, Bulvik S, Phillip M. Chronic autoimmune thyroiditis in children and adolescents: at presentation and during long-term follow-up. Arch Dis Child. 2009;94(1):33-7. PubMed | Google Scholar
- ÖZSU CAVGA EL, Mutlu R, Çizmeci F, Hatun S. Characteristics of our patients with Hashimoto thyroiditis. Turk Arch Ped. 2011;46(3). Google Scholar
- Jordan B, Uer O, Buchholz T, Spens A, Zierz S. Physical fatigability and muscle pain in patients with Hashimoto thyroiditis. J Neurol. 2021;268(7):2441-2449. PubMed | Google Scholar
- Cappa M, Bizzarri C, Crea F. Autoimmune thyroid diseases in children. J Thyroid Res. 2010 Dec 14;2011:675703. PubMed | Google Scholar
- Skarpa V, Kousta E, Tertipi A, Anyfandakis K, Vakaki M, Dolianiti M et al. Epidemiological characteristics of children with autoimmune thyroid disease. Hormones (Athens). 2011;10(3):207-14. PubMed | Google Scholar
- DeBoer MD, LaFranchi S. Differential presentation for children with autoimmune thyroiditis discovered because of symptom development or screening. J Pediatr Endocrinol Metab. 2008;21(8):753-61. PubMed | Google Scholar
- Wiersinga WM. Clinical Relevance of Environmental Factors in the Pathogenesis of Autoimmune Thyroid Disease. Endocrinol Metab (Seoul). 2016;31(2):213-22. PubMed | Google Scholar
- Ruggeri RM, Trimarchi F, Giuffrida G, Certo R, Cama E, Campenn� A et al. Autoimmine comorbidities in Hashimoto´s thyroididtis: different pattern of association in adulthood and childhood/adolescence. Eur J Endocrinol. 2017 Feb;176(2):133-141. PubMed | Google Scholar
- Dundar B, Boyaci A, Sangun O, Dundar N. Hashimoto thyroiditis in children and adolescents: evaluation of clinical and laboratory findings. Turk Pediatri Ars. 2011;46(4):318-22. Google Scholar
- Wasniewska M, Corrias A, Salerno M, Mussa A, Capalbo D, Messina MF et al. Thyroid function patterns at Hashimoto´s thyroiditis presentation in childhood and adolescence are mainly conditioned by patients´ age. Horm Res Paediatr. 2012;78(4):232-6. PubMed | Google Scholar
- Crisafulli G, Gallizzi R, Aversa T, Salzano G, Valenzise M, Wasniewska M et al. Thyroid function test evolution in children with Hashimoto´s thyroiditis is closely conditioned by the biochemical picture at diagnosis. Ital J Pediatr. 2018;44(1):22. PubMed | Google Scholar
- Wang SY, Tung YC, Tsai WY, Lee JS, Hsiao PH. Long-term outcome of hormonal status in Taiwanese children with Hashimoto´s thyroiditis. Eur J Pediatr. 2006 Jul;165(7):481-3. PubMed | Google Scholar
- Demirbilek H, Kandemir N, Gonc EN, Ozon A, Alikasifoglu A. Assessment of thyroid function during the long course of Hashimoto´s thyroiditis in children and adolescents. Clin Endocrinol (Oxf). 2009 Sep;71(3):451-4. PubMed | Google Scholar
- Aversa T, Corrias A, Salerno M, Tessaris D, Di Mase R, Valenzise M et al. Five-Year Prospective Evaluation of Thyroid Function Test Evolution in Children with Hashimoto's Thyroiditis Presenting with Either Euthyroidism or Subclinical Hypothyroidism. Thyroid. 2016 Oct;26(10):1450-1456. PubMed | Google Scholar
- Crisafulli G, Aversa T, Zirilli G, Pajno GB, Corica D, De Luca F et al. Subclinical hypothyroidism in children: when a replacement hormonal treatment might be advisable. Front Endocrinol (Lausanne). 2019;10:109. PubMed | Google Scholar
- Fiore E, Rago T, Latrofa F, Provenzale MA, Piaggi P, Delitala A et al. Hashimoto´s thyroiditis is associated with papillary thyroid carcinoma: role of TSH and of treatment with L-thyroxine. Endocr Relat Cancer. 2011;18(4):429-37. PubMed | Google Scholar
- Bruellman RJ, Watanabe Y, Ebrhim RS, Creech MK, Abdullah MA, Dumitrescu AM et al. Increased Prevalence of TG and TPO Mutations in Sudanese Children With Congenital Hypothyroidism. J Clin Endocrinol Metab. 2020 May 1;105(5):1564-72. PubMed | Google Scholar