Situation analysis of urogenital bilharzia in West Africa (2010-2021) and control strategies and prospects: systematic review and meta-analysis
Josias Olutobi Ahamide, Charles Sossa, Yolande Sissinto, Virginie Mongbo, Victorien Dougnon, Boris Legba, Edgard-Marius Ouendo
Corresponding author: Josias Olutobi Ahamide, Institut Régional de Santé Publique, Université d'Abomey-Calavi, Abomey-Calavi, Bénin 
Received: 14 Feb 2022 - Accepted: 20 Dec 2022 - Published: 18 Jan 2023
Domain: Epidemiology,Infectious diseases epidemiology
Keywords: Bilharzia, strategies, prevalence, systematic review, meta-analysis, West Africa
©Josias Olutobi Ahamide et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Josias Olutobi Ahamide et al. Situation analysis of urogenital bilharzia in West Africa (2010-2021) and control strategies and prospects: systematic review and meta-analysis. Pan African Medical Journal. 2023;44:35. [doi: 10.11604/pamj.2023.44.35.33766]
Available online at: https://www.panafrican-med-journal.com//content/article/44/35/full
Review 
Situation analysis of urogenital bilharzia in West Africa (2010-2021) and control strategies and prospects: systematic review and meta-analysis
Situation analysis of urogenital bilharzia in West Africa (2010-2021) and control strategies and prospects: systematic review and meta-analysis
Josias Olutobi Ahamide1,&, ![]() Charles Sossa1, Yolande Sissinto2, Virginie Mongbo1,
Charles Sossa1, Yolande Sissinto2, Virginie Mongbo1, ![]() Victorien Dougnon3,
Victorien Dougnon3, ![]() Boris Legba3, Edgard-Marius Ouendo1
Boris Legba3, Edgard-Marius Ouendo1
&Corresponding author
Schistosomes are parasitic diseases caused by flatworms (schistosomes or bilharzia), transmitted in the urine or in the faeces, and involving intermediate hosts (freshwater molluscs). Their recrudescence in endemic areas is no longer in question and remains a crucial public health problem in the world in general and in West Africa in particular. In order to eradicate bilharzia, many control strategies and policies have been implemented on both sides. The objective of this systematic literature review is to synthesize the existing evidence on control strategies implemented by West African countries. To achieve this, data were collected from PubMed, Direct Science, Web of Sciences, Google Scholar, PloS and Banque de Données de Santé Publique (BDSP), using appropriate keywords. Academic articles and theses written in French or English that evaluated the analysis of a bilharzia situation in West Africa were selected. Sixteen scientific papers were selected for the study, ten of which were used for a meta-analysis. The systematic review revealed that bilharzia is still an endemic disease in West Africa. Clearly, it continues to wreak havoc on the population, especially among school children. Rural areas are the most affected by the disease. Strategies to control bilharzia are based on preventive and curative treatment of the infection with chemotherapy and vector control of soil molluscs (host and vector of bilharzia eggs). Praziquantel is the main known antibilharzian. Also, the species most frequently found in analyses are S. haematobium and S. mansonii. This review has allowed to evaluate the control strategies carried out and to deduce the strengths and weaknesses, in order to define the perspectives for the efficiency of the anti-bilharzia control for the eradication of bilharzia in the endemic zones of West Africa.
In sub-Saharan Africa, bilharzia or schistosomiasis is the second most prevalent and important neglected tropical disease in terms of public health, after hookworm [1-3]. The number of people exposed worldwide is estimated at 600 million, with more than 200 million people infected and nearly 280,000 deaths each year, 97% of which occur in Africa, south of the Sahara [2,3]. Schistosomiasis accounts for 1.9 million disability-adjusted life years (Dalys) per year [4] with 90% of the current burden concentrated in Africa. It is endemic in more than 70 countries and territories in the tropics and subtropics and therefore remains a major public health problem worldwide [5]. Schistosoma japonicum is widespread in China and parts of South-East Asia [6]. In addition, it has been reported that more than 40 species of wild and domestic animals can be infected with S. japonicum [7,8]. The last decade has been marked by an extraordinary surge of awareness and funding for neglected tropical diseases, especially bilharzia. On a large scale, the fight against schistosomiasis has become more than ever the concern of sovereign States, guarantors of the health of populations, international institutions as well as humanitarian and non-profit organizations.
However, the number of people still needing treatment is not encouraging [9]. The increasing incidence of the disease seems to be leading governments to intensify health policies in the area of prevention and care. Thus, the overall objective of current public health strategies for schistosomiasis is to reduce morbidity through preventive chemotherapy (PC) [10]. Large-scale periodic administration of praziquantel, focusing in this case on the school-age population but also on adults living in high-risk (highly endemic) areas, aims to reduce the prevalence and intensity of infection [11]. However, its usefulness is sometimes limited enough in areas with high rates of reinfection and resistance. In addition, the low susceptibility of Schistosoma mansoni and S. japonicum to Praziquantel (PZQ) has been induced by mass drug administration programmes [5,12]. The strategies implemented to control bilharzia in West Africa have certainly contributed to a decrease in the incidence of the disease, although the challenges remain [12,13]. From an optimization perspective, it is necessary to take stock of these strategies, to evaluate their impact, their strength and weakness. This situation motivates the authors who are trying to identify and analyse the gaps in the knowledge which is intended to be filled with this review. In fact, they plan to enumerate the assets, to evaluate the strengths and weaknesses of previous studies and make objective proposals to motivate public authorities and organizations involved in the fight against bilharzia to refocus control strategies on an integrated approach (sensitization-chemotherapy-sensitization), to strengthen the sanitary and behavioral hygiene system through the construction of social and health infrastructures in endemic areas.
Indeed, the following study intends to fill up the gaps in the knowledge developed by the formers authors as far as the strategies used to fight against bilharziasis are concerned. This systematic review has to synthesize existing evidence on control strategies carried out by West African countries. It is supposed to inventory the means and techniques implemented and to study their limitations in order to develop a more adequate control plan capable of reducing or even definitively eradicating bilharzia from endemic countries. More over the meta-analysis aims to combine numerical data from multiple separate studies from the reviews, to capitalize on what has been learned, to analyze in a descriptive way the global level of the phenomenon (bilharziasis) and its variation in West Africa and finally to identify the nature of the interventions implemented. Therefore, it has a particular design to consider the summary of the studies concerned and the robustness of the conclusion and the recommendation of the meta analysis. This study was initiated as a systematic literature review and meta-analysis to identify lists of bilharziasis reviewer studies and presents as overall objective to develop a systematic literature review and meta-analysis of interventional studies conducted on urogenital bilharzia 2010-2021 in West African countries. Specifically the study aims to: produce a synthesis of control strategies carried out in West Africa from 2010 to 2021 and determine the effect of interventions carried out in the framework of schistosome control from 2010 to 2021 in West African countries.
PRISMA guidelines [14] were used to conduct the systematic review. A literature search was conducted in the published literature (peer-reviewed journal articles) and grey literature (conference reports, theses and dissertations). Selected electronic databases including Google, Google Scholar, PubMed, Direct Science, Web of Sciences, PloS, Medline, and BDSP were targeted with appropriate keywords.
Eligibility and ineligibility criteria: a study is included if: it proposes a theme centred essentially on bilharziasis in general or urogenital bilharziasis in particular as well as anti-bilharzian control techniques (curative treatment and preventive control methods); it is taken into account by publications (journals and articles, theses and dissertations), in the period 2010 to 2020, and whose themes meet the expectations of the objectives of the systematic review and it is carried out in West Africa or sub-Saharan Africa. He was excluded from this study: studies on topics other than bilharzia; studies published in a language other than English, French; studies that represent just a narrative review; studies less than 2010 and those completed after 2020 and articles that do not accurately reflect the period of study.
Search strategy: the search was done in July 2021 and was conducted on scientific databases. Some Boolean operators were used to make the necessary interconnections to facilitate the selection of the best studies that best meet the journal writing. In accordance with the study question, a search equation was used in scientific databases. Almost all of the searches were done online and with syntax and keywords from the search theme. We decrypted results from a number of search engines, the main ones being PubMed, PloS, ScienceDirect, Web of Sciences, Google, Google Scholar and BDSP. Some Boolean operators were used to make the necessary interconnections to facilitate the selection of the best studies that best meet the editorial needs of the journal. The themes addressed in these different research areas concern NTDs in general and bilharzia in particular, as well as data from epidemiological studies. The research was later complemented by an epidemiological study done on bilharzia in Benin in 2020.
Selection of studies: the selection was made independently but on the basis of titles, abstracts and full texts, following the eligibility criteria listed above. The results were then pooled. Studies that may have been selected by only one or the other researcher were discussed for inclusion or rejection. Articles were included on the basis of the predefined selection criteria: publications addressing the targeted themes and published in French or English between 2010 and 2021; reports on estimates of the association between prevalence and the impact of socio-demographic and economic factors on the resurgence of bilharzia or the prevalence of the underlying infection, with a measure of statistical significance (e.g. p-value or 95% confidence interval) [15].
Risk of bias assessment: to avoid methodological errors and errors in the analysis of the data collected that would have systematic consequences on the quality of this literature review, biases were identified to assess the quality of the studies selected for the systematic review. These include confounding by poor analysis or by an incriminating factor that is not causally related to the study.
Data extraction: data extraction was done by simultaneous reading of the documents by the researchers. The data collected concerned the participants (number, age, gender, duration of the intervention, level of prosperity, socio-demographic factors) and the mode of transmission, anti-bilharzia strategies, inclusion criteria, and interventions (frequency, duration, types).
Study variables
Dependent variable: the dependent variable considered in the present study is the "prevalence" of the disease and in this case, it is represented by the number of positive and negative cases of bilharzia obtained before and after the interventions.
Independent variables: regarding the independent variables "countries" and "years" are defined to make the analysis of the subgroups.
Data processing and analysis: the forest plot is used to highlight the overall effect of the interventions on the prevalence of bilharzia as well as the estimated effect size and confidence interval for each study. For this purpose, the Odd Ratio (OR) is the indicator used to measure the effect size with a 95% confidence interval (CI). Subgroup analyses were also performed by different characteristics such as country and study year. Heterogeneity and publication bias were captured through the I2 statistic and the Egger test. From the forest plot, the heterogeneity test was performed. The STATA 16 software was used for the analysis.
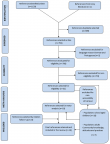
Selection of studies: the selection of studies is summarized in the flow chart (Figure 1). Entering the search equation into the various search engines mentioned above yielded 1120 results. After removing duplicates, 752 studies were obtained. After reading the titles and abstracts and applying the eligibility criteria, 31 studies were retained. Sixteen references (Figure 1) were then excluded because the full text was not available. Thus, in the end, 16 articles were included in the systematic review (Figure 1).
Source 1: compiled by the authors based on the processing of epidemiological data and interventions in the framework of bilharzia control in West Africa from 2010 to 2021.
Data synthesis: the works included in this review come from different countries in sub-Saharan Africa, such as: Benin, Ghana, Mali, Senegal, Burkina Faso, Sierra Leone and Niger. Four (04) studies were conducted in Mali, five (05) studies were conducted in Niger, two (02) studies were conducted in Benin and two (02) studies were conducted in Burkina Faso Table 1, Table 1 (suite), Table 1 (suite 1), Table 1 (suite 2) provides a summary of the articles reviewed.
Impact of anti-bilharzia interventions or strategies: data collected from the various articles show that bilharzia is one of the most important neglected tropical diseases (NTDs) in terms of morbidity and mortality. Epidemiological data from these papers show that the disease is endemic in many developing countries, affecting mostly children, farmers and women who are in frequent contact with waters that may harbour the intermediate host molluscs [16-30]. Millions of people worldwide are infected with different species of schistosomes [31].
The studies included in this systematic literature review were conducted in Benin, Ghana, Senegal, Mali, Burkina Faso, Sierra Leone and Niger. In all these studies, the interventions were related to the diagnosis of bilharzia in the urine and stool of the target population and treatment with praziquantel. In Benin, of the two studies included in this study, one was concerned with malacological research, i.e. mollusc intermediate hosts of the parasite before infecting humans [15]. The Kato Kartz method was the most widely used for diagnosis [26]. In Mali, a study also looked at the evolution of the disease in the population without any treatment [19]. Schistosoma haematobium and Schistosoma mansoni are the two most identified species in these studies [25].
It has been noted that any control measures for bilharzia should involve three major components: chemotherapy treatment, improvement of the health situation, vector control and health education. Addressing these components would help reduce transmission and reinfection by encouraging individuals to observe protective health behaviors [16,21]. Chemotherapy treatment was the most discussed component in this literature review. Three anti-bilharzia drugs are used for this purpose. Praziquantel is the main known antibilharzian. Data show that it is effective on all species of schistosome. Its cure rate varies from 80% to 100% [28]. Metrifonate is only effective on S. haematobium but resistance has been reported in Mali and Senegal [20,21,24]. Oxamniquine is active on S. mansoni. An experimental treatment with Epiquantel carried out in Niger has shown its efficacy, accompanied by undesirable effects, such as abdominal pain and bloody diarrhea in school children [28]. It is also important to emphasize that the strategies implemented for chemotherapy allow the targeting of interventions at the geographical level and at the level of risk groups. In general, all studies have shown that chemotherapy is the most effective means of controlling bilharzia because it has been shown to decrease the prevalence of bilharzia after the studies. These studies have also shown that praziquantel could be used by at-risk populations as a preventive treatment, accompanied by health education, especially that related to hygiene measures.
Meta analysis of bilharzia interventions: in order to optimize the results of the various bilharzia interventions over the study period, a meta-analysis of ten studies was conducted on all bilharzia interventions in West Africa.
Forest plot of the effect of the intervention on the studied phenomenon: Figure 2 presents the forest plot of the random effect model of the level of intervention on the studied phenomenon of bilharzia. There is a thematic correlation between all articles included in this study. All the works selected for this systematic literature review addressed the epidemiology, diagnostic methods and control measures (chemotherapy) of bilharzia in the study regions. There was no bias in the selected articles.
Source 2: compiled by the authors based on the processing of epidemiological data and interventions in the framework of bilharzia control in West Africa from 2010 to 2021 (Figure 2).
Risk of bias: Figure 3 presents the result of the Egger test for possible publication bias and shows that there is a homogeneity of studies with themes that respect the context of the systematic review (Figure 3). It has moreover demonstrated that the model is significant at the 5% level (p = 0.00) and the I2 statistic is 99.51% (Figure 3), which suggests strong heterogeneity. Similarly, the Egger test indicates the absence of publication bias (Prob > | Z | = 0.7405) (Figure 3). Thus, Figure 2 shows that there is no small sample size bias in this meta-analysis. Overall, the control interventions had a positive effect on the prevalence of bilharzia. Compared to the pre-intervention effects, an individual is 0.72 times less likely to contract the disease after intervention. However, some interventions had effects contrary to those normally expected. This trend was observed in Sierra Leone, Benin, Ghana, Senegal and Niger where a higher prevalence was observed after the intervention than before the intervention. Furthermore, the overall Odd-Ratio is 0.72 CI [0.19; 2.78] (Figure 3) which translates into a 0.72-fold lower risk of contracting bilharzia for an individual who has undergone the intervention compared to his or her counterpart living in an endemic area and not having undergone the intervention. The same trend was observed regardless of the country and year of study. The risk is higher respectively in Ghana in 2015, 15.57 CI [10.35; 23.41] (Figure 3) and Benin 2016-2020; 4.00 CI [3.82; 4.19] (Figure 3) while the lowest value is recorded in Benin in 2010 with 0.02 CI [0.01; 0.02] (Figure 3).
Source 3: compiled by the authors based on the processing of epidemiological data and interventions in the framework of bilharzia control in West Africa from 2010 to 2021 (Figure 3).
Subgroup analysis of the effect of interventions on the phenomenon studied: Figure 4 presents the subgroup analysis of the effect of the intervention, which here represents Praziquantel Mass Treatment (PMT), on reducing the number of people who contracted bilharzia in West Africa. Depending on the subgroup considered, we find both Odds-inferior to 1 for some values and superior to 1 for others and the p- value ratio is 0.000, which means that an individual who has undergone treatment is at less risk than one who has not received treatment. The overall trend observed is maintained within the subgroups and the extreme effect sizes are obtained in Burkina Faso (9.94; CI [8.88; 11.12]) (Figure 4) and Benin (0.26; CI [0.00; 54.73]) (Figure 4). Moreover, we also note a more or less significant progression of the odd with the lowest value observed in 2012 (0.14; CI [0.04; 0.51]) (Figure 4) and the value (4.00; CI [3.82; 4.19]) (Figure 4) obtained in 2020 with a peak of (9.94; CI [8.88; 11.12]) observed in the year 2013. This study therefore reveals that the prevalence of bilharzia has progressively increased from 2010 to 2020 which confirms that the risk of bilharzia contamination has remained constant throughout the last ten years with a very high level of endemicity. All West African countries considered in the study are affected by the disease and regardless of their geographical location, the odd ratio has increased significantly. When we look at the inter-group variations, we record fairly significant differences between countries, considering the effect of the interventions on the phenomenon studied on the one hand and the geographical location of the countries on the other (p = 0) (Figure 4). It follows that the effect of the interventions carried out on the phenomenon studied in West Africa did not remain constant over the study period considered (Figure 4).
Source 4: compiled by the authors based on the processing of epidemiological data and interventions in the framework of bilharzia control in West Africa from 2010 to 2021 (Figure 4)
Discussion
The aim of this systematic review was to synthesize existing evidence on the control strategies of West African countries against bilharzia. To ensure the quality of the methodology, the Prefered Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guideline criteria were followed at all stages of this systematic review. The main criterion was the situational analysis of the current situation through an assessment of control strategies and prospects for the eradication of bilharzia [32].
In general, health outcomes are unevenly distributed. In fact, several determinants have a considerable influence on the occurrence and recrudescence of bilharzia. These include behavioural hygiene, the availability of socio-community infrastructure (access to drinking water and sanitation facilities), environmental hygiene, exposure through working conditions by considering the case of populations forced to use waterways in the exercise of their fishing or farming profession and access to health services. As a result, this disease systematically hinders school attendance and performance.
The analysis of the data collected from the articles made it possible to identify several studies carried out in West African countries. These include Benin, Niger, Mali, Burkina Faso, Senegal, and Ghana [16,20,21,32]. These regions represent endemic areas for the occurrence of bilharzia. The prevalence study carried out shows a very high incidence of schistosomiasis by mass diagnostic surveys [13,15,17,18,20,25]. The African regions constitute indeed zones of predilection of the intermediate hosts of S. haematobium and S. mansoni [33]. The control strategies were essentially the identification of intermediate hosts and chemotherapy with PZQ. [22,25,30,31]. Currently mass drug administration campaigns remain the most studied and used strategy according to WHO guidelines [1,3,12].
The most studied target populations are school children under 12 years of age. The management of bilharziasis focuses on preventive and curative treatment [20]. Biannual treatment with praziquantel is one of the effective control strategies for bilharzia [34]. Anti-infective treatment has been shown to significantly reduce the parasite load of S. mansoni, but this is not always the case for S. haematobium in infested children under 6 years of age [21]. In West Africa, despite these measures, the control of schistosomiasis becomes complicated because Schistosoma spp. is capable of infecting several definitive hosts [20]. Vector control is therefore becoming a strategy of choice in the fight against schistosomiasis in the face of the many environmental changes that African countries are witnessing [19]. Drinking water supply is an important strategy in pest control, as bilharzia vectors have water as their natural habitat. Riverine populations without access to safe water are therefore at much greater risk of the disease [19,35]. As noted in several cohort studies, when shellfish control is combined with population-based screening and selective or mass drug therapy, prevalence would be reduced more rapidly and incidence would decrease in the population [21,36]. However, transmission was often not eliminated. Successful shellfish control programmes have significantly reduced the local prevalence of Schistosome infection [37]. But some control programmes have had minimal impact on local prevalence [33]. Although there were some apparent differences in effects by region and parasite species, the mollusc species identified in the included studies were too diverse to make meaningful comparisons for prevalence or incidence results stratified at the intermediate host species level [36].
According to the standard control rule, children aged 5-14 years have a 50% or greater chance of being infected after preventive chemotherapy performed once a year [22]. Thus, the basic control strategy for bilharzia is to choose the initial frequency of treatment based on the baseline prevalence and then re-evaluate after 5-6 years of the same drug. Depending on the prevalence of infection, the community is then given a possible change in the frequency of chemotherapy administration. The latter is re-evaluated after an additional 4 years of treatment to see if the objectives of the program have been achieved beacause the rate of infection has dropped considerably. Indeed, the objective of this control strategy is to achieve a morbidity of less than 5% of prevalence of heavy infection in children aged 5 to 14 years or the objective of eliminating less than 1% of prevalence of heavy infection [12,22]. A study carried out in 2021 in Mali on the massive distribution of drugs to school children reported a decrease in S. haematobium infection, however, the WHO requires that praziquantel not be used in preventive chemotherapy campaigns in school children because, to date, there is no suitable formulation. [1]. It would therefore be appropriate to re-evaluate the control of schistosomiasis in children with praziquantel.
In fact, the meta-analysis showed that overall, the interventions had a positive effect on bilharzia because of the way it spreads and its extent in the world, it is considered as a public health problem that undoubtedly impacts economic and socio-demographic conditions in a world development. Furthermore, it is an evidence that bilharzia is one of the neglected tropical diseases targeted by the WHO with endemic areas where the prevalence has reached alarming proportions [38,39]. The results indicate that the interventions carried out in the framework of the control of bilharzia with PZQ have an effect on the reduction of the prevalence in several countries whatever the country and the period of study. Indeed, the risk of bilharzia infection is reduced when the subject undergoes a preventive intervention (administration of praziquantel). However, the risk could be further reduced when the administration of PZQ is accompanied by multiple household sensitization on water use and relations as well as the promotion of socio-sanitary infrastructures.
In addition, according to WHO, current strategies for schistosomiasis control aim to prevent morbidity in later life through regular population monitoring by mass drug administration to at-risk populations in so-called homogeneous ecological zones [40,41]. This confirms the importance of the interventions implemented on both sides in the studies reviewed for the meta-analysis. However, according to studies conducted by Sturrock et al. it is clear that Mass Drug Treatment (MDT) alone is not the only solution to reduce the spread of schistosomiasis and therefore additional interventions are needed and should be implemented [40].
These results are consistent with previous studies by Danso-Appiah et al. [23] on the threshold prevalence observed during a MDT in May 2021 which showed that when Praziquantel is administered, the prevalence of S. mansoni and S. haematobium in school children decreases, but with no apparent difference between baseline and forty-eight months of treatment. The same is true for annual MDT administration for S. mansoni, which resulted in a reduction in children and adults, but no apparent reduction after twelve months of repeated MDT. At the community level, treatment has produced similar results with annual mass drug administration for nine years, but this has not reduced prevalence to the target of elimination of S. mansoni and S. haematobium in settings where baseline prevalence was 10% or more [31].
A malacological study carried out in the town of Péhunko in Benin in 2014 has shown results [15] much lower than those obtained by Ibikounlé et al. in 2009 [31,41] with a high prevalence of the condition (96%) in the village of Doh alone with a Z statistical test of the Stat View software (Z=2.555; p>0.05). It appears that this decrease in the infestation rate could be linked to the sensitization of the population and the mass treatments with praziquantel organized in the commune [42-44]. While the control strategy for schistosomes is preferably characterized by a mass survey followed by the administration of Praziquantel to communities, the use of molluscicides is also a control strategy recommended by the World Health Organization for the development of effective and practical measures for the control of schistosomiasis through snail elimination.
In this perspective, King et al. in a non-randomized study conducted in the United States in November 2015 under the control of the Center for Global Health and Diseases and the Schistosomiasis Consortium for Operational Research and Evaluation (SCORE) [35] had found that there is a positive correlation between the use of molluscicide in the control of snails and mass treatments with praziquantel. Thus, according to its authors, this synergy in the bilharzia control strategy is an effective method of reducing Schistosoma infections over time, with an additive effect on prevalence when population-based drug control is also carried out [45,46].
On the other hand, Knopp et al. in a study carried out in April 2013 in Zanzibar went further by highlighting that in the context of school-based treatment programmes, only the implementation of triple therapy consisting of snail control by molluscicides, preventive chemotherapy campaigns, and probably also the improvement of sanitary infrastructures, have reduced the prevalences of schistosomiasis from very high levels (50%) in the 1980s to a low level today [43-46]. It is therefore obvious that according to the authors, the reduction of the prevalence cannot only be the prerogative of a MDT with PZQ, nor the use of molluscicides against the hosts hosting miracidium, and that it is necessary to closely associate the teaching of hygiene notions by multiple sensitization sessions for a change of behaviour.
The results obtained from the present study deserve to be used taking into account some limitations common to schistosome control interventions. In general, however, it should be noted that the interventions identified are aimed to reduce the prevalence of schistosomiasis in endemic areas. Nevertheless, the resurgence and re-emergence of the disease despite the types of interventions commonly implemented seem to indicate the inadequacy of the strategies used in schistosomiasis control. It appears that raising awareness of good hygiene practices to encourage behavioural change and complementing traditional control strategies would be the most important means of accelerating the reduction of the prevalence rate. This study is innovative with its major assets being the elaboration of a synthetic view of the control strategies implemented in the fight against schistosomiasis, to bring out the positive effects and the impact of the treatments administered to the populations and to make a comparison of the interventions carried out in the different countries of West Africa.
Limitations: the major limitation is the very low number of studies of an experimental nature highlighting comparative data before and after interventions which does not favoring a broad-spectrum comparison. We therefore suggest that anti-schistosomiasis control strategies be more multispectral and carried out in a context of interventions including all the main actors such as religious, community leaders, traditional chiefs and in particular public authorities. Each intervention must be carried out in the context of evaluation "before and after intervention" so as to map the impacts in order to hope for better results tending to the elimination of schistosomiasis.
Annual antiparasitic distribution campaigns remain the most widely used control strategy, however, host-vector and human-to-human transmission related to poor hygiene result in a continuous rebound in infection prevalence. Repeated implementation of annual mass distribution campaigns based on WHO guidelines allows for an initial reduction in prevalence in two to three rounds. However, subsequent rounds do not allow for further reduction in prevalence of infection, which has prevented the elimination of the public health problem. Uncertainties regarding aspects of human and shellfish biology and exposure factors must be considered for effective control. It would therefore be important to encourage vector control in schistosomiasis control strategies.
What is known about this topic
- In the medical community the bilharziasis disease is sufficiently well known;
- Bilharziasis is well known as an NTD disease commonly in endemic area;
- The bilharziasis disease is known to be cured by the administration of PZQ.
What this study adds
- This study evaluates the strengths and weaknesses of the strategies developed to fight against bilharziasis;
- The study notes the inadequacies of the implemented control strategies;
- This study is initiated to highlight the implementing of an integrated control strategy against bilharziasis for better results.
The authors declare no competing interests.
All authors read and approved the final version of the manuscript and also contributed to its content.
We would like to aknowledge Dr Adogbo-Medagbe Estonie for assisting us all the time when we were working by feeding us every day. We equally acknowledge Mr Ahonon Nicaise, computer engineer who contributed towards the study by making substantial contributions, conception, design and his promptitude in computer problems. Same greatoussness towards Mr Acotcheou Pacôme who does not meet the criteria for authorship for acquisition of data, analysis and interpretation of data, who was involved in drafting the manuscript and revising it critically for important intellectual content. Finally, it is very important for us to acknowledge the reviewers and editors of PAMJ who were also involved in drafting the manuscript and have participated in giving the document its scientific character.
Table 1: summary and characteristics of studies included in the systematic review
Table 1 (suite): summary and characteristics of studies included in the systematic review
Table 1 (suite 1): summary and characteristics of studies included in the systematic review
Table 1 (suite 2): summary and characteristics of studies included in the systematic review
Figure 1: numbers of titles and studies reviewed in preparation of the current systematic review and meta-analysis bilharziasis and of chemical mollusciciding effects on Schistosoma-endemic area
Figure 2: forest plot of the effect of interventions (chemotherapy through mass drug administration and community awareness) on bilharziasis studied as the phenomenon
Figure 3: results of Egger's test for publication bias showing small study effects for the primary outcome (bias coefficient for the main analysis 95% confidence interval)
Figure 4: subgroup analysis of the effect of interventions on the phenomenon studied showing the effect of the intervention represented by Praziquantel Mass Treatment (PMT) on bilharzia
- World Health Organization (WHO). Schistosomiasis and soiltransmitted helminthiases: number of people treated in 2016. Accessed 25th August 2021.
- Hay SI, Abajobir AA, Abate KH, Abbafati C, Abbas KM, Abd-Allah F et al. Global, regional, and national disability-adjusted life-years (DALYs) for 333 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet. sept 2017;390(10100):1260-344. PubMed | Google Scholar
- Organisation Mondiale de la Santé. RAPPORT DE SITUATION 2001–2011 ET PLAN STRATÉGIQUE 2012–2020. Accessed 25th August 2021.
- Fenwick A, Rollinson D, Southgate V. Implementation of Human Schistosomiasis Control: Challenges and Prospects. Adv Parasitol. 2006;61:567-622. PubMed | Google Scholar
- Cao Z-G, Zhao Y-E, Lee Willingham A, Wang T-P. Towards the Elimination of Schistosomiasis japonica through Control of the Disease in Domestic Animals in The People's Republic of China: A Tale of over 60 Years. Adv Parasitol. 2016;92:269-306. PubMed | Google Scholar
- Zhang S-Q, Sun C-S, Wang M, Lin D-D, Zhou X-N, Wang T-P. Epidemiological Features and Effectiveness of Schistosomiasis Control Programme in Lake and Marshland Region in The People's Republic of China. Adv Parasitol. 2016;92:39-71. PubMed | Google Scholar
- Li H-J, Liang Y-S, Dai J-R, Wang W, Qu G-L, Li Y-Z et al. [Studies on resistance of Schistosoma to praziquantel XIV experimental comparison of susceptibility to praziquantel between PZQ-resistant isolates and PZQ- susceptible isolates of Schistosoma japonicum in stages of adult worms, miracidia and cercariae]. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 1 Dec 2011;23(6):611-9. PubMed | Google Scholar
- Couto FF, Coelho PMZ, Araújo N, Kusel JR, Katz N, Jannotti-Passos LK et al. Schistosoma mansoni: a method for inducing resistance to praziquantel using infected Biomphalaria glabrata snails. Mem Inst Oswaldo Cruz. March 2011;106(2):153-7. PubMed | Google Scholar
- Savioli L, Albonico M, Engels D, Montresor A. Progress in the prevention and control of schistosomiasis and soil-transmitted helminthiasis. Parasitology International. June 2004;53(2):103-13. PubMed | Google Scholar
- Mohamed-Ali Q, Doehring-Schwerdtfeger E, Abdel-Rahim IM, Schlake J, Kardorff R, Franke D et al. Ultrasonographical investigation of periportal fibrosis in children with Schistosoma mansoni infection: reversibility of morbidity seven months after treatment with praziquantel. Am J Trop Med Hyg. 1991 Apr;44(4):444-51. PubMed | Google Scholar
- Chen MG. Assessment of morbidity due to Schistosoma japonicum infection in China. Infect Dis Poverty. 2014 Feb 14;3(1):6. PubMed | Google Scholar
- World Health Organization. Working to overcome the global impact of neglected tropical diseases: first WHO report on neglected tropical diseases. Working to overcome the global impact of neglected tropical diseases: first WHO report on neglected tropical diseases. Google Scholar
- Bah YM, Paye J, Bah MS, Conteh A, Redwood-Sawyerr V, Sonnie M et al. Achievements and challenges of lymphatic filariasis elimination in Sierra Leone. Turner JD, editor. PLoS Negl Trop Dis. 2020 Dec 28;14(12):e0008877. PubMed | Google Scholar
- Moher D, Liberati A, Tetzlaff J, Altman DG, Altman D, Antes G et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement (Chinese edition). Journal of Integrative Medicine. 2009 Sep;7(9):889- 96. Google Scholar
- Ibikounlé M, Ogouyèmi-Hounto A, de Tové YSS, Dansou A, Courtin D, Kindé- Gazard D et al. Epidemiology of urinary schistosomiasis in schoolchildren in the commune of Péhunco in northern Benin: malacological survey. Bull Soc Pathol Exot. 2014;107(3):177-84. PubMed | Google Scholar
- Ouedraogo H, Drabo F, Zongo D, Bagayan M, Bamba I, Pima T et al. Schistosomiasis in school-age children in Burkina Faso after a decade of preventive chemotherapy. Bull World Health Organ. 1 Jan 2016;94(1):37-45. PubMed | Google Scholar
- Garba A, Kinde-Gazard D, Makoutodé M, Boyer N, Ernould JC, Chippaux JP et al. Preliminary evaluation of morbidity due to S. haematobium and S. mansoni in the area of the future Adjarala Dam in Benin. Santé. 2000 Sep-Oct;10(5):323-8. PubMed | Google Scholar
- Gyan B, Boakye D, Ayi I, van der Puije W, Asuming-Brempong E, Bimi L et al. Relationship between eosinophil cationic protein and infection intensity in a schistosomiasis endemic community in Ghana. RRTM. jan 2015;1. Google Scholar
- Dabo A, Diarra AZ, Machault V, Touré O, Niambélé DS, Kanté A et al. Urban schistosomiasis and associated determinant factors among school children in Bamako, Mali, West Africa. Infect Dis Poverty. dec 2015;4(1):4. PubMed | Google Scholar
- Catalano S, Leger E, Fall CB, Borlase A, Diop SD, Berger D et al. Multihost transmission of Schistosoma mansoni in Senegal, 2015-2018. Emerg Infect Dis. 2020 Jun;26(6):1234-1242. PubMed | Google Scholar
- Dabo A, Diallo M, Agniwo PK, Danté S, Traoré A, Diawara S et al. Mass Drug Distribution Strategy Efficacy for Schistosomiasis Control in Mali (West Africa). Researcg Square. 2021. Google Scholar
- Li EY, Gurarie D, Lo NC, Zhu X, King CH. Improving public health control of schistosomiasis with a modified WHO strategy: a model-based comparison study. The Lancet Global Health. oct 2019;7(10):e1414-. PubMed | Google Scholar
- Danso-Appiah A, Garba AD, Orso M, Akuffo KO, Fleming FM, Jiangang G et al. Prevalence threshold that should be applied when deciding schistosomiasis mass drug administration: systematic review and meta-analysis. Accessed 24th August 2021.
- Bintou LY, Yaro AS, Sodio B, Sacko M. Persistence of urinary schistosomiasis in endemic areas subjected to repeated mass treatment in Mali. Int J Bio Chem Sci. 23 May 2019;13(1):369. Google Scholar
- MINISTERE DE LA SANTE DU BENIN. PLAN DIRECTEUR NATIONAL DE LUTTE INTEGREE CONTRE LES MALADIES TROPICALES NEGLIGEES [Internet]. Benin: Direction Nationale de la Santé; 2017. Accessed 25th August 2021.
- Garba A, Touré S, Dembelé R, Boisier P, Tohon Z, Bosqué-Oliva E et al. Present and future schistosomiasis control activities with support from the Schistosomiasis Control Initiative in West Africa. Parasitology. Nov 2009;136(13):1731-7. PubMed | Google Scholar
- Garba A, Lamine MS, Barkiré N, Djibo A, Sofo B, Gouvras AN et al. Efficacy and safety of two closely spaced doses of praziquantel against Schistosoma haematobium and S. mansoni and re-infection patterns in school-aged children in Niger. Acta Trop. Nov 2013;128(2):334-44. PubMed | Google Scholar
- Garba A, Lamine MS, Djibo A, Tahirou A, Aouami MA, Alfari A et al. Safety and efficacy of praziquantel syrup (Epiquantel®) against Schistosoma haematobium and Schistosoma mansoni in preschool-aged children in Niger. Acta Trop. Nov 2013;128(2):318-25. PubMed | Google Scholar
- Bagayan M, Zongo D, Savadogo B, Ouedat A, Sorgho H, Kabre BG et al. Prevalence of urinary schistosomiasis in a school environment in a periurban area of Ouagadougou: Case of the Yamtenga district (Burkina Faso). Science and Technique, Health Sciences. 2014;37(1-2):93-100.
- Bah YM, Paye J, Bah MS, Conteh A, Saffa S, Tia A et al. Schistosomiasis in School Age Children in Sierra Leone After 6 Years of Mass Drug Administration With Praziquantel. Front Public Health. 2019 Feb 12;7:1. PubMed | Google Scholar
- Ibikounlé M, Mouahid G, Sakiti NG, Massougbodji A, Moné H. Freshwater snail diversity in Benin (West Africa) with a focus on human schistosomiasis. Acta Trop. 2009 Jul;111(1):29-34. PubMed | Google Scholar
- Badran RA. The interactive effect of some molluscicides on the inermediate host of human bilharziasis and their associated zoosporic fungi in the river Nile, Egypt. Bull Soc Pathol Exot. 1996;89(3):227-31. PubMed | Google Scholar
- Toure S. Two-year impact of single praziquantel treatment on infection in the national control programme on schistosomiasis in Burkina Faso. Bull World Health Organ. 2008 Oct;86(10):780-7. PubMed | Google Scholar
- Barbosa FS, Costa DPP. A long-term schistosomiasis control project with molluscicide in a rural area of Brazil. Ann Trop Med Parasitol. 1981 Feb;75(1):41-52. PubMed | Google Scholar
- King CH, Sutherland LJ, Bertsch D. Systematic Review and Meta-analysis of the Impact of Chemical-Based Mollusciciding for Control of Schistosoma mansoni and S. haematobium Transmission. Loker ES, editor. PLoS Negl Trop Dis. 28 Dec 2015;9(12):e0004290. PubMed | Google Scholar
- Gundersen SG, Birrie H, Torvik HP, Scherbaum H. Control of Schistosoma mansoni in the Blue Nile Valley of western Ethiopia by mass chemotherapy and focal snail control: a primary health care experience. Transactions of the Royal Society of Tropical Medicine and Hygiene. Nov 1990;84(6):819-25. PubMed | Google Scholar
- World Health Organization (WHO). Sustaining the drive to overcome the global impact of neglected tropical diseases: second WHO report on neglected diseases. Accessed 30th November 2021.
- World Health Organization (WHO). Helminth control in school-age children: a guide for managers of control programmes, 2nd ed. Accessed 30th November 2021.
- World Health Organization. Schistosomiasis: progress report 2001-2011. 2013. Google Scholar
- Sturrock RF. The control of schistosomiasis: epidemiological aspects of reinfection. Mem Inst Oswaldo Cruz. 1989;84(suppl 1):134-48. PubMed | Google Scholar
- Warren KS, Siongok TKA, Houser HB, Ouma JH, Peters PA. Quantification of Infection with Schistosoma haematobium in Relation to Epidemiology and Selective Population Chemotherapy. I. Minimal Number of Daily Egg Counts in Urine Necessary to Establish Intensity of Infection. J Infect Dis. 1 Dec 1978;138(6):849-55. PubMed | Google Scholar
- Prentice MA, Barnish G. Granule formulations of molluscicides for use in developing countries. Ann Trop Med Parasitol. Feb 1980;74(1):45-51. PubMed | Google Scholar
- Knopp S, Stothard JR, Rollinson D, Mohammed KA, Khamis IS, Marti H et al. From morbidity control to transmission control: time to change tactics against helminths on Unguja Island, Zanzibar. Acta Trop. nov 2013;128(2):412-22. PubMed | Google Scholar
- Mgeni AF, Kisumku UM, McCullough FS, Dixon H, Yoon SS, Mott KE. Metrifonate in the control of urinary schistosomiasis in Zanzibar. Bull World Health Organ. 1990;68(6):721-30. PubMed | Google Scholar
- Stothard JR, French MD, Khamis IS, Basáñez M-G, Rollinson D. The epidemiology and control of urinary schistosomiasis and soil-transmitted helminthiasis in schoolchildren on Unguja Island, Zanzibar. Trans R Soc Trop Med Hyg. Oct 2009;103(10):1031-44. PubMed | Google Scholar
- Mc Cullough FS, Krafft JG. Schistosomiasis in Zanzibar and Pemba. Geneva: World Health Organization; 1976.