Efficacy of resveratrol in women with polycystic ovary syndrome: a systematic review and meta-analysis of randomized clinical trials
Hammad Ali Fadlalmola, Amal Mohammed Elhusein, Khaled Mohammed Al-Sayaghi, Muayad Saud Albadrani, Duggahatti Veerabhadra Swamy, Daniel Mon Mamanao, Ehab Ibrahim El-Amin, Salma Elhadi Ibrahim, Siddiqa Mohammed Abbas
Corresponding author: Hammad Ali Fadlalmola, Nursing College, Taibah University, Madinah, Saudi Arabia 
Received: 12 Nov 2021 - Accepted: 09 Mar 2023 - Published: 16 Mar 2023
Domain: Family Medicine,Gynecology,Obstetrics and gynecology
Keywords: Meta-analysis, polycystic ovary syndrome, resveratrol, systematic review
©Hammad Ali Fadlalmola et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Hammad Ali Fadlalmola et al. Efficacy of resveratrol in women with polycystic ovary syndrome: a systematic review and meta-analysis of randomized clinical trials. Pan African Medical Journal. 2023;44:134. [doi: 10.11604/pamj.2023.44.134.32404]
Available online at: https://www.panafrican-med-journal.com//content/article/44/134/full
Review 
Efficacy of resveratrol in women with polycystic ovary syndrome: a systematic review and meta-analysis of randomized clinical trials
Efficacy of resveratrol in women with polycystic ovary syndrome: a systematic review and meta-analysis of randomized clinical trials
![]() Hammad Ali Fadlalmola1,&, Amal Mohammed Elhusein2,
Hammad Ali Fadlalmola1,&, Amal Mohammed Elhusein2, ![]() Khaled Mohammed Al-Sayaghi1, Muayad Saud Albadrani3, Duggahatti Veerabhadra Swamy2,
Khaled Mohammed Al-Sayaghi1, Muayad Saud Albadrani3, Duggahatti Veerabhadra Swamy2, ![]() Daniel Mon Mamanao1, Ehab Ibrahim El-Amin4, Salma Elhadi Ibrahim5, Siddiqa Mohammed Abbas5
Daniel Mon Mamanao1, Ehab Ibrahim El-Amin4, Salma Elhadi Ibrahim5, Siddiqa Mohammed Abbas5
&Corresponding author
Polycystic ovarian syndrome (PCOS) is a metabolic and hormonal condition affecting women of a reproductive age. It causes an abnormal menstrual cycle, anovulation, infertility, acne, hirsutism, obesity, hyperlipidemia, and cardiovascular disorders. Because resveratrol decreases testosterone levels, it may be of value in treating PCOS. We aimed to evaluate the efficacy of resveratrol in treating women with PCOS. We searched for randomized clinical trials (RCTs) in PubMed, Cochrane CENTRAL, Scopus and Web of Science. With 95% confidence intervals, the data was retrieved and analyzed as a mean difference (MD) or a standardized mean difference (SMD). Four RCTs with 218 women were included in the analysis. Resveratrol significantly reduced testosterone (SMD = -0.40; 95% CI [-0.71, -0.10], P = 0.009), luteinizing hormone (LH) (SMD = -0.32; 95% CI [-0.62, 0.01], P = 0.04), and dehydroepiandrosterone sulfate (DHEAS) (MD = -0.85; 95% CI [-1.25, -0.45], P < 0.0001) compared with the placebo. Resveratrol is effective in treating women with PCOS due to reducing the levels of testosterone, LH, and DHEAS. In combination with other treatments, especially for hyperlipidemia, resveratrol is beneficial for women diagnosed with PCOS.
Polycystic ovary syndrome (PCOS) is a complex hormonal and metabolic disorder affecting women of reproductive age. The major features of PCOS are abnormal menstrual cycles, anovulation, infertility, acne, hirsutism, obesity, hyperlipidemia, and cardiovascular disorders (as part of the metabolic syndrome) [1-3]. Although the prevalence is 5%-7%, the etiology of the condition is not yet understood [4]. Hyperandrogenism is considered a critical aspect of this syndrome. Hyperplasia of the theca-interstitial (T-I) compartment in the ovary and the associated overexpression of many enzymes involved in steroidogenesis can result in an increased production of ovarian androgens [5-7]. PCOS may also be caused by increased levels of both fasting insulin as well as the relative ratio of the gonadotropin luteinizing hormone (LH) and follicle-stimulating hormone (FSH) [8]. Recently, evidence supported the theory that chronic low-grade inflammation plays an etiological role in the pathogenesis of PCOS. Many PCOS studies reported a strong association between inflammation and hyperandrogenism [9,10].
Efficient management for patients with PCOS is still controversial. Current treatments involve antiandrogens for androgen-related indications, including acne and hirsutism, clomiphene to induce ovulation, combined oral contraceptive pills (OCP) for both menstrual dysfunction and hyperandrogenism, and metformin as an insulin-sensitizing agent that may reduce the secretion of ovarian androgen [8]. Antiandrogens, such as flutamide and spironolactone, are well tolerated and have significant effects on hirsutism, but they may cause loss of libido, breast tenderness, irregular menstrual cycle, and the risk of changing a male fetus to a female. In pregnancy, there are many other adverse effects related to each drug in this category [11,12]. Clomiphene is considered one of the first options for managing anovulation; however, about 30% of the patients do not respond. In addition, it has particular constraints in patients with advanced age and a body mass index (BMI) >30 kg/m2 [8,13]. OCP induces a negative feedback of LH secretion and ovarian androgen synthesis may decrease. They could inhibit the peripheral conversion of testosterone to dihydrotestosterone (the active form). However, OCPs have several potential side effects, including insulin resistance, glucose tolerance, venous thrombosis, and vascular reactivity [14].
Alternative treatments are urgently required for PCOS patients. Resveratrol (trans-3,5,4�-trihydroxystilbene) is a natural polyphenol supplement found in grapes, nuts, and berries with recognized anti-inflammatory, antioxidant, anti-cancer, and cardioprotective activities [15]. In the last few years, the promising effects of resveratrol treatment for PCOS have been investigated. In vitro studies [16] indicated that exposing rat theca-interstitial cells (T-I) to resveratrol led to reduced androgen production by increasing executioner caspases, decreasing the number of viable cells, and inhibiting deoxyribonucleic acid (DNA) synthesis [17]. Recently, resveratrol administration decreased the level of testosterone in rats with dehydroepiandrosterone (DHEA)-induced PCOS. Banaszewska et al. performed the first clinical trial assessing the effect of resveratrol on PCOS and found a significant decline in ovarian and adrenal androgens [18]. The effect of resveratrol on ovarian steroidogenesis is selective, because it has no impact on the creation of progesterone by the theca cells [19].
Despite the beneficial results obtained by some clinical trials, the available data is still inconclusive and inadequate. Consequently, we performed systematic review and meta-analysis with all available RCTs to investigate whether resveratrol can improve menstrual irregularity, and symptoms related to hyperandrogenism and reduce the levels of androgens and insulin.
Search strategy and data collection: we searched four databases: Cochrane CENTRAL, PubMed, Scopus, and Web of Science (WOS) for published clinical trials. The search included literature related to any current studies on the National Institutes of Health trials registry. The following search approach was used: (resveratrol or SRT501 or SRT-501 or resvida or stilbenetriol) and (“polycystic ovary syndrome” or “polycystic ovarian syndrome” or “Stein-Leventhal syndrome” or “sclerocystic ovary syndrome” or “sclerocystic ovarian degeneration” or “sclerocystic ovary” or “polycystic ovary disease” or “functional ovarian hyperandrogenism” or “ovarian hyperthecosis” or “Stein-Leventhal syndrome” or PCOS).
Selection criteria: we incorporated all RCTs for women with PCOS and evaluated the efficacy of the different doses of resveratrol to placebo. There was no age, gender, publishing date or location limits. Observational studies, animal studies, thesis reviews, non-English studies, non-accessible studies, and study abstracts were omitted.
Study outcomes: the efficacy of resveratrol in women with PCOS was measured through the following outcomes. Firstly, laboratory tests (hormones) including total testosterone (nmol/L), LH (mIU/mL), FSH (mIU/mL), TSH (mIU/mL), prolactin (ng/mL), and DHEAS (mol/L). Secondly, pregnancy occurrence rates including clinical and chemical pregnancy. Thirdly, lipid profiles including cholesterol (mg/dl), low-density lipoprotein-cholesterol (LDL-C) (mg/dl), high-density lipoprotein-cholesterol (HDL-C) (mg/dl), and triglycerides (mg/dL). Other parameters included an acne score, C-reactive protein (CRP), insulin (mIU/mL), and sex hormone binding globulin (SHBG) (nmol/L).
Quality assessment: the quality of the RCTs included were assessed by the Cochrane´s Risk of Bias Tool (Version 1, Chapter 8.5) [20]. According to the funnel-plot-based methods of Egger, due to the limited number of eligible studies, publication bias could not be evaluated [21].
Statistical analysis: the inverse-variance method was used to analyze the continuous outcomes as mean difference (MD) or the standardized mean difference (SMD) in cases of different units with a 95% confidence interval (CI). The Mantel-Haenszel method was used to analyze the dichotomous outcomes as relative risk (RR) with a 95% CI, and the fixed-effect model if the data were homogenous, if not, the random-effects model. We conducted the analysis with the Review Manager Software (version 5.3). In case of a missing standard deviation, we used Altman´s equation to calculate it from the 95% CI [22]. The equation is given as follows:
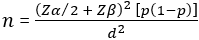
Where n = sample size; Zα/2 = critical value of the standard normal distribution for a given level of significance; Zβ = critical value of the standard normal distribution for a given power (1-p); p = expected proportion of individuals with the outcome of interest in the population; and d = desired level of precision or effect size.
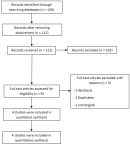
Literature search results: after removing the duplications, our search retrieved 111 unique citations; through title and abstract screening 102 were excluded and the 9 were eligible for full-text screening. Four RCT were eligible for inclusion [18,23-25]. Figure 1 displays the flow chart of the data collection and screening processes.
Summary of the eligible RCTs: the trials included compared different resveratrol doses (800, 1000, and 1500 mg daily) with the placebo. The total sample size was 218 women: 109 in each arm. All women were diagnosed with PCOS and received resveratrol or placebo orally, once or twice daily for at least 40 days. The women´s mean age ranged from 26.3 to 30.8 years. Table 1 and Table 2 display a summary of the trials as well as the baseline characteristics of the eligible participants.
Quality assessment: according to the Cochrane Risk of Bias tool, the eligible trials were high quality trials. All risk of bias were at low in terms of attrition, performance, reporting, and other sources of bias [18,23-25]. However, one trial [24] was categorized as an unclear risk of selection bias (randomization domain) and detection bias. Regarding selection bias (allocation domain), three trials [23-25] were low unclear and only one [18] was low risk. Figure 2 displays the risk of bias graph and summary.
Outcomes
A. Laboratory tests (hormones)
1. Total testosterone: three studies [23-25] with analyzable data reported this outcome with a total of 169 patients; 84 in the resveratrol arm and 85 in the placebo arm. The pooled effect estimate indicated that resveratrol significantly reduced the level of testosterone, compared with the placebo (SMD = -0.40; 95% CI [-0.71, -0.10], P = 0.009) (Figure 3). The pooled results were homogenous (I2 = 0%, P = 0.75).
2. Follicle-stimulating hormone (FSH) (mIU/mL): three studies [23-25] with analyzable data reported this outcome with a total of 169 patients; 84 in resveratrol arm and 85 in the placebo arm. The pooled effect estimate revealed no effect of resveratrol on FSH (SMD = -0.02; 95% CI [-0.78, 0.74], P = 0.96) (Figure 3). The pooled results were heterogenous (I2 = 83%, P = 0.003), which was resolved by excluding Bahramrezaie 2019 [25] (I2 = 0%, P = 0.93) and the results became significantly favoring resveratrol in term of reducing FSH (SMD = -0.41; 95% CI [-0.79, -0.02], P = 0.04).
3. Luteinizing hormone LH (mIU/mL): three studies [23-25] with analyzable data reported this outcome with a total of 169 patients; 84 in resveratrol arm and 85 in the placebo arm. Using the random effect model, the pooled effect estimate revealed that resveratrol significantly reduced testosterone compared to the placebo (SMD = -0.32; 95% CI [-0.65, 0.01], P = 0.06) (Figure 3). Using the fixed effect model, the pooled effect estimate indicated that resveratrol significantly lowered LH compared with the placebo (SMD = -0.32; 95% CI [-0.62, 0.01], P = 0.04). The pooled results were homogenous (I2 = 0%, P = 0.75). The pooled results were homogenous (I2 = 13%, P = 0.32).
4. Prolactin (ng/mL): three studies [23-25] with analyzable data reported this outcome with a total of 169 patients; 84 in the resveratrol arm and 85 in the placebo arm. The pooled effect estimate revealed no effect of resveratrol on prolactin (SMD = 0.04; 95% CI [-0.26, 0.35], P = 0.77) (Figure 3). The pooled results were homogenous (I2 = 5%, P = 0.35).
5. Thyroid stimulating hormone TSH (mIU/mL): two studies [24,25] with analyzable data reported this outcome with a total of 139 patients; 69 in resveratrol arm and 70 in the placebo arm. The pooled effect estimate revealed that no effect of resveratrol on TSH (MD = 0.25; 95% CI [-0.09, 0.58], P = 0.15) (Figure 3). The pooled results were heterogenous (I2 = 70%, P = 0.07), but the heterogeneity could not be resolved with only two trials included in the analysis of this outcome.
6. Dehydroepiandrosterone sulfate (DHEAS): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed that resveratrol significantly reduced DHEAS compared with placebo (MD = -0.85; 95% CI [-1.25, -0.45], P < 0.0001) (Figure 3). The pooled results were heterogenous (I2 = 65%, P = 0.09), but the heterogeneity could not be resolved with only two trials included in the analysis of this outcome.
B. Pregnancy occurrence
7. Positive clinical pregnancy: two studies [23,25] with analyzable data reported this outcome with a total of 101 patients; 50 in the resveratrol arm and 51 in the placebo arm. The pooled relative risk (RR) indicated no effect of resveratrol regarding clinical pregnancy rates (RR = 0.89; 95% CI [0.65, 1.23], P = 0.49) (Figure 4). The pooled results were homogenous (I2 = 31%, P = 0.23).
8. Positive chemical pregnancy: two studies [23,25] with analyzable data reported this outcome with a total of 100 patients; 50 in each arm. The pooled relative risk (RR) indicated that was no effect of resveratrol regarding clinical pregnancy rates (RR = 0.84; 95% CI [0.60, 1.17], P = 0.30) (Figure 4). The pooled results were homogenous (I2 = 51%, P = 0.15).
C. Lipid profiles
9. Cholesterol (mg/dl): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on cholesterol (MD = -5.91; 95% CI [-18.54, 6.72], P = 0.36) (Figure 5). The pooled results were homogenous (I2 = 48%, P = 0.17).
10. High-density lipoprotein- cholesterol (HDL-C) (mg/dl): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on HDL (MD = 0.24; 95% CI [-3.53, 4.00], P = 0.90) (Figure 5). The pooled results were homogenous (I2 = 0%, P = 0.73).
11. Low-density lipoprotein-cholesterol (LDL-C) (mg/dl): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on LDL (MD = -2.03; 95% CI [-10.24, 6.18], P = 0.63) (Figure 5). The pooled results were homogenous (I2 = 53%, P = 0.14).
12. Triglycerides (mg/dl): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on triglycerides (MD = -8.25; 95% CI [-37.25, 20.74], P = 0.58) (Figure 5). The pooled results were homogenous (I2 = 0%, P = 0.51).
D. Other parameters
13. Acne score: two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on the acne score (MD = -0.15; 95% CI [-0.39, 0.08], P = 0.21) (Figure 6). The pooled results were homogenous (I2 = 0%, P = 0.70).
14. C-reactive protein (CRP): three studies [18,23,24] with analyzable data reported this outcome with a total of 148 patients; 74 in each group. The pooled effect estimate revealed no effect of resveratrol on the CRP (MD = 0.00; 95% CI [-1.31, 1.31], P = 1.00) (Figure 6). The pooled results were heterogenous (I2 = 70%, P = 0.04). Brenjian et al. 2018 was excluded to resolve the heterogeneity [23] (I2 = 0%, P = 0.42) without affecting the significance (P = 0.18).
15. Insulin (mIU/ml): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on insulin (MD = -1.82; 95% CI [-5.60, 1.96], P = 0.35) (Figure 6). The pooled results were homogenous (I2 = 41%, P = 0.19).
16. Sex hormone binding globulin (SHBG) (nmol/L): two studies [18,24] with analyzable data reported this outcome with a total of 108 patients; 54 in each group. The pooled effect estimate revealed no effect of resveratrol on the SHBG (MD = -2.81; 95% CI [-16.12, 10.50], P = 0.68) (Figure 6). The pooled results were homogenous (I2 = 28%, P = 0.24).
Based on the analysis, resveratrol had a significant effect on decreasing testosterone, LH, and DHEAS compared with placebo, indicating a positive effect on managing the hormonal imbalance responsible for most of the disease symptoms. However, there was a non-significant difference between the resveratrol and placebo for the FSH, prolactin, and TSH level. Resveratrol had no effect on the clinical or chemical pregnancy rates, the lipid profiles assessed by cholesterol, hormone-sensitive lipase (HSL), LDL, and triglyceride levels, or other parameters such as the acne score, CRP, insulin and SHBG.
The current treatment strategies used for managing women with PCOS include flutamide, spironolactone, metformin, clomiphene citrate, and combined OCP [8]. In patients with PCOS, anovulation is caused by high local androgen concentrations directly affecting the ovary [26,27]. Flutamide is a nonsteroidal antiandrogen of known efficacy in patients with hirsutism. It is not known yet whether it functions by only blocking the androgen receptors or through inhibiting androgen biosynthesis [27]. As flutamide therapy may reestablish ovulatory cycles and fertility, contraception may be required as reported by De Leo et al. [27]. Combined OCPs are the most frequently used treatment modality for PCOS as they suppress androgen production, and improve skin androgenic symptoms and menstrual dysfunction [28]. Vrbíková et al. reported prospective data about the effect of combined OC indicating a reduction in the risk of coronary artery disease and endometrial cancers in PCOS patients [28]. Clomiphene citrate (CC) is also one of the first therapeutic options for treating anovulation in PCOS patients due to its low cost, limited dose-dependent side effects, and ease of administration. Should the woman be CC resistant, metformin alone or with clomiphene is a feasible alternative [29]? According to Johnson 2014 [29], metformin is an appropriate first line of treatment in non-obese PCOS women with anovulatory infertility.
Regarding the current outcomes of the different hormone levels in PCOS, some hormone levels measured during the PCOS diagnosis include LH, FSH, total and free testosterone, prolactin, DHEAS, TSH, progesterone, androstenedione, and estrogen. Regarding the testosterone hormone level, the levels of both the total and free testosterone is often increased. A slight increase in testosterone can suppress normal menstruation and ovulation. In PCOS, the testosterone values will be ≤150 ng/dL (≤5.2 nmol/L) [30]. Our analysis revealed a significant effect of resveratrol, compared with placebo, in reducing the level of testosterone. The DHEAS values may be normal or slightly higher in PCOS [30]. The current analysis indicated a superiority of resveratrol over the placebo in decreasing the DHEAS level.
In terms of the LH and FSH hormones, which supports ovulation, women with PCOS typically have a LH level of 18 mlU/ml and a FSH level of 6 mlU/ml, which is within the normal range of 5-20 mlU/ml, and indicated as an elevated LH to FSH ratio or a ratio of 3:1 [31]. Our analysis revealed that there was no significant difference between resveratrol and placebo in terms of FSH, with a marginal reducing effect on LH. Regarding the prolactin hormone, mild hyperprolactinemia occurs in 5% to 30% of PCOS patients [30,32]. Our results showed no superiority of resveratrol compared with placebo in decreasing the prolactin levels.
For the occurrence of pregnancy, clinical pregnancy decreases in PCOS due to hormonal disturbance. However, there was no difference between resveratrol and placebo. The lipid profile is frequently abnormal in PCOS patients [33], but the abnormalities are not consistent in all populations. According to Kiranmayee et al. [33] the majority (80%) of PCOS patients demonstrated abnormal lipid profiles, and in 70%, the abnormality included a low level of high-density lipoprotein (HDL) [33]. No superiority of resveratrol over placebo was observed in our analysis regarding a decrease in lipid profiles.
However, resveratrol produces significant results in some important outcomes. Bahramrezaie et al. [25] reported that resveratrol improves the PCOS symptoms, possibly through the expression of specific genes in the angiogenesis pathway of the granulosa cells or altering the serum levels of some sex hormones. Banaszewska et al. [18] concluded that the ovarian and adrenal androgens were significantly reduced by resveratrol, supporting an improvement in the condition. In addition, Mansour et al. [24] found that resveratrol improved the menstrual cyclicity and loss of hair. These findings could predict the potential effect of resveratrol, especially if combined with other lines of therapy to achieve the outcome, which cannot be obtained with resveratrol alone.
Regarding the strengths and limitations of our study, we conducted a comprehensive search to obtain a substantial level of evidence. The quality of the clinical trials we included complied with the currently accepted level of evidence. All the available outcomes in the final sample were reported. We performed a sensitivity analysis to detect the level of significance, and where heterogeneity occurred, it was resolved. However, some limitations should be considered. The publication bias could not be assessed due to the limited sample size. We included a small number of studies with a relatively small sample size. We recommend future clinical trials investigating the efficacy of resveratrol in combination with other lines of treatment, with a larger sample size and a longer follow-up duration.
Resveratrol is a promising and effective drug in the treatment of women with PCOS due to its effect on the testosterone, LH, and DHEAS levels. Combinations with other treatment, especially for hyperlipidemia, are required to improve the outcome in women with PCOS.
What is known about this topic
- Polycystic ovary syndrome (PCOS) is a complex hormonal and metabolic disorder affecting women of reproductive age;
- The major features of PCOS are abnormal menstrual cycles, anovulation, infertility, acne, hirsutism, obesity, hyperlipidemia, and cardiovascular disorders.
What this study adds
- Resveratrol is effective in treating women with PCOS due to reducing the levels of testosterone, LH, and DHEAS;
- In combination with other treatments, especially for hyperlipidemia, resveratrol is beneficial for women diagnosed with PCOS.
The authors declare no competing interests.
Hammad Ali Fadlalmola was the study leader, planned the design, and conducted the analysis; Hammad Ali Fadlalmola, Muayad Saud Albadrani screened the papers, extracted the data, and wrote the paper; Duggahatti Veerabhadra Swamy, Amal Mohammed Elhusein, Khaled Mohammed Al-Sayaghi, Salma Elhadi Ibrahim, Siddiqa Mohammed Abbas, Ehab Ibrahim El-Amin, and Daniel Mon Mamanao performed the following tasks independently; searching databases, screening records, and data extraction; Hammad Ali Fadlalmola, Muayad Saud Albadrani, Khaled Mohammed Al-Sayaghi, Salma Elhadi Ibrahim, Siddiqa Mohammed Abbas, Ehab Ibrahim El-Amin and Amal Mohammed Elhusein were responsible for the editing, peer reviewing, and supervision. All the authors read and approved the final version of this manuscript.
We thank all the librarians who helped us in prepare our search strategy in deferent databases.
Table 1: complete summary of the included studies and their findings
Table 2: baseline characteristics of enrolled patients in the included studies
Figure 1: PRISMA flow chart which summarizes the literature search, and number of the obtained records
Figure 2: summary of risk of bias for each included study
Figure 3: forest plot of laboratory tests (hormones) including total testosterone, FSH (mIU/mL), LH (mIU/mL), prolactin (ng/mL), TSH (mIU/mL), and DHEAS
Figure 4: forest plot of pregnancy occurrence including positive clinical pregnancy and positive chemical pregnancy
Figure 5: forest plot of lipid profiles including cholesterol (mg/dl), HDL-C (mg/dl), LDL-C (mg/dl), and triglycerides (mg/dl)
Figure 6: forest plot of other parameters including acne score, CRP, Insulin (mIU/mL), SHBG (nmol/L)
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum Reprod. 2004 Jan;19(1):41-7. PubMed | Google Scholar
- Norman RJ, Dewailly D, Legro RS, Hickey TE. Polycystic ovary syndrome. Lancet. 2007 Aug 25;370(9588):685-97. PubMed | Google Scholar
- Teede HJ, Misso ML, Costello MF, Dokras A, Laven J, Moran L et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum Reprod. 2018 Sep 1;33(9):1602-1618. PubMed | Google Scholar
- Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, Azziz R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016 Jul;106(1):6-15. PubMed | Google Scholar
- Gilling-Smith C, Story H, Rogers V, Franks S. Evidence for a primary abnormality of thecal cell steroidogenesis in the polycystic ovary syndrome. Clin Endocrinol (Oxf). 1997 Jul;47(1):93-9. PubMed | Google Scholar
- Nelson VL, Legro RS, Strauss JF 3rd, McAllister JM. Augmented androgen production is a stable steroidogenic phenotype of propagated theca cells from polycystic ovaries. Mol Endocrinol. 1999 Jun;13(6):946-57. PubMed | Google Scholar
- Wickenheisser JK, Quinn PG, Nelson VL, Legro RS, Strauss JF 3rd, McAllister JM. Differential activity of the cytochrome P450 17α-hydroxylase and steroidogenic acute regulatory protein gene promoters in normal and polycystic ovary syndrome theca cells. J Clin Endocrinol Metab. 2000 Jun;85(6):2304-11. PubMed | Google Scholar
- Arentz S, Abbott JA, Smith CA, Bensoussan A. Herbal medicine for the management of polycystic ovary syndrome (PCOS) and associated oligo/amenorrhoea and hyperandrogenism; a review of the laboratory evidence for effects with corroborative clinical findings. BMC Complement Altern Med. 2014 Dec 18;14:511. PubMed | Google Scholar
- González F. Inflammation in Polycystic Ovary Syndrome: Underpinning of insulin resistance and ovarian dysfunction. Steroids. 2012 Mar;77(4):300-5. PubMed | Google Scholar
- González F, Rote NS, Minium J, Kirwan JP. Evidence of proatherogenic inflammation in polycystic ovary syndrome. Metabolism. 2009 Jul;58(7):954-62. PubMed | Google Scholar
- Spritzer PM, Lisboa KO, Mattiello S, Lhullier F. Spironolactone as a single agent for long-term therapy of hirsute patients. Clin Endocrinol (Oxf). 2000 May;52(5):587-94. PubMed | Google Scholar
- Rosenfield RL. Clinical practice. Hirsutism. N Engl J Med. 2005 Dec 15;353(24):2578-88. PubMed
- Legro RS, Barnhart HX, Schlaff WD, Carr BR, Diamond MP, Carson SA et al. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N Engl J Med. 2007 Feb 8;356(6):551-66. PubMed | Google Scholar
- Badawy A, Elnashar A. Treatment options for polycystic ovary syndrome. Int J Womens Health. 2011 Feb 8;3:25-35. PubMed | Google Scholar
- Manna SK, Mukhopadhyay A, Aggarwal BB. Resveratrol suppresses TNF-induced activation of nuclear transcription factors NF-κB, activator protein-1, and apoptosis: potential role of reactive oxygen intermediates and lipid peroxidation. J Immunol. 2000 Jun 15;164(12):6509-19. PubMed | Google Scholar
- Wong DH, Villanueva JA, Cress AB, Duleba AJ. Effects of resveratrol on proliferation and apoptosis in rat ovarian theca-interstitial cells. Mol Hum Reprod. 2010 Apr;16(4):251-9. PubMed | Google Scholar
- Furat Rencber S, Kurnaz Ozbek S, Eraldemlr C, Sezer Z, Kum T, Ceylan S et al. Effect of resveratrol and metformin on ovarian reserve and ultrastructure in PCOS: an experimental study. J Ovarian Res. 2018 Jun 29;11(1):55. PubMed | Google Scholar
- Banaszewska B, Wrotynska-Barczynska J, Spaczynski RZ, Pawelczyk L, Duleba AJ. Effects of Resveratrol on Polycystic Ovary Syndrome: A Double-blind, Randomized, Placebo-controlled Trial. J Clin Endocrinol Metab. 2016 Nov;101(11):4322-4328. PubMed | Google Scholar
- Ortega I, Wong DH, Villanueva JA, Cress AB, Sokalska A, Stanley SD et al. Effects of resveratrol on growth and function of rat ovarian granulosa cells. Fertil Steril. 2012 Dec;98(6):1563-73. PubMed | Google Scholar
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ et al. Cochrane Handbook for Systematic Reviews of Interventions version 6.3. Cochrane, 2022. Updated February 2022. Accessed 17th February 2023.
- Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997 Feb;315(7109):629-34. PubMed | Google Scholar
- Altman DG, Bland JM. Standard deviations and standard errors. BMJ. 2005 Oct;331(7521):903. PubMed | Google Scholar
- Brenjian S, Moini A, Yamini N, Kashani L, Faridmojtahedi M, Bahramrezaie M et al. Resveratrol treatment in patients with polycystic ovary syndrome decreased pro-inflammatory and endoplasmic reticulum stress markers. Am J Reprod Immunol. 2020;83(1):e13186. PubMed | Google Scholar
- Mansour A, Samadi M, Sanginabadi M, Gerami H, Karimi S, Hosseini S et al. Effect of resveratrol on menstrual cyclicity, hyperandrogenism and metabolic profile in women with PCOS. Clin Nutr. 2021 Jun;40(6):4106-4112. PubMed | Google Scholar
- Bahramrezaie M, Amidi F, Aleyasin A, Saremi A, Aghahoseini M, Brenjian S et al. Effects of resveratrol on VEGF & HIF1 genes expression in granulosa cells in the angiogenesis pathway and laboratory parameters of polycystic ovary syndrome: a triple-blind randomized clinical trial. J Assist Reprod Genet. 2019 Aug;36(8):1701-1712. PubMed | Google Scholar
- Ehrmann DA, Barnes RB, Rosenfield RL. Polycystic ovary syndrome as a form of functional ovarian hyperandrogenism due to dysregulation of androgen secretion. Endocr Rev. 1995 Jun;16(3):322-53. PubMed | Google Scholar
- De Leo V, Lanzetta D, D´Antona D, La Marca A, Morgante G. Hormonal effects of flutamide in young women with polycystic ovary syndrome. J Clin Endocrinol Metab. 1998 Jan;83(1):99-102. PubMed | Google Scholar
- Vrbíková J, Cibula D. Combined oral contraceptives in the treatment of polycystic ovary syndrome. Hum Reprod Update. 2005 May-Jun;11(3):277-91. PubMed | Google Scholar
- Johnson NP. Metformin use in women with polycystic ovary syndrome. Ann Transl Med. 2014 Jun;2(6):56. PubMed | Google Scholar
- Sheehan MT. Polycystic ovarian syndrome: diagnosis and management. Clin Med Res. 2004 Feb;2(1):13-27. PubMed | Google Scholar
- Saadia Z. Follicle Stimulating Hormone (LH: FSH) Ratio in Polycystic Ovary Syndrome (PCOS) - Obese vs. Non- Obese Women. Med Arch. 2020 Aug;74(4):289-293. PubMed | Google Scholar
- Franks S. Polycystic ovary syndrome: a changing perspective. Clin Endocrinol (Oxf). 1989 Jul;31(1):87-120. PubMed | Google Scholar
- Kiranmayee D, Kavya K, Himabindu Y, Sriharibabu M, Madhuri GLJ, Venu S. Correlations Between Anthropometry and Lipid Profile in Women With PCOS. J Hum Reprod Sci. 2017 Jul-Sep;10(3):167-172. PubMed | Google Scholar