Prolonged SARS-CoV-2 infection during autologous hematopoietic stem cell transplantation from Hodgkin´s lymphoma: a case report
Ouadii Abakarim, Fatima Ezzahra Lahlimi, Oumaima Maghnouj, Illias Tazi
Corresponding author: Ouadii Abakarim, Department of Clinical Hematology and Bone Marrow Transplantation, University Hospital Center Mohammed VI, Faculty of Medicine and Pharmacy, Cadi Ayyad University, Marrakesh, Morocco 
Received: 10 Jul 2022 - Accepted: 11 Mar 2023 - Published: 16 Mar 2023
Domain: Infectious disease,Oncology
Keywords: SARS-CoV-2, autologous hematopoietic stem cell transplantation, Hodgkin´s lymphoma, chemotherapy, case report
©Ouadii Abakarim et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Ouadii Abakarim et al. Prolonged SARS-CoV-2 infection during autologous hematopoietic stem cell transplantation from Hodgkin´s lymphoma: a case report. Pan African Medical Journal. 2023;44:133. [doi: 10.11604/pamj.2023.44.133.36277]
Available online at: https://www.panafrican-med-journal.com//content/article/44/133/full
Case report 
Prolonged SARS-CoV-2 infection during autologous hematopoietic stem cell transplantation from Hodgkin´s lymphoma: a case report
Prolonged SARS-CoV-2 infection during autologous hematopoietic stem cell transplantation from Hodgkin's lymphoma: a case report
![]() Ouadii Abakarim1,&,
Ouadii Abakarim1,&, ![]() Fatima Ezzahra Lahlimi1, Oumaima Maghnouj1,
Fatima Ezzahra Lahlimi1, Oumaima Maghnouj1, ![]() Illias Tazi1
Illias Tazi1
&Corresponding author
Autologous hematopoietic stem cell transplantation (HSCT) for relapsed Hodgkin's lymphoma increases the risk of infection by using intensive chemotherapy. This risk is obviously ongoing given the increased virulence of severe COVID-19. We report a case of a young man with Hodgkin's lymphoma who received conditioning chemotherapy followed by autologous HSCT and tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) during the early phase of aplasia with persistence of COVID-19 beyond 30 days with favorable follow-up and clinical improvement on treatment. For this type of patient with hematologic malignancy, viral infection can be fatal and strict medical precautions with isolation rules must be maintained, especially for SARS-CoV-2.
Immunocompromised patients with hematologic malignancies have historically been more susceptible to viral respiratory disease [1,2]. Autologous hematopoietic stem cell transplantation (HSCT) for relapsed Hodgkin's lymphoma increases the risk of infection by using intensive chemotherapy [3]. This risk is ongoing given the increased virulence of severe SARS-CoV-2 [4]. The Center for International Blood and Marrow Transplant Research (CIBMTR) has concluded that coronavirus infection in patients receiving autologous hematopoietic stem cell transplantation has a poor prognosis [5]. Here we report a case of relapsed Hodgkin's lymphoma who developed COVID-19 viral pneumonitis in bone marrow aplasia induced by conditioning followed by autologous HSCT.
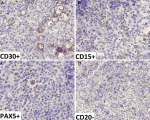
Patient information: a 26-year-old man with relapsed Hodgkin's lymphoma (Figure 1 (A, B, C, D)), initially stage VI by pleura, who received 6 cycles of ABVD (adriamycin, bleomycin sulfate, vinblastine sulfate, and dacarbazine), 3 cycles of ICE (ifosfamide, carboplatin, etoposide), 3 cycles of DHAP (dexamethasone (steroid), high-dose cytarabine (Ara-C), cisplatin (platinum)), and 2 cycles of GEMOX (gemcitabine-oxaliplatin), in complete remission, was admitted to our bone marrow transplant unit for autologous HSCT.
Clinical findings: on admission, the patient was in good general condition, the clinical examination was without particularity, and the paraclinical assessment. The nasopharyngeal swab, performed by reverse transcription-polymerase chain reaction (RT-PCR), was negative for SARS-CoV-2. Myeloablative conditioning was done by the BAM protocol (Busulfan, Cytarabine, Melphalan) without incident. Hematopoietic stem cell transplantation was performed on day 0 with a total of 2.89 x 106 CD34+/kg bw cells infused. On day 7, the patient presented with a fever and dry cough. Oxygen saturation was normal. A physical examination, including the chest, was normal.
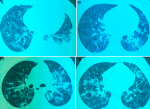
Diagnostic assessment: viral serologies were performed, and RT-PCR for SARS-CoV-2 was positive. On chest CT, the nodular ground-glass focus visible in the right dorsobasal segment was consistent with minimal COVID-19 viral pneumonitis and superimposed bacterial infection. The extent of parenchymal involvement was estimated to be between 10% and 25% (Figure 2 (A, B)). In the biological workup, C-reactive protein was elevated to 138.22 mg/L and ferritin to 4466 ng/ml. Procalcitonin levels were low at 0.52 ng/ml and D-dimer at 0.75 ug/ml.
Therapeutic interventions: due to the development of febrile neutropenia, the patient was initially put on: ceftriaxone 2 g/day for 3 days (day 7 to day 9) with fluconazole 400 mg on day 7, then 200mg/day for 4 days (day 8 to day 11) against oral thrush. Then, we put ceftazidime 1g x 4/day for 8 days (day 10 to day 17); ciprofloxacin 400mg x 2/day for 4 days (day 11 to day 14) in the event of persistent fever and signs of superinfection. Faced with the worsening of the infectious signs, the patient was put on amikacin 1g/day (day 12 to day 16) and voriconazole 400mg first 24h then 200mg/day for 5 days (day 12 to day 17). According to the National Guidelines on Management of Coronavirus Disease COVID-19 in Morocco, the patient was placed on azithromycin 500mg 1st day and 250mg/day for 7 days (day 8 to day 14); levofloxacin 500mg x 2/day (day 14 to day 27). Corticosteroid therapy was set up with methyl-prednisolone 40mg x 2/day for 8 days (day 14 to day 21), and then 20mg x 2/day for 3 days (day 23 to day 25). The duration of the aplasia was 22 days. On day 30, the patient's condition became more complicated. He developed stage III NYHA dyspnea with dysphagia. The physical examination showed a fever of 38.5°C, polypnoea at 28 cycles/min, whitish deposits on the tongue and throat, and bilateral basithoracic crackles on auscultation. Chest CT showed a scannographic appearance consistent with COVID-19 viral pneumonitis, with extensive parenchymal involvement estimated to be between 25% and 50% (Figure 2 (C, D)). Bacterial and mycotic blood cultures were all negative. The multiplex PCR was positive for SARS-CoV-2. According to the COVID-19 Guidelines in Morocco, the patient was put on hydroxychloroquine 100mg x 2/day for 7 days, azithromycin 500mg 1st day, then 250mg/day for 7 days. Heparin prophylaxis was administered during this period.
Follow-up and outcome of interventions: the evolution was marked by a favorable response with clinical improvement.
Patient perspective: during treatment, the patient was satisfied with the level of care provided to him.
Informed consent: the purpose of the study was explained to the patient, and informed consent was received before samples were collected. The patient was made aware that her medical records would be kept confidential.
The report presented here presents a patient who recovered from COVID-19 pneumonia with prolonged viral shedding during the neutropenic period of an autologous HSCT. Few studies have been published on the clinical outcomes of HSCT recipients with COVID-19 [5-7]. Patients receiving autologous HSCT have a higher risk of severe outcomes from COVID-19 in comparison with the general population. Despite this, the risk of contracting SARS-CoV-2 appears similar to the normal population [8]. Sharma et al. [5] studied 318 patients who underwent autologous HSCT diagnosed with COVID-19. They found that nearly half of the patients had the mild disease (49%), whereas 14% of patients had severe disease and required intensive care. At 30 days of follow-up, overall survival (OS) was 68% in allogeneic transplant recipients and 67% in autologous transplant recipients. Older age (>50 years), male gender, and patients with lymphoma had higher mortality in autologous HSCT than myeloma. There was no significant increase in mortality secondary to COVID-19 infection in patients who underwent myeloablative chemotherapy during autologous HSCT [6]. Patients who are candidates for autologous HSCT must be hospitalized for a long time. This makes them more susceptible to nosocomial infections [9]. This was the case with our patient. Thus, frequent testing of healthcare providers, including nurses and other members of the transplant team, for asymptomatic carriers of COVID-19 would be wise. The treatment of COVID-19 is controversial, and the guidelines differ. No clear recommendations exist for HSCT recipients, particularly in the early transplant setting, mainly due to the paucity of data and an unknown risk-benefit ratio [5].
Based on the recommendations of the scientific and technical committee of the national program for the prevention and control of influenza and other acute respiratory infections, the Moroccan Ministry of Health adopted a therapeutic protocol based on chloroquine and hydroxychloroquine. Approaches such as chloroquine (500 mg every 12 hours) or hydroxychloroquine sulfate (200 mg every 8 hours) for a period of 10 days, along with azithromycin 500 mg on day 1, followed by 250 mg daily from day 2 to day 7, have been suggested. In the default of clinical and/or PCR improvement, treatment will be discontinued. Lopinavir/Ritonavir at a dosage of 400mg x 2 daily for 10 days was also used as a second-line treatment. Antibiotics were not routinely used and were recommended only in case of bacterial superinfection. For patients at a high risk of thromboembolic complications, it may be important to combine low molecular weight heparin (LMWH). Patients are managed as inpatients, with strict clinical, biological, and radiological monitoring to detect any signs of deterioration at an early stage [10]. Several trials have well demonstrated the feasibility and efficacy of early management with combination therapy of hydroxychloroquine and azithromycin to prevent COVID-19-related deaths [11]. Cohorts have shown, as in the largest published outpatient series, that hydroxychloroquine-based therapy is associated with the lowest mortality rates and was not associated with severe cardiac side effects but was associated with a significant decrease in an infection fatality rate of 75% [12,13].
In contrast, those countries using hydroxychloroquine plus azithromycin or Lopinavir/Ritonavir as therapy at the onset of the epidemic have experienced a much slower dynamic in daily deaths [14,15]. In the reverse direction, numerous meta-analyses have shown the formal ineffectiveness of hydroxychloroquine alone or along with azithromycin with an increased risk of mortality [16]. The World Health Organization (WHO) does not recommend the use of hydroxychloroquine alone or along with azithromycin for COVID-19 [17]. The use of corticosteroids reduces inflammation but delays the immune reconstitution necessary for viral clearance. Corticosteroids are the most widely agreed-upon treatment in a cytokine storm [18]. A cautious compromise between immunosuppression for managed immune reconstitution and the limitation of inflammation that can damage the lungs is crucial [19]. The use of corticosteroids slightly reduces the number of deaths from all causes up to 60 days after treatment [20]. It also improves people's symptoms in patients with SARS-CoV-2 infection [18]. Our patient survived well with severe and prolonged COVID-19 under this protocol despite the presence of Hodgkin's lymphoma and post-conditioning aplasia, and controversies related to the chosen treatment.
We see above the case of a patient with severe and prolonged COVID-19 after conditioning and autologous HSCT. For these types of patients with hematologic malignancies, viral infection can be severe and strict medical precautions and isolation rules should be maintained especially for SARS-CoV-2.
The authors declare no competing interests.
Patient management: Ouadii Abakarim, Fatima Ezzahra Lahlimi, Oumaima Maghnouj and Illias Tazi. Data collection: Ouadii Abakarim, Fatima Ezzahra Lahlimi, and Oumaima Maghnouj. Manuscript drafting: Ouadii Abakarim. Manuscript revision: Fatima Ezzahra Lahlimi and Illias Tazi. All authors approved the final version of the manuscript.
We thank all clinical staff who participated in the treatment of the patient.
Figure 1: (A, B, C, D) immunohistochemistry of lymph node biopsy showing relapsed Hodgkin lymphoma in patient
Figure 2: A) first chest CT scan showing minimal COVID-19 viral pneumonitis; B) second chest CT scan showing extensive COVID-19 viral pneumonitis
- Martino R, Rámila E, Rabella N, Muñoz JM, Peyret M, Portos JM et al. Respiratory virus infections in adults with hematologic malignancies: a prospective study. Clin Infect Dis. 2003 Jan 1;36(1): 1-8. PubMed | Google Scholar
- Hakki M, Rattray RM, Press RD. The clinical impact of coronavirus infection in patients with hematologic malignancies and hematopoietic stem cell transplant recipients. J Clin Virol. 2015 Jul;68:1-5. PubMed | Google Scholar
- Linch DC, Winfield D, Goldstone AH, Moir D, Hancock B, McMillan A et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin´s disease results of a BNLI randomised trial. Lancet 1993;341:1051-1054. PubMed | Google Scholar
- Wang XA, Binder AF, Gergis U, Wilde L. COVID-19 in Patients With Hematologic Malignancies: A Single Center Retrospective Study. Front Oncol. 2021 Oct 28;11:740320. PubMed | Google Scholar
- Sharma A, Bhatt NS, Martin AS, Abid MB, Bloomquist J, Chemaly RF et al. Clinical characteristics and outcomes of COVID-19 in haematopoietic stem-cell transplantation recipients: An observational cohort study. Lancet Haematol. 2021 Mar;8(3):e185-e193. PubMed | Google Scholar
- Sultan AM, Mahmoud HK, Fathy GM, Abdelfattah NM. The outcome of hematopoietic stem cell transplantation patients with COVID-19 infection. Bone Marrow Transplant. 2021 Apr;56(4):971-973. PubMed | Google Scholar
- Ljungman P, Mikulska M, de la Camara R, Basak GW, Chabannon C, Corbacioglu S et al. The challenge of COVID-19 and hematopoietic cell transplantation; EBMT recommendations for management of hematopoietic cell transplant recipients, their donors, and patients undergoing CAR T-cell therapy. Bone Marrow Transplant. 2020 Nov;55(11):2071-2076. PubMed | Google Scholar
- Silfverberg T, Wahlin B, Carlson K, Cherif H. Impact of COVID-19 on patients treated with autologous hematopoietic stem cell transplantation: A retrospective cohort study. Ups J Med Sci. 2022 Aug 25;127. PubMed | Google Scholar
- Kanellopoulos A, Ahmed MZ, Kishore B, Lovell R, Horgan C, Paneesha S et al. COVID-19 in bone marrow transplant recipients: reflecting on a single centre experience. Br J Haematol. 2020 Jul;190(2):e67-e70. PubMed | Google Scholar
- Naciri A, Sine H, Baba MA, Bouchriti Y, Kharbach A, Achbani A. National Guidelines on Management of Coronavirus Disease COVID-19 in Morocco. European Journal of Medical and Educational Technologies 2020;13(1):em20.
- Million M, Lagier JC, Tissot-Dupont H, Ravaux I, Dhiver C, Tomei C et al. Early combination therapy with hydroxychloroquine and azithromycin reduces mortality in 10,429 COVID-19 outpatients. Rev. Cardiovasc. Med. 2021;22(3):1063-1072. PubMed | Google Scholar
- McCullough PA, Kelly RJ, Ruocco G, Lerma E, Tumlin J, Wheelan KR et al. Pathophysiological Basis and Rationale for Early Outpatient Treatment of SARS-CoV-2 (COVID-19) Infection. Am J Med. 2021 Jan;134(1):16-22. PubMed | Google Scholar
- Mokhtari M, Mohraz M, Gouya MM, Namdari Tabar H, Tabrizi J, Tayeri K et al. Clinical outcomes of patients with mild COVID-19 following treatment with hydroxychloroquine in an outpatient setting. Int Immunopharmacol. 2021 Jul;96:107636. PubMed | Google Scholar
- Arshad S, Kilgore P, Chaudhry ZS, Jacobsen G, Wang DD, Huitsing K et al. Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19. Int J Infect Dis. 2020 Aug;97:396-403. PubMed | Google Scholar
- Mikami T, Miyashita H, Yamada T, Harrington M, Steinberg D, Dunn A et al. Risk factors for mortality in patients with COVID-19 in New York City. J Gen Intern Med. 2021 Jan;36(1):17-26. PubMed | Google Scholar
- Sivapalan P, Ulrik CS, Lapperre TS, Bojesen RD, Eklöf J, Browatzki A et al. Azithromycin and hydroxychloroquine in hospitalised patients with confirmed COVID-19: a randomised double-blinded placebo-controlled trial. Eur Respir J. 2022 Jan 6;59(1):2100752. PubMed | Google Scholar
- World Health Organization. WHO COVID-19 Solidarity Therapeutics Trial. Accessed February 21, 2023.
- COVID-19 Treatment Guidelines: Panel Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Accessed February 21, 2023.
- Wei J, Zhao J, Han M, Meng F, Zhou J. SARS-CoV-2 infection in immunocompromised patients: humoral versus cell-mediated immunity. J Immunother Cancer. 2020 Jul;8(2):e000862. PubMed | Google Scholar
- Cochrane Haematology Group, Wagner C, Griesel M, Mikolajewska A, Mueller A, Nothacker M et al. Systemic corticosteroids for the treatment of COVID-19. Cochrane Database Syst Rev. 2021 Aug 16;8(8):CD014963. PubMed | Google Scholar