Seroprevalence of hepatitis C virus antigen in patients with chronic liver disease and hepatocellular carcinoma at 12th week of treatment: a cross-sectional study
Donatien Serge Mbaga, Jacky Njiki Bikoï, Etienne Atenguena Okobalemba, Justin Olivier Essindi, Chris André Mbongue Mikangue, Alexandra Emmanuelle Membangbi, Aïcha Ngoutane, Arnaud Franck Elang, Sabine Aimée Touangnou-Chamda, Carole Stéphanie Sake, George Ikomey Mondinde, Sebastien Kenmoe, Sara Honorine Riwom Essama
Corresponding author: Sara Honorine Riwom Essama, Department of Microbiology, The University of Yaoundé I, Yaoundé, Cameroon 
Received: 14 May 2022 - Accepted: 24 Sep 2022 - Published: 11 Oct 2022
Domain: Epidemiology,Infectious diseases epidemiology
Keywords: Hepatitis C virus Ag, chronic hepatitis, cirrhosis, hepatocellular carcinoma
©Donatien Serge Mbaga et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Donatien Serge Mbaga et al. Seroprevalence of hepatitis C virus antigen in patients with chronic liver disease and hepatocellular carcinoma at 12th week of treatment: a cross-sectional study. Pan African Medical Journal. 2022;43:72. [doi: 10.11604/pamj.2022.43.72.35450]
Available online at: https://www.panafrican-med-journal.com//content/article/43/72/full
Research 
Seroprevalence of hepatitis C virus antigen in patients with chronic liver disease and hepatocellular carcinoma at 12th week of treatment: a cross-sectional study
Seroprevalence of hepatitis C virus antigen in patients with chronic liver disease and hepatocellular carcinoma at 12th week of treatment: a cross-sectional study
![]() Donatien Serge Mbaga1,
Donatien Serge Mbaga1, ![]() Jacky Njiki Bikoï1,
Jacky Njiki Bikoï1, ![]() Etienne Atenguena Okobalemba2, Justin Olivier Essindi1,
Etienne Atenguena Okobalemba2, Justin Olivier Essindi1, ![]() Chris André Mbongue Mikangue1,
Chris André Mbongue Mikangue1, ![]() Alexandra Emmanuelle Membangbi1,
Alexandra Emmanuelle Membangbi1, ![]() Aïcha Ngoutane1,3,
Aïcha Ngoutane1,3, ![]() Arnaud Franck Elang1,
Arnaud Franck Elang1, ![]() Sabine Aimée Touangnou-Chamda1,
Sabine Aimée Touangnou-Chamda1, ![]() Carole Stéphanie Sake1, George Ikomey Mondinde4,
Carole Stéphanie Sake1, George Ikomey Mondinde4, ![]() Sebastien Kenmoe5,
Sebastien Kenmoe5, ![]() Sara Honorine Riwom Essama1,&
Sara Honorine Riwom Essama1,&
&Corresponding author
Introduction: epidemiological data suggests that more than 50% of hepatitis C virus (HCV) patients fail treatment. The objective of the study was to highlight the seroprevalence of hepatitis C virus antigen (HCV Ag) at the 12th week of treatment.
Methods: during a cross-sectional study, participants with chronic liver disease and hepatocellular carcinoma (HCC) were recruited between December 2020 and March 2022 at the Yaoundé General Hospital (HGY) and the University Teaching Hospital of Yaounde (UTHY). Five millilitres of blood samples were taken from each consenting participant and then a qualitative search for HCV Ag by Enzyme-Linked Immuno Assay (ELISA) was performed. Analysis of the results was performed using SPSS Version 25.0 software.
Results: out of the 192 participants selected for the study, only 92 (47,9%) participants were at 12 weeks of treatment, including 69 (75%) participants positive for the hepatitis C virus antibody (HCV Ab) by RDT. Of these participants, 44 (47.8%) participants were positive for HCV Ag by ELISA, respectively 19/37 (51.3%), 14/19 (73.6%), 11/13 (84.6%) with chronic hepatitis (HC), Cirrhosis, and HCC (P<0.0001).
Conclusion: our results showed a high prevalence of HCV Ag in patients at their 12th week of treatment which predicts treatment failure and calls for public policy to develop new management strategies to prevent HCV treatment failure in our context.
Hepatitis C virus (HCV) is one of the main viruses responsible for liver disease [1]. An estimated 71.1 million people are living with HCV infection worldwide [2]. Chronic hepatitis C infection is the leading cause of death in people with chronic hepatitis (CH), cirrhosis, and hepatocellular carcinoma (HCC) [3], and this chronic infection causes 25% of HCC cases [4]. WHO Africa estimates that HCV is one of the viruses responsible for 96% of mortality in people with cirrhosis and those with HCC [5]. In Cameroon, HCV affects 6.5% of people [6] and shifts considerably across the population in the country according to the most recent Cameroonian studies [7-13]. The primary objective of HCV treatment with direct-acting antivirals (DAAs) is to cure infected people. This healing can be measured by the sustained virological response (RSV), which is defined as an absence of detectable HCV ribonucleic acid (RNA) in the serum at the 12th or 24th week of treatment [14]. Current DAA-based treatments have shown promising results, with a cure rate of more than 95% of patients from the 8th week of treatment [15]. This prompted the World Health Organization (WHO) to establish a global strategy for the elimination of viral hepatitis which aims to reduce the number of new infections, and deaths and increase by 80% the proportion of people treated by 2030 [3]. Unfortunately, some studies have shown that more than 50% of HCV patients fail treatment and systematically relapse and later develop treatment resistance [16,17]. This is mainly observed in certain hard-to-cure populations, such as patients with severe hepatic decompensation, active HCC, chronic HCV genotype 3 infection, and those with multiple DAA failures [18].
To date, multiple obstacles that may hinder the success of HCV treatment have been identified [19] and constitute the major limiting factors that may contribute to a significant increase in HCV morbidity and mortality over the next 15 years in developing countries if nothing is done [20]. Screening and diagnosis of HCV infection rely on three different approaches which can be used alone or in combination. These are the detection of an HCV IgG antibody (HCV-Ab), the detection of the capsid antigen of the HCV (HCV-Ag) which is highly retained from one viral strain to another, and the detection of HCV RNA. Of these approaches only HCV Ag and RNA can confirm an active infection [21] even the recent WHO guidelines continue to recommend the use of the Ac-HCV test for first-line screening [22]. Hepatitis C virus Ag in serum or plasma is an indirect marker of HCV replication that can be used as an alternative to HCV RNA to diagnose HCV viremia at the 12th or 24th week of treatment to assess whether the patient eliminates HCV in a context where HCV viral load assessment is very expensive for poor people [23]. We therefore set the objective to highlight the seroprevalence of HCV Ag in patients with chronic liver disease and hepatocellular carcinoma at the 12th week of treatment.
Study design and setting: this study was cross-sectional. It took place in the city of Yaoundé, and the hepato-gastroenterology departments of the Yaoundé General Hospital (YGH) and internal medicine of the University Teaching Hospital of Yaoundé (UTHY) served as a recruitment site for participants between December 2020 and March 2022.
Study population and sampling: all participants were informed of the aim of the study before their inclusion in the study. The study population was composed of participants with chronic liver disease and those with hepatocellular carcinoma (HCC) who came for follow-up at the 12th week of treatment either the UTHY or the HGY, the consenting participants were divided into 3 groups. Group 1 consisted of participants with chronic hepatitis C (CHC) who were negative for cirrhosis or HCC. Group 2 consisted of participants with cirrhosis diagnosed by radiographic, biochemical, and clinical examinations and confirmed by a physician. Group 3 was composed of participants with HCC diagnosed by histological, radiographic, and biochemical examinations and confirmed by a physician. These participants were chosen, regardless of their sex, age (21 years or older), nationality, and treatment. Any participant who was having others aetiologies or comorbidities was excluded from the study.
Procedure for collecting socio-demographic and clinical data: data collection from each participant was done consecutively and prospectively. Each of the participants spoke in the consultation box with the principal investigator. The information leaflet was presented, to those unable to read, the principal investigator read the contents of the leaflet and then explained the aim of the study. The consenting patient gave his signed agreement to participate in the study. Then a pre-established and standardized questionnaire was administered to him.
Variables collected: for each participant, qualitative and quantitative variables were collected either by interview or blood samples analysis. The qualitative variables collected were: sex, marital status, scarification, tattoos, piercing, alcohol consumption before disease; tobacco consumption before disease; surgical history; history of blood transfusion; the treatment submitted, patients with chronic hepatitis; cirrhosis; HCC; HCV-Ab; HCV-Ag. The quantitative variables collected were age; alanine aminotransferase (ALT) glutamic-pyruvic transaminase (GPT); aspartate aminotransferase (AST) glutamic-oxaloacetic transaminase (GOT); alpha-fetoprotein (AFP).
Collection and fate of blood samples: overall, five millilitres of blood samples were taken from the crook of the elbow of each consenting participant, in ethylenediamine tetraacetic acid (EDTA) tubes. The samples were transferred to the Centre for the Study and Control of Communicable Diseases (CSCCD) of the Faculty of Medicine and Biomedical Sciences of Yaoundé (FMBS) where they were divided into three aliquots of the same volume each. The first aliquot was used for the search for HCV Ab and HCV Ag, the second aliquot was used for the quantification of ALT, AST, and AFP. The third aliquot was divided into 2 parts and then saved for further analysis.
Biological analysis of samples
Search for hepatitis C virus antibody and hepatitis C virus antigen: we carried out a qualitative search for HCV-Ab by immunochromatography according to the designers of rapid diagnostic tests (RDT) VOXTUR BIO LTD for HCV manufactured in India. Only positive results were confirmed by Monolisa™ HCV Ag-Ab ultra for the detection of capsid antigen of HCV in human plasma manufactured in the USA. Samples whose optical density was greater than or equal to the threshold (ratio ≥1) were considered positive. We did not retest samples due to a lack of reagent.
Alpha-fetoprotein, alanine aminotransferase, and aspartate aminotransferase quantification: quantification of AFP and transaminases (ALT and AST) was carried out only on ELISA-positive samples. Indeed, 30 HCV were selected randomly using a random number generator in Microsoft Excel. The codes corresponding to the aliquots were used for analyses. alpha-fetoprotein quantification was performed using Finecare™ Rapid AFP quantitative Strip (made in China) which uses a fluorescence immunochromatography Assay. Quantification of transaminases (ALT and AST) was performed using sprinreact reagents (made in Spain).
Ethical considerations: to respect the ethics of medical research, the study was authorized by the University Teaching Hospital of Yaoundé (N° 3235/AR/CHUY/DG/DGA/CAPRC), Yaoundé General Hospital (N/Ref: 213-21/HGY/DG/DPM/MA-TR). The CSCCD allowed the laboratories analysis. Each patient who participated in this study read the information sheet then gave their signed consent and finally an anonymity code was assigned to them.
Sample size calculation: the minimum sample size was 148 participants. The calculation of this sample size was made using the prevalence of HCV, which are 6.5% in Cameroon [6], and according to GLOBOCAN data from 2020 which provided a prevalence of 4.2% for liver cancer in Cameroon [24]. We used the following formula [25]:
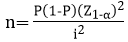
Z = the level of statistical significance with a 95% confidence interval (CI) of 1.96; i= the level of precision of 0.05; P =prevalence of outcomes.
Statistical analysis: for each participant recruited, qualitative and quantitative variables were collected by either interviewing the participant or analyzing blood samples. The collected data were recorded and processed using Excel Version 2016 (Microsoft Corp., USA). Analyses were done using the biostatistical software Statistical Package for Social Sciences (SPSS) Version 25.0. The Chi-square test allowed us to compare the proportions between the different groups and the non-parametric Kruskal Wallis test allowed us to compare the means ± standard deviations between the different groups. A p-value of < 0.05 was considered statistically significant.
Participants: we offered the study to 619 patients who came for consultation at the UTHY or the YGH, respectively 331 patients at the UTHY and 288 patients at the YGH. Of these 619 patients, 41 refused to participate in the study, which gave a non-response rate of 6.6%, 92 were excluded because they were on their first day of consultation and 294 did not meet the inclusion criteria. The remaining 192 participants were on anti-HCV treatment, of which 32 were positive for Human Immunodeficiency Virus (HIV) and were excluded. Of the remaining 160 participants, 28 were at less than twelve weeks of treatment, 92 were at 12 weeks of treatment and 40 were at more than 12 weeks of treatment. Among the patients who were at 12 weeks of treatment, 69 (75%) tested positive for HCV Ab, out of these participants, only 44 (47.8%) were positive for HCV Ag (19 CH, 14 cirrhosis, and 11 HCC). We randomly selected 10 chronic hepatitis, 10 cirrhosis, and 10 HCC samples to quantify AFP and transaminases (Figure 1).
Sociodemographic and clinical characteristics of study participants: overall, participants with chronic hepatitis (CH) were the majority with 19 HCV-positive participants. But the HCC group had the highest average age with 65.8 ± 4.2 years. The 50-59 and 60-69 age groups were the most represented with 8 (42.1%) and 8 (72.7%) respectively for CH and HCC. The male gender 11 (57.9%) of the CH group was most represented. According to marital status, married participants were the most represented in the CH group with 12 (63.1%). For scarification, the CHC group was the most represented with 4 (36.3%) and 5 (26.3%) of CH participants having tattoos. Seven (36.8%) CH participants reported that they consumed alcohol before their illness while only 2 (18.1%) HCC participants reported smoking cigarettes before their illness. Two participants both CH and Cirrhosis groups respectively (10.5) and (14.2%) had received a blood transfusion before their disease. Concerning anti-HCV treatment, 8 (42.1%) participants were taken sofosbuvir + ribavirin (Table 1).
HCV profile of study participants: out of the 192 participants on HCV antiviral treatment, 92 were HIV-negative at the 12th week of their treatment. Of these participants, 58 had chronic hepatitis (CH), 21 had cirrhosis, and 13 had HCC. Sixty-nine participants were positive for the HCV Ab test respectively 37/58 (63.7%) CH; 19/21 (90.4%) Cirrhosis, and 13/13 (100%) HCC (P<0.009), and 23 tests were invalid results. We excluded these samples from our analysis. Out of positive HCV Ab participants, only 44 were positive for HCV Ag with ELISA with optical densities equal to or greater than the threshold, ratios ≥ 1 (Table 2). These samples were not retested following a lack of reagents. They were declared positive for HCV in this study respectively 19/58 (51.3%) CH, 14/21 (73.6%) Cirrhosis, and 11/13 (84.6%) HCC (P<0.000 1) (Table 3).
Biochemical markers of HCV-positive participants simultaneously on RDT and ELISA: participants whose samples were ELISA positive were randomly selected. Each group was made up of 10 participants. Transaminase was tested for each group ALT (GPT) and ASAT (GOT) were higher in participants with HCC (75.4±7.3 IU/L and 72.2 ± 13.8 IU/L respectively) compared to participants with CH and Cirrhosis, with P<0.0001. Regarding AFP, participants with HCC had also a higher mean of AFP of 249.8±6.1 ng /ml, followed by participants with cirrhosis 82.3±10.7 ng /ml, and finally participants with CH 58.5±26.3 ng /ml (P<0.0001) (Table 4).
By 2030, with the treatment of hepatitis C using DAAs, the WHO goal is to eliminate HCV. Therefore, unsatisfactory HCV treatment outcomes must be reported to facilitate better management of those seeking care. This study aimed to estimate the seroprevalence of HCV Ag in participants with liver disease and HCC at the 12th week of treatment. Of the 69 samples positive for HCV Ab with the RDT, only 44 samples were positive for HCV Ag with ELISA, respectively 19/37 (51.3%) HC, 14/19 (73.6%) Cirrhosis, 11/13 (84.6%) HCC (P<0.0001). These results show a high prevalence of HCV Ag, which translates into a long and very expensive treatment for patients. Since some patients achieve healing rather at the 24th week this would mean that the treatment of these patients will have to be prolonged until the 24th week. Since treatment against HCV is expensive in our context, this appeals to public authorities to bear the costs of treatment more because infected patients come from the most disadvantaged sections of the population. In other words, to remain in the policy of eradicating HCV, the governments of developing countries must set up solid policies for managing the treatment of people with HCV so that they can achieve the sustained virologic response (SVR). This implies that patients whose HCV Ag results by ELISA are positive at the 12th week of their therapy require special follow-up. Indeed, several studies have been interested in assessing the role of HCVA in the diagnosis of hepatitis C. It has been demonstrated that the HCV Ag marker can be used to diagnose hepatitis C and of evaluating HCV SVR [3] because there is a correlation between the titter of HCVAg and the viral load of HCV, which makes the capsid antigen a marker of viremia and viral replication of HCV [23]. It, therefore, remains important to carefully follow up with people receiving HCV treatment so that increased efforts can be made to identify people who are successful or unsuccessful to converge toward the international HCV elimination goals. Although HCV-Ag testing can potentially replace viral load for HCV diagnosis or monitoring in some settings, national and international guidelines still advocate the use of PCR as the gold standard test for monitoring progress during and after treatment and is currently still recommended [21].
Our results may have been underestimated given the fact that we only considered ELISA positive for all samples whose optical density was greater than or equal to the threshold (ratio ≥ 1) and that we did not retest the samples with the threshold (ratio between 0.9 and 1). Indeed, it is known that the treatment of hepatitis C using DAAs very rarely eliminates HCV in all people treated. This is probably due to certain risk factors such as on the one hand the advanced age and health status of the patient and on the other hand, the cost, the duration, and the missed doses can negatively affect the results of the treatment [23,26-28]. Added to this, the lack of inexpensive viral diagnostic tests and universally accessible treatment adapted to resource-limited settings represent an obstacle to the implementation of HCV surveillance in difficult contexts [22].
Regarding biochemical markers to assess the liver health status of study participants at their 12th week of treatment, CHC participants had the highest ALT (GPT), AST (GOT), and AFP. high with respectively 75.4±7.3 IU/L; 72.2±13.8 IU/L; 249.8±6.1 ng/mL compared to participants with CH and Cirrhosis (P<0.0001). The transaminase test (ALT and AST) is simple, practical, inexpensive, and non-invasive but not specific to liver disease. Indeed, transaminases are present respectively in the cytoplasm and mitochondria of hepatocytes and muscle cells. When hepatocytes are damaged by the cytopathic effects of oncogenic hepatitis viruses, transaminases are released into the bloodstream resulting in increased serum levels of ALT and AST in the peripheral blood. It is also possible that inflammatory stimulation due to tumour progression stimulates hepatocytes to produce abundant amounts of transaminases compared to what would be found in patients with early liver disease [29]. Besides its role in the diagnosis of HCC, higher serum AFP levels were also observed in participants with HCC. Alpha-fetoprotein is currently the most widely used serum biomarker for screening and early diagnosis of HCC, as well as for evaluating the efficacy and prognosis of HCC treatment. However, not all HCC tumours contribute to an increase in AFP. This makes this marker non-specific to the CHC facility. It is therefore important to explore the search for new biomarkers capable of overcoming the shortcomings of transaminases and AFP to complement X-ray examinations in the hope of achieving HCC screening rates close to those that would be obtained using the biopsy [22,30].
The results obtained in our study can be used to update and add epidemiological data to influence policymakers in the policy of prevention and control of HCV during treatment in Cameroon. We excluded all patients with comorbidities such as diabetes, and HIV because these could play a major role in the ineffectiveness of the treatment. Only plasma samples were considered in this work, we could have obtained more revealing results if we had used the serum, whole blood, or biopsy fragments. The fact of not having retested positive samples as indicated by the Monolisa™ HCV Ag-Ab ultra-protocol constitutes a limitation insofar as certain samples declared positive would have been negative and vice versa. As other limitations, we can note the lack of staging of cirrhotic participants and those with HCC. To achieve the WHO objectives plan to eliminate HCV by 2030, sub-Saharan Africa, in particular Cameroon, will have to actively prioritize the implementation of several strategies such as the reduction or even total coverage of treatment costs. of HCV by public authorities. Identify groups at high risk of therapeutic failure, so that they benefit from special attention in the management of their treatment and indirectly prevent possible resistance to antiviral treatment, and monitor the rebound of viral load and the diversion towards occult hepatitis C.
Our results showed a high prevalence of HCV Ag in patients with liver disease and hepatocellular carcinoma at their 12th week of treatment, suggesting a high prevalence of early treatment failure. These results challenge public policies to develop treatment management strategies to prevent treatment failure and treatment desertion due to Anti HCV treatment costs in our context and encourage the development of the vaccine against HCV. All this is to prevent mortality from cirrhosis, liver failure, and hepatocellular carcinoma.
What is known about this topic
- Sustained virological response measured at 12th or 24th weeks of treatment;
- More than 50% of HCV patients fail treatment.
What this study adds
- Highlight a high prevalence of HCV Ag at 12 weeks of treatment.
The authors declare no competing interests.
Donatien Serge Mbaga, Jacky Njiki Bikoï, and Sara Honorine Riwom Essama were responsible for the conception and design of the study as well as project administration. Donatien Serge Mbaga, Etienne Atenguena Okobalemba, were responsible for the data collection. Donatien Serge Mbaga, Jacky Njiki Bikoï, Justin Olivier Essindi, and George Ikomey Mondinde, were involved in the laboratory analysis and interpretation of results. Donatien Serge Mbaga was responsible for statistical analysis. Donatien Serge Mbaga wrote the original draft. Sebastien Kenmoe, Justin Olivier Essindi, Mbongue Mikangue Chris André, Alexandra Emmanuelle Membangbi, Aicha Ngoutane, Arnaud Franck Elang, Sabine Aimée Touangnou-Chamda, and Carole Stéphanie Sake, reviewed the first draft. Sara Honorine Riwom Essama oversees all steps. All the authors have read and agreed to the final manuscript.
We thank the University Teaching Hospital of Yaoundé, Yaoundé General Hospital, the CSCCD of FMBS of the University of Yaoundé, and the Laboratory of Microbiology of the University of Yaoundé I for respectively authorizing this work and giving use of the workspace. We also thank all participants for agreeing to participate in this work.
Table 1: sociodemographic and clinical characteristics of HCV Ag positive participants after 12 weeks of treatment
Table 2: profile of HCV markers sought in participants at 12 weeks of treatment
Table 3: seroprevalence of HCV Ag tested in participants at 12 weeks of treatment
Table 4: biochemical markers of HCV Ag positive participants by ELISA at 12 weeks of treatment
Figure 1: flow Chart of selection of participants
- WHO. Hepatitis: overview. 2022. Accessed April 13, 2022.
- WHO. Hepatitis C. 2021. Accessed April 26, 2022.
- Wasitthankasem R, Vichaiwattana P, Siripon N, Posuwan N, Auphimai C, Klinfueng S et al. Liver disease burden and required treatment expenditures for hepatitis C virus (HCV) infection in Thailand: Implications for HCV elimination in the new therapeutic era, a population-based study. PLoS ONE. 2018;13(4):e0196301. PubMed | Google Scholar
- Mbaga DS, Kenmoe S, Kengne-Ndé C, Ebogo-Belobo JT, Mahamat G, Foe-Essomba JR et al. Hepatitis B, C and D virus infections and risk of hepatocellular carcinoma in Africa: a meta-analysis including sensitivity analyses for studies comparable for confounders. PLoS One. 2022;17(1):e0262903. PubMed | Google Scholar
- Organisation mondiale de la Santé, Comité Regional Afrique. Rapport de situation sur la mise en œuvre du cadre d´action de la stratégie mondiale du secteur de la santé pour la prévention, les soins et le traitement de l´hépatite virale 2016-2021 dans la région Africaine. Bureau Régional de l´Afrique. 2018. Accessed April 16, 2022.
- Bigna JJ, Amougou MA, Asangbeh SL, Kenne AM, Nansseu JR. Seroprevalence of hepatitis C virus infection in Cameroon: a systematic review and meta-analysis. BMJ Open. 2017;7(8):e015748. PubMed | Google Scholar
- Djuidje Ngounoue M, Fepa Kwesseu AG, Nwobegahay J, Moundipa Fewou P. HBV and HCV seroprevalence and the predominant HCV genotypes in a Hospital setting in Cameroon. Virol Mycol. 2018;7(178):2161-0517. Google Scholar
- Kowo MP, Andoulo FA, Ngek LT, Sizimboue DT, Ndam AN, Ondo BE et al. Prevalence of hepatitis C virus and associated risk factors among inmates at New Bell Prison, Douala, Cameroon. Open Journal of Epidemiology. 2019 May 6;9(02):119. Google Scholar
- Tsague MK, Fotio AL, Bomgning CLK, Nguefack-Tsague G, Fopa F, Nguelefack TB. Prevalence of viral and non-viral hepatitis in Menoua Division, West Region, Cameroon: a retrospective hospital-based study. Pan Afr Med J. 2019 Apr 29;32:212. PubMed | Google Scholar
- Ngoupa JB, Njukeng PA, Akwa EN, Kengne M, Tamoufe U, Ter Goon D et al. Seroprevalence and associated risk factors for hepatitis B virus infection among barbers and their clients in two cities in Cameroon. Southern African Journal of Infectious Diseases. 2019 Mar 1;34(1):1-5. Google Scholar
- Njouom R, Siffert I, Texier G, Lachenal G, Tejiokem MC, Pépin J et al. The burden of hepatitis C virus in Cameroon: spatial epidemiology and historical perspective. J Viral Hepat. 2018;25(8):959-968. PubMed | Google Scholar
- Ndifontiayong AN, Ali IM, Sokoudjou JB, Ndimumeh JM, Tume CB. The effect of HBV/HCV in response to HAART in HIV patients after 12 months in Kumba Health District in the South West Region of Cameroon. Trop Med Infect Dis. 2021;6(3):150. PubMed | Google Scholar
- Eyong EM, Yankam BM, Seraphine E, Ngwa CH, Nkfusai NC, Anye CS et al. The prevalence of HBsAg, knowledge and practice of hepatitis B prevention among pregnant women in the Limbe and Muyuka Health Districts of the South West region of Cameroon: a three-year retrospective study. Pan Afr Med J. 2019;32:122. PubMed | Google Scholar
- Pockros PJ. Management of patients who have achieved sustained virologic response for hepatitis C virus infection. Gastroenterology & Hepatology. 2018 May;14(5):305. Google Scholar
- Lee JS, Choi HJ, Lee HW, Kim BK, Park JY, Kim DY et al. Screening, confirmation, and treatment rates of hepatitis C virus infection in a tertiary academic medical center in South Korea. Journal of Gastroenterology and Hepatology. 2021;36(9):2479-2485. PubMed | Google Scholar
- Rocco A, Compare D, Coccoli P, Esposito C, Di Spirito A, Barbato A et al. Vitamin B 12 supplementation improves rates of sustained viral response in patients chronically infected with hepatitis C virus. Gut. 2013;62(5):766-773. PubMed | Google Scholar
- Innes HA, Hutchinson SJ, Allen S, Bhattacharyya D, Bramley P, Delahooke TES et al. Excess liver-related morbidity of chronic hepatitis C patients, who achieve a sustained viral response, and are discharged from care. Hepatology. 2011;54(5):1547-1558. PubMed | Google Scholar
- Nahon P, Layese R, Cagnot C, Asselah T, Guyader D, Pol S et al. HCV Eradication in primary or secondary prevention optimizes hepatocellular carcinoma curative management. Cancer Prevention Research. 2021;14(5):581-592. PubMed | Google Scholar
- Lee HW, Lee HJ, Hwang JS, Sohn JH, Jang JY, Han KJ et al. Lamivudine maintenance beyond one year after HBeAg seroconversion is a major factor for sustained virologic response in HBeAg-positive chronic hepatitis B. Hepatology. 2010;51(2):415-421. PubMed | Google Scholar
- Chan HL, Wong VW, Tse AM, Tse C, Chim AM, Chan H et al. Serum hepatitis B surface antigen quantitation can reflect hepatitis B virus in the liver and predict treatment response. Clinical Gastroenterology and Hepatology. 2007;5(12):1462-1468. PubMed | Google Scholar
- Adland E, Jesuthasan G, Downs L, Wharton V, Wilde G, McNaughton AL et al. Hepatitis virus (HCV) diagnosis and access to treatment in a UK cohort. BMC Infect Dis. 2018;18(1):461. PubMed | Google Scholar
- Pawlotsky J-M, Negro F, Aghemo A, Berenguer M, Dalgard O, Dusheiko G et al. EASL recommendations on treatment of hepatitis C: final update of the series. Journal of Hepatology. 2020;73(5):1170-1218. PubMed | Google Scholar
- Rossetti B, Loggi E, Raffaelli CS, Mercinelli S, Gandolfo C, Savellini GG et al. Hepatitis C virus core antigen (HCVAg): an affordable assay to monitor the efficacy of treatment in DAAs era. 2021;44(2):89-94. Google Scholar
- GLOBOCAN. Estimated number of prevalent cases (3-year) as a proportion in 2020, liver, both sexes, all ages, Africa. 2020. Accessed February 23, 2022.
- Wang X, Ji X. Sample size estimation in clinical research: from randomized controlled trials to observational studies. Chest. 2020;158(1S):S12-S20. PubMed | Google Scholar
- Ghenea AE, Ungureanu AM, Turculeanu A, Popescu M, Carsote M, Ţieranu ML et al. Predictors of early and sustained virological response of viral hepatitis C. Rom J Morphol Embryol. 2020;61(4):1185-1192. PubMed | Google Scholar
- Nabulsi NA, Martin MT, Sharp LK, Koren DE, Teply R, Zuckerman A et al. Predicting treatment failure for initiators of hepatitis C virus treatment in the era of direct-acting antiviral therapy. Front Pharmacol. 2020;11:551500. PubMed | Google Scholar
- Tmu N, Kumar A, Sharma P, Singla V, Bansal N, Arora A. Results of sofosbuvir plus ribavirin in patients with hepatitis C related decompensated cirrhosis. Journal of Clinical and Experimental Hepatology. 2019;9(1):4-12. PubMed | Google Scholar
- Chan HLY, Chen CJ, Omede O, Al Qamish J, Al Naamani K, Bane A et al. The present and future disease burden of hepatitis C virus infections with today´s treatment paradigm. Journal of Viral Hepatitis. 2017;24(S2):25-43. PubMed | Google Scholar
- Huang CF, Yu ML. Unmet needs of chronic hepatitis C in the era of direct-acting antiviral therapy. Clin Mol Hepatol. 2020;26(3):251-260. PubMed | Google Scholar
Search
This article authors
On Pubmed
- Donatien Serge Mbaga
- Jacky Njiki Bikoï
- Etienne Atenguena Okobalemba
- Justin Olivier Essindi
- Chris André Mbongue Mikangue
- Alexandra Emmanuelle Membangbi
- Aïcha Ngoutane
- Arnaud Franck Elang
- Sabine Aimée Touangnou-Chamda
- Carole Stéphanie Sake
- George Ikomey Mondinde
- Sebastien Kenmoe
- Sara Honorine Riwom Essama
On Google Scholar
- Donatien Serge Mbaga
- Jacky Njiki Bikoï
- Etienne Atenguena Okobalemba
- Justin Olivier Essindi
- Chris André Mbongue Mikangue
- Alexandra Emmanuelle Membangbi
- Aïcha Ngoutane
- Arnaud Franck Elang
- Sabine Aimée Touangnou-Chamda
- Carole Stéphanie Sake
- George Ikomey Mondinde
- Sebastien Kenmoe
- Sara Honorine Riwom Essama












