Performance of immunochromatographic and immunoenzymatic techniques in the diagnosis of toxoplasmosis in pregnant women in Cameroon: need for harmonization
Jacqueline Félicité Yéngué, Claudine Mireille Ntsama Essomba, Georgia Elna Ambada Ndzengue, Nelson Sonela, Elise Elong Lobè, Michel Carlos Tommo Tchouaket, Aude Christelle Ka´e, Jules Colince Tchadji, Sebastien Kenmoe, Aubin Joseph Nanfack, Charles Félix Bilong Bilong
Corresponding author: Jacqueline Félicité Yéngué, Department of Animal Biology and Physiology, University of Yaoundé I, Yaoundé, Cameroon 
Received: 24 Aug 2022 - Accepted: 17 Oct 2022 - Published: 16 Dec 2022
Domain: Infectious diseases epidemiology
Keywords: Toxoplasmosis, pregnant women, diagnosis, RDT, ELISA, performance, Cameroon
©Jacqueline Félicité Yéngué et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Jacqueline Félicité Yéngué et al. Performance of immunochromatographic and immunoenzymatic techniques in the diagnosis of toxoplasmosis in pregnant women in Cameroon: need for harmonization. Pan African Medical Journal. 2022;43:195. [doi: 10.11604/pamj.2022.43.195.36996]
Available online at: https://www.panafrican-med-journal.com//content/article/43/195/full
Research 
Performance of immunochromatographic and immunoenzymatic techniques in the diagnosis of toxoplasmosis in pregnant women in Cameroon: need for harmonization
Performance of immunochromatographic and immunoenzymatic techniques in the diagnosis of toxoplasmosis in pregnant women in Cameroon: need for harmonization
![]() Jacqueline Félicité Yéngué1,&, Claudine Mireille Ntsama Essomba1, Georgia Elna Ambada Ndzengue1,2,
Jacqueline Félicité Yéngué1,&, Claudine Mireille Ntsama Essomba1, Georgia Elna Ambada Ndzengue1,2, ![]() Nelson Sonela2,3, Elise Elong Lobè2,
Nelson Sonela2,3, Elise Elong Lobè2, ![]() Michel Carlos Tommo Tchouaket2,4, Aude Christelle Ka´e2,3,
Michel Carlos Tommo Tchouaket2,4, Aude Christelle Ka´e2,3, ![]() Jules Colince Tchadji1,2,
Jules Colince Tchadji1,2, ![]() Sebastien Kenmoe5, Aubin Joseph Nanfack2, Charles Félix Bilong Bilong1
Sebastien Kenmoe5, Aubin Joseph Nanfack2, Charles Félix Bilong Bilong1
&Corresponding author
Introduction: in order to contribute to the improvement of the management of toxoplasmosis in pregnant women in Cameroon, performance of two techniques commonly used in the diagnosis of toxoplasmosis was evaluated.
Methods: a total of 541 pregnant women were recruited from seven hospitals in two Regions of Cameroon, of which 63% (341: Batch1) were from health facilities (HF) using a immunochromatographic technique (ICT) as a screening test for toxoplasmosis, and 37% (200: Batch2) from those using an immunoenzymatic technique (IEZ). On each sample, Ig (Immunoglobulin) G (IgG) and IgM were tested by three techniques: a a Rapid Diagnostic Test (RDT), an Enzyme Linked Immuno Sorbent Assay (ELISA) and a VIDAS Enzyme-linked fluorescent assay taken as reference (VIDAS/ELFA. The results from the health facilities were recorded.
Results: for the IgG assay, our two laboratory methods were sensitive (96.0% and 97.5%) and specific (64.2% and 59.7%). Their concordance rates with the VIDAS/ELFA reference were above 60% (P<0.001). Moreover, for the IgM assay, the performances of the two methods were equivalent: Se= 18.2%, Sp= 99.4% with a low concordance rate (Kappa = 0.24). Considering the results provided by the selected hospitals, the ELISA used in Batch2 showed similar performances to the two techniques used in reference lab while the performances were low for the RDT used in Batch1.
Conclusion: both methods showed similar performances (good for (IgG) and poor for IgM). However, for the immunochromatographic method, differences in performance were found between our results and those provided by the selected health facilities. These differences suggest a harmonization of diagnostic techniques for toxoplasmosis in pregnant women in Cameroonian health facilities.
Toxoplasmosis, anthropozoonosis, remains a public health problem. It is a cosmopolitan infectious disease, affecting about one third of the world's population [1,2]. It is caused by Toxoplasma gondii (T. gondii), an obligate intracellular protozoan parasite [3]. This parasite, whose definitive host is the cat or other felids, can affect all warm-blooded animals, including humans [4]. Toxoplasmosis prevalence is less than 10% in hot and dry climates and between 60 and 80% in humid areas [5]. It is generally higher in the Americas and sub-Saharan Africa due to the abundance of cats and a climate favorable to oocysts survival [6]. Toxoplasmosis is asymptomatic in immunocompetent individuals, but is serious in immunocompromised individuals and pregnant women. In the latter, the absence of immunity to T. gondii exposes the individual to primary infection and the risk of foetal transmission, which can lead to early abortion and congenital malformations [7,8]. The risk of foetal infection increases with gestational age at the time of infection, and is often highest in the third trimester. Foetal damage is more severe when maternal infection occurs in the first trimester [9]. It is therefore of great interest to monitor the immunological status of toxoplasmosis in pregnant women. In the latter, immunological monitoring is based on the detection of markers of infection, notably IgG and IgM antibodies to T. gondii. One week after the onset of infection, IgM, a marker of recent infection, is the first to appear; and IgG, a marker of old infection, appears 2 to 3 weeks after IgM. Depending on the pattern presented, infection is considered acute (IgG+/IgM+), recent (IgG-/IgM+), chronic (IgG+/IgM-), and absent (IgG-/IgM-). [10,11]. In Cameroon, confirmation of the first two profiles leads to drug treatment. Thus, T. gondii is investigated in pregnant women using serological techniques. However, the use of these tools remains a concern. Indeed, the technique are numerous; they differ both in their principles and in the nature of the reagents used; also there are even differences in the interpretation of the results [12,13]. In Cameroon, some studies have shown that the prevalence of toxoplasmosis in pregnant women varies from one year to another, from one region to another, and from one city to another. Thus, from 1981, it was 78.6% in Douala [14], 65% in Penka-Michel [15], and 35.77% in Buea, [16]. The serological diagnostic techniques applied in local studies have been diverse: Enzyme Immuno Assay (EIA) [15], RDT [17], and ELISA [18]. In the absence of national planning, do current strategies for diagnosing toxoplasmosis in pregnant women improve management in Cameroon? In order to contribute to the improvement of the management of toxoplasmosis in pregnant women in Cameroon, we evaluated the performance of two techniques immunochromatographic (ICT) and immunoenzymatic (IEZ) commonly used in health facilities.
A multicenter, cross-sectional, descriptive and analytical study was conducted from June 2019 to February 2020 in seven Catholic health facilities in the Center (03) and Littoral (04) regions. The health facilities in the Center were located in urban areas. Those in the Littoral were equally divided between urban and rural areas. In the health facilities using the ICT method (by RDT), the pregnant women recruited were grouped in batch 1 (341), and those using the IEZ method (by ELISA) in batch 2 (200).
Inclusion criteria: the study population consisted of pregnant women aged 14-46 years who were seen for the first time at antenatal clinics (ANC) in the health facilities selected for the study.
Sample size: based on the average of the toxoplasmosis prevalence values published by Njunda et al. [19] in the Center i.e. 65.5% and Nguefack et al. [14] in the Littoral i.e. 78.6%, a minimum sample size of 310 participants was obtained from the formula:
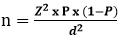
In this formula, n = sample size, Z (1.96) = the statistic corresponding to level of confidence with 95%, P = expected prevalence and d = precision´s level at 0.05.
Sample analysis: for each participant, socio-demographic data were collected on individual survey sheets. Next, 3mL x 2 of venous blood were collected and divided into two dry tubes. The first tube was used for serological analysis (RDT or ELISA) in the health facilities. The description of the kits used is given in Annex 1. The second tube of blood was sent to the reference laboratory: the Immunology and Microbiology Laboratory (LIM) of the Chantal BIYA International Reference Center (CIRCB). In the laboratory, the tube was left to rest for 1 hour. After coagulation, it was centrifuged at 3000 rpm for 15 minutes; the serum obtained was aliquoted in cryotubes and stored at -80°C. Anti-toxoplasma IgG and IgM antibodies were tested by ICT (TDRLIM), EIA (ELISALIM), and ELFA methods of VIDAS Biomérieux the latter being taken as reference method. In the health facilities included in this study, the serological diagnosis of toxoplasmosis in pregnant women is carried out by the ICT or IEZ techniques. As the harmonization of these methods is not effective in our context, we choose the ELFA method from VIDAS Biomérieux as a reference. This is an automated test based on a two-step IEZ assay for IgG and immunocapture for IgM with a final detection by fluorescence [21-23]. The description of the kits used at the LIM is given in Annex 2.
Data analysis: the categorical variables are presented as numbers and frequencies. Chi-square and Fisher's exact tests were used to compare the proportions of the different categorical variables using SPSS version 25 software (SPSS, Inc., Chicago, IL, USA). The Z value (Z) was calculated for the comparison of the two methods with matched data. Sensitivity (Se) and specificity (Sp) were calculated using the software [24]. The Youden index and the kappa agreement coefficient (Ka) were also calculated. The significance level for differences was set at p < 0.05.
Ethical considerations: the protocol of this study received the approval of the National Ethics Committee through the issuance of the Ethical Clearance N°2019/05/18/CE/CNERSH/SP, as well as the administrative authorisations from the managers of the different health facilities retained. Written informed consent was also obtained from each eligible and willing pregnant woman.
Sociodemographic profile of pregnant women: the project was presented to 1070 pregnant women, 50.6% (541) consented to participate. Their ages ranged from 14 to 46 years, with an average of 27.67 ± 5.58 years. The modal class was (21-31) years representing 61.4% (332/541) of the study sample; 63.2% were unmarried; 58.4% had attained secondary education; 57.9% were registered in the 2nd trimester of pregnancy; 72.2% were in at least their second pregnancy; 73.9% had no knowledge of toxoplasmosis; 64% did not recognize the presence of a cat in their environment.
Toxoplasmosis infection rates obtained in the health facilities with RDT: only 13.3% of samples were positive. With the VIDAS ELFA method, the infection rate was 6 times higher (80.4%). The difference between the two methods was significant between the infection rates of IgG (p<0.0001) (Table 1).
Toxoplasmosis infection rates obtained in health facilities using ELISA: the infection rates of toxoplasmosis obtained by ELISA Batch2 and ELFA were similar at 75% and 74% respectively. For IgG, the difference between the infection rates determined by these two methods was not significant (p>0.05), but was significant for IgM (p<0.05). (Table 2).
Toxoplasmosis infection rates obtained in the Immunology and Microbiology Laboratory of the CIRCB on the two batches of samples: in the subpopulation 1, the toxoplasmosis infection rate was 76.8% with TDRLIM and 86.8% with ELISALIM. In subpopulation 2, these rates ranged from 71% with TDRLIM to 81.5% with ELISALIM. For both anti-T. gondii antibody assays, the IgG levels obtained by the two methods were different in the two batches (p < 0.0001). On the other hand, the IgM frequencies obtained by the two methods were statistically similar in Batch 1 (p=0.32), and for Batch 2 (p=0.65) (Table 3).
Performance of immunochromatographic and enzyme immunoassay methods
IgG detection: for the detection of IgG in the first batch of samples, the RDTBatch1 applied in the health facilities was not very efficient and less concordant with the reference, it was highly specific and poorly sensitive. Its positive predictive value (PPV) was above 80%, and the negative predictive value (NPV) very low (20%). On the other hand, the two methods tested at LIM on the same samples showed similar performances; they were very sensitive but less specific. Their predictive values were similar, over 90% for PPV, and over 70% for NPV. The values of the Youden index (0.6) and the kappa coefficient (≥ 0.7) indicate that both experimental methods are much better (Table 4). However, in the second batch, the ELISA technique applied in the hospitals and our two experimental methods showed similar and good performance in the IgG assay. For all three techniques, the sensitivity was > 90%, the specificity > 70%. In this second batch, PPV and NPV were above 80%. Furthermore, the Youden index values (≥ 0.7) indicate that all three methods are good and well suited for IgG detection (Table 4).
IgM detection: for the IgM assay, the methods used both in the health facilities and in the Immunology and Microbiology Laboratory of the CIRCB were specific (Sp > 91%) but not very sensitive (Se < 38%). For the IgM assay, the different methods presented in both batches showed NPVs above 90%. In contrast, the PPVs were below 30% for the RDTbatch 1 and in all 3 methods in batch 2. All of them also appeared to be not very effective, since the Youden index ≤ 0.3 (Table 4).
In order to contribute to the improvement of the management of toxoplasmosis in pregnant women in Cameroon, we determined the prevalence of toxoplasmosis with two techniques routinely used in hospitals; we further evaluated the performance of these technique. This is the first multicenter study of its kind in the diagnosis of toxoplasmosis in Cameroon. Two groups of serological test results collected respectively from health facilities (RDTBatch1 and ELISABatch2) and at the Immunology and Microbiology Laboratory (LIM) of the CIRCB (RDTLIM and ELISALIM) are presented. The participants were recruited from 7 health facilities; 5 of which were tested with RDT and 2 with ELISA for biological diagnosis of toxoplasmosis. Of these pregnant women, 341 were recruited in five health facilities (Batch 1) and 200 in the other two (Batch 2). In Batch1, the prevalence of toxoplasmosis in pregnant women using the ICT/RDT method was 13.3%. This rate is much lower than those reported by previous studies in pregnant women in Cameroon using the same technics, i.e. 22.84% in Yaoundé [25], 35.4% in Bamenda [26], 22.9% in Buea [16], and 32.4% in Yaoundé [27]. Our prevalence (13.3%) is also lower than other studies conducted with the ICT method in West Africa, namely 44.4% in Nigeria [28] and 48.5% in Benin [29]. At LIM/CIRCB, the ICT and EIA methods applied to the same samples gave infection rates of 76.8% and 86.8% respectively, well above 13.3%. This discrepancy could be related to management of inventories, storage procedures, validity of the tests at the time of use, and the competence of the laboratory staff. Several cases of toxoplasmosis would therefore be ignored, which would give rise to risks of neonatal infections, premature abortions, etc.
Using the IEZ/ELISA method, the seroprevalence of toxoplasmosis in Batch 2 was 75%. This result is close to the data from other studies carried out in pregnant women using the same method, namely 70% and 78.6% in the city of Douala [14,30] in Cameroon. It is also close to 71% obtained by the ICT method tested at the LIM of CIRCB. Nevertheless, this result (75%) collected from health facilities is slightly lower than 81.4%, the infection rate reported in Ethiopia [31], and 81% obtained at LIM with the IEZ technique. However, this seroprevalence (75%) is higher than the 36.5% obtained in Mbouo-Mbandjoun (West Region of Cameroon) [18] and 65.5% obtained in Yaoundé [19] determined by the same technics. Moreover, it is also higher than that of studies conducted outside Cameroon using the same method in pregnant women, i.e. 69% in Brazil [32] and 48.9% in Nigeria [6]. The locality or environment, cultural dietary habits, thus seem to have an impact on the probability of contacting toxoplasmosis [5].
The results collected from the health facilities and those obtained at LIM/CIRCB show differences in the prevalence of anti-toxoplasmic IgG/IgM. In the health facilities as well as in the Immunology and Microbiology Laboratory of the CIRCB, for the ELISA technique, the number of sera containing IgG was higher than with the RDT. However, both methods were equivalent in the detection of IgM. The differences observed between the infection rates reported by the health facilities in our study and those obtained at the LIM of the CIRCB, using the same methods and on the same samples, illustrate the difficulties in interpreting serological results for toxoplasmosis [13]. In Batch1, the sensitivity of the RDT (12%) for IgG detection is much lower than the 54.4% obtained in Egypt [33], the 46.7% found in Nigeria [34], and the 96% obtained at LIM/CIRCB with the same technique. These differences could be related to the handling conditions on the one hand and the quality of the kits used on the other. The value of Se/RDTBatch1=12% is also lower than 97% obtained in Benin [29]. This other difference in sensitivity would be related to the difference in the principles of the methods applied [35]; indeed, the RDT applied in the Benin study was based on an enzyme-linked immunosorbent method, which could justify its best performance. This is supported by the high sensitivity level of 97.45% obtained at LIM with the ELISA technique tested on the same batch of samples (Table 4). In subpopulation 2, the ELISABatch2 technique applied in health facilities showed similar performance to our two experimental methods against IgG (Se ≥ 90% and Sp ≥ 70%). A study carried out in Nantes (France) showed comparable performance (Se=98.87% and Sp=88.46%) when the authors compared the Platelia microplate technique based on the Elisa principle and the VIDAS Biomérieux automated system [36].
Regarding IgM detection, our two experimental techniques and those applied to the two batches of health facilities showed equivalent performance. Their sensitivity was 33.3%. This result is close to 33.3% and 29.03%, sensitivities obtained respectively in Benin [29] and Egypt [33]. The sensitivity of the TDRBatch1 towards IgM therefore still appears to be low in this case. This could be partly due to the large number of false negatives recorded. This low sensitivity of the TDRBatch1 to detect IgM could pose a problem in the diagnosis of acute toxoplasma infections in pregnant women; these results suggest the use of a confirmatory test for IgM [37,38]. In the second batch of samples, the ELISABatch2 technique used in hospital centres and the two technique tested at the LIM had also very low sensitivity (Se ≤ 30%) although quite specific (Sp ≥ 90%). This result differs from that of Martin & Morin [36] who found a sensitivity of 87.08% and a specificity of 96.97% with the same technique. The low sensitivity of IgM recorded in the current study is already reflected in the low positivity rate (10%) (Table 2). This could be explained by the fact that the majority of women recruited in this study were highly immunized on the one hand, RDT and ELISA tests applied both in the hospitals retained in this study and at the LIM of the CIRCB might not be sufficiently sensitive to detect acute infections on the other hand.
Limitations: the bias of this study is that the two groups of results were not obtained on identical samples. The non-uniformity of the numbers in the different health facilities did not allow a good comparison by sampling site and by kit.
The infection rate of toxoplasmosis among pregnant women in health facilities that used RDTBatch1 was 13.2%; and 75% for those that used ELISABatch2. In the laboratory, the prevalences obtained in this study were above 70% for RDTLIM and ELISALIM. Both experimental techniques showed similar performance (good for IgG and poor for IgM). Moreover, only the ELISABatch2 technique showed similar performance to our experiments. The differences in performances observed between the RDTLIM and the one applied in the health facilities suggest that evaluations should be extended with a view of harmonizing diagnostic technique for toxoplasmosis in pregnant women in Cameroonian health facilities.
What is known about this topic
- Toxoplasmosis is a public health problem with severe consequences in pregnant women;
- The use of a variety of serological assays remains a concern and presents a real difficulty to interpret the results, especially in countries with limited resources.
What this study adds
- For IgG detection, the ICT method used in the health facilities was less efficient than the one we tested, with good performance (sensitive and specific). On the other hand, the IEZ method applied in the health facilities and the one used in our experiments showed similar performance (sensitive and specific) in the IgG assay;
- Both methods (ICT and IEZ) were less sensitive and less efficient for IgM detection;
- There is a need for harmonisation of serological diagnostic methods in health facilities in Cameroon.
The authors declare no competing interests.
Jacqueline Félicité Yéngué, Claudine Mireille Ntsama Essomba and Charles Félix Bilong Bilong participated in the design of the study and the drafting of the protocol. Aubin Joseph Nanfack, Georgia Elna Ambada Ndzengue supervised the analyses at the CIRCB laboratory. Jacqueline Félicité Yéngué collected the data and performed the RDT analyses. Michel Carlos Tommo Tchouaket, Aude Christelle Ka´e and Jacqueline Félicité Yéngué performed the serological test on the Biomérieux VIDAS machine. Elise Elong Lobè, Georgia Elna Ambada Ndzengue, Nelson Sonela and Jacqueline Félicité Yéngué were responsible for the ELISA analysis. Jacqueline Félicité Yéngué initiated and performed the statistical analyses under the supervision of Charles Félix Bilong Bilong. The manuscript was drafted and finalised by Jacqueline Félicité Yéngué, Aubin Joseph Nanfack, Georgia Elna Ambada Ndzengue, Sebastien Kenmoe, and Jules Colince Tchadji under the supervision of Claudine Mireille Ntsama Essomba and Charles Félix Bilong Bilong. All authors read and approved the final version of the article.
The authors would like to thank The Douala Sisters´ Servants of Mary for various academic facilities (1), the managers and staff of the ANC and laboratory departments of the following hospitals: Nicolas Barré of Ekounou, Saint Martin de Porès of Mvog Mbetsi, Saint André of Pouma, Saint Padre Pio, Saint Paul of Douala, CASS of Nkoldongo, and the Safacam Health Center in Dizanguè (2), the pregnant women who participated in this study (3), VIDAS BIOMÉRIEUX Company who graciously offered the Toxo IgG/IgM VIDAS kits (4); the CIRCB who provided us with an adequate technical platform for the practical phase of this work (5).
Table 1: infection rates of anti-toxoplasma IgG in RDT facilities
Table 2: frequencies of anti-toxoplasma IgG and IgM in health facilities under ELISA
Table 3: toxoplasmosis infection rates obtained in the laboratory with the RDT and ELISA on the two batches
Table 4: performance of the RDT and ELISA for the detection of IgG and IgM
Annex 1: description of the kits used in the health facilities (319 KB)
Annex 2: description of the kits used at the CIRCB Laboratory (320 KB)
- Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363(9425):1965-76. PubMed
- Robert-Gangneux F, Dardé ML. Epidemiology of and Diagnostic Strategies for Toxoplasmosis. Clin Microbiol Rev. Clin Microbiol Rev. 2012 Apr;25(2):264-96. PubMed | Google Scholar
- Passos ADC, Bollela VR, Furtado JMF, Lucena MM de, Bellissimo-Rodrigues F, Paula JS et al. Prevalence and risk factors of toxoplasmosis among adults in a small Brazilian city. Rev Soc Bras Med Trop. déc 2018;51(6):781-7. PubMed | Google Scholar
- Webster JP. Review of “Toxoplasmosis of Animals and Humans (Second Edition)” by J.P. Dubey. Parasites Vectors. Dec 2010;3:112. Accessed August 16, 2022
- Pappas G, Roussos N, Falagas ME. Toxoplasmosis snapshots: Global status of Toxoplasma gondii seroprevalence and implications for pregnancy and congenital toxoplasmosis. International Journal for Parasitology. 2009;39(12):1385-94. PubMed | Google Scholar
- Nasir IA, Aderinsayo AH, Mele HU, Aliyu MM. Prevalence and Associated Risk Factors of Toxoplasma gondii Antibodies among Pregnant Women Attending Maiduguri Teaching Hospital, Nigeria. J of Medical Sciences. 15 avr 2015;15(3):147-54. Google Scholar
- Nizard J. Toxoplasmose et grossesse. Journal de Gynécologie Obstétrique et Biologie de la Reproduction. 2008 Mar;37 Suppl 1:4-9. PubMed | Google Scholar
- Torgerson PR, Mastroiacovo P. The global burden of congenital toxoplasmosis: a systematic review. Bull World Health Organ. 1 juill 2013;91(7):501-8. PubMed | Google Scholar
- Tourdjman M, Tcheandjieu C, de Valk H, Goulet V, Le Strat Y. Toxoplasmose chez les femmes enceintes en France: évolution de la séroprévalence et des facteurs associés entre 1995 et 2010, à partir des enquêtes nationales périnatales. Bulletin Epidemiologique Hebdomadaire. 2015;15-16:264-72.
- Teimouri A, Mohtasebi S, Kazemirad E, Keshavarz H. Role of Toxoplasma gondii IgG Avidity Testing in Discriminating between Acute and Chronic Toxoplasmosis in Pregnancy. Pritt BS, éditeur. J Clin Microbiol. 2020;58(9):e00505-20. PubMed | Google Scholar
- Villard O, Cimon B, L´Ollivier C, Fricker-Hidalgo H, Godineau N, Houze S et al. A Serological diagnosis of Toxoplasma gondii infection: recommendations from the French National Reference Center for Toxoplasmosis. Diagn Microbiol Infect Dis. Diagnostic Microbiology and Infectious Disease. janv 2016;84(1):22-33. PubMed | Google Scholar
- Bessières MH, Chemla C, Cimon B, Marty P, Gay-Andrieu F, Pellouv H et al. Les difficultés d´interprétation de la sérologie de la toxoplasmose. Revue Francophone des Laboratoires. 1 juin 2006;2006(383):43-9. Google Scholar
- Villard O, Jung-Étienne J, Cimon B, Franck J, Fricker-Hidalgo H, Godineau N et al. Sérodiagnostic de la toxoplamose en 2010: conduite à tenir et interprétation en fonction des profils sérologiques obtenus par les méthodes de dépistage. Feuillets de Biologie. 2011;52(298):43-9.
- Nguefack CT, Meumeu IK, Ngaba GP, Kongnyuy E, Nana Njamen T, Halle Ekane G et al. Prevalence and Factors Associated with Toxoplasma gondii Immunization among Pregnant Women in Douala - Cameroon. J Womens Health, Issues Care. 2016;05(06). Google Scholar
- Kouitcheu Mabeku LB, Tchakounte C, Bonsi ST, Etoa FX. Prevalence of Toxoplasma gondii and Associated Risk Factors among Pregnant Women Attending Hospital Centers in Penka-Michel, Cameroon. JSRR. 26 nov 2018;21(2):1-11. Google Scholar
- Ndamukong-Nyanga JL, Jacqueline N, Flavia D, Ndamukong NC, Nji DN. Socio-Demographic Factors and Attitudes Influencing the Seroprevalence of Toxoplasmosis among Pregnant Women in Buea, Sw Cameroon. South Asian Journal of Parasitology. 2019;3(4). Google Scholar
- Abongwa LE, Signang A, Tibi S, Ngenwi A. Socio-demographic and Obstetric Variations of T. gondii and HIV-1 Co-infection among Pregnant Women in Cameroon. JAMB. 2019;16(3):1-11. Google Scholar
- Guemgne Todjom F, Makou Tsapi E, Gamago GA, Vignoles P, Wabo Pone J, Djuikwo Teukeng FF. Seroprevalence of Toxoplasmosis and associated risk factors in pregnant women at the Protestant Hospital, Mbouo-Bandjoun, Cameroon. Af J Clin Exp Micro. 2019;20(3):221-30. Google Scholar
- Njunda AL, Nsagha DS, Assob JCN, Kamga HLF, Tafili RT, Achidi EA. Seroepidemiology of Toxoplasmosis in Pregnant Women Attending the University Teaching Hospital in Yaounde, Cameroon | International Journal of Health Research. International Journal of Health Research. 2011a;1-9. Google Scholar
- Pourhoseingholi MA, Vahedi M, Rahimzadeh M. Sample size calculation in medical studies. Gastroenterol Hepatol Bed Bench. 2013;6(1):14-7. PubMed | Google Scholar
- Gharavi M, Oormazdi H, Roointan E. A Comparative Study on Sensitivity and Specificity of Conventional and Unconventional IgG and IgM Assays for Diagnosis of Toxoplasmosis. 2008;42-5. Google Scholar
- Weber B, Badiel M, Alvarez-Otero Y, Thulliez P, Montoya LG. Comparative evaluation of AxSYM, VIDAS and VIDIA toxoplasmosis reagent performance in a high seroprevalence Latin American country. 2010;27-34. Google Scholar
- Murat JB, Dard C, Fricker Hidalgo H, Dardé ML, Brenier-Pinchart MP, Pelloux H. Comparison of the Vidas System and Two Recent Fully Automated Assays for Diagnosis and Follow-Up of Toxoplasmosis in Pregnant Women and Newborns. Clin Vaccine Immunol. août 2013;20(8):1203-12. PubMed | Google Scholar
- Schoonjans F. MedCalc´s Diagnostic test evaluation calculator. MedCalc. Accessed Dec 13, 2022.
- Ndamukong-Nyanga JL, Linda KF, Lucile STC, Dolly-Misper DT. The Severity of Malaria and Toxoplasmosis Co-Infections among Pregnant Women in Yaounde, Cameroon. JSRR. 2021;22-32.
- Nguemaïm NF, Takang WA, Dobgima WP, Guebidiang BM, Foumane P, Kamga FHL. Seroprevalence of Toxoplasma gondii infection and associated risk factors among pregnant women attending antenatal clinic at the Bamenda Regional Hospital, Cameroon. Af J Clin Exp Micro. 2020;21(2):123-31. Google Scholar
- Kamga Wouambo R, Lili Motué L, Petang Keutcha D. Séroprévalence de la toxoplasmose et facteurs de risque associés chez les femmes enceintes en zone semi-urbaine: cas du district de santé de cité verte | Cameroon Health Research Forum. 2017. Accessed Feb, 8, 2022.
- Uttah EC, Ajang R, Ogbeche J, Etta H, Etim L. Comparative Seroprevalence and Risk Factors of Toxoplasmosis among Three Subgroups in Nigeria. Journal of Natural Sciences Research. 2013. Google Scholar
- Ogouyèmi-Hounto A, Agbayahoun-Chokki F, Sissinto Savi de Tove Y, Biokou Bankole B, Adinsi de Souza V, Assogba M et al. Évaluation d´un test de diagnostic rapide pour le diagnostic de la toxoplasmose chez les femmes enceintes à Cotonou (Bénin). Bull Soc Pathol Exot. 2014;107(2):85-9. Google Scholar
- Gebremedhin EZ, Abebe AH, Tessema TS, Tullu KD, Medhin G, Vitale M et al. Seroepidemiology of Toxoplasma gondii infection in women of child-bearing age in central Ethiopia. BMC Infect Dis. 2013 Feb 26;13:101. PubMed | Google Scholar
- Njunda AL, Assob JC, Nsagha DS, Kamga HL, Nde PF, Yugah VC. Seroprevalence of Toxoplasma gondii infection among pregnant women in Cameroon. J Public Health Africa. 2011b;2(2):e24. PubMed | Google Scholar
- Sroka S, Bartelheimer N, Winter A, Heukelbach J, Ariza L, Ribeiro H et al. Prevalence and Risk Factors of Toxoplasmosis among Pregnant Women in Fortaleza, Northeastern Brazil. Am J Trop Med Hyg. 2010;83(3):528-33. PubMed | Google Scholar
- Hassaneina F, Shehata AI. Rapid Immunochromatographic Test (RDT) Versus ELISA Technique for Diagnosing Toxoplasmosis among Individuals with Mental Disabilities. IJIRD. 2018;7(6). Google Scholar
- Onosakponome EO, Wokem GN, Abah AE. Comparison of Elisa and Rapid Immunochromatographic Tests in Diagnosis of Toxoplasmosis in Port Harcourt, Nigeria. IJTDH. 2020;41(1):54-9. Google Scholar
- Gomez CA, Budvytyte LN, Press C, Zhou L, McLeod R, Maldonado Y et al. Evaluation of Three Point-of-Care Tests for Detection of Toxoplasma Immunoglobulin IgG and IgM in the United States: Proof of Concept and Challenges. Open Forum Infectious Diseases. 2018;5(10):ofy215. PubMed | Google Scholar
- Martin C, Morin O. Sérologie de la toxoplasmose: comparaison de deux techniques: microplaque Platelia® (Biorad) et automate Liaison® (Diasorin) étu de de l´avi dite des anticorps IgG anti-toxoplasmiques. Revue Francophone des Laboratoires. 2006;2006(386):71-6. Google Scholar
- Duffy KT, Wharton PJ, Johnson JD, New L, Holliman RE. Assessment of immunoglobulin-M immunosorbent agglutination assay (ISAGA) for detecting toxoplasma specific IgM. Journal of Clinical Pathology. 1989;42(12):1291-5. PubMed | Google Scholar
- Bamba S, Some DA, Chemla C, Geers R, Guiguemde TR, Villena I. Analyse sérologique de la toxoplasmose pergravidique: évaluation des risques et perspectives du dépistage prénatal au centre hospitalier universitaire de Bobo Dioulasso au Burkina Faso. Pan Afr Med J. 2012;12:43. Epub 2012 Jun 22. PubMed | Google Scholar











