Impact of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with gestational diabetes: a randomized controlled trial
Sihem Chahed, Latifa Lassouad, Maha Dardouri, Ali Mtiraoui, Amel Maaroufi, Hedi Khairi
Corresponding author: Sihem Chahed, Université de Sousse, Faculté de Médecine de Sousse, Laboratoire de Recherche LR12ES03 “Qualité des Soins et Management des Services de Santé Maternelle”, Sousse, Tunisia 
Received: 02 Mar 2022 - Accepted: 22 Oct 2022 - Published: 08 Nov 2022
Domain: Obstetrics and gynecology,Maternal and child health,Reproductive Health
Keywords: Gestational diabetes, pregnant women, patient education, midwifery care
©Sihem Chahed et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Sihem Chahed et al. Impact of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with gestational diabetes: a randomized controlled trial. Pan African Medical Journal. 2022;43:128. [doi: 10.11604/pamj.2022.43.128.34084]
Available online at: https://www.panafrican-med-journal.com//content/article/43/128/full
Research 
Impact of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with gestational diabetes: a randomized controlled trial
Impact of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with gestational diabetes: a randomized controlled trial
![]() Sihem Chahed1,2,&, Latifa Lassouad1,3,
Sihem Chahed1,2,&, Latifa Lassouad1,3, ![]() Maha Dardouri1, Ali Mtiraoui1, Amel Maaroufi4, Hedi Khairi1,3
Maha Dardouri1, Ali Mtiraoui1, Amel Maaroufi4, Hedi Khairi1,3
&Corresponding author
Introduction: patient education is a key component of positive pregnancy and childbearing experiences, especially in women with gestational diabetes (GDM). Scant studies assessed the impact of tailored self-care education on pregnancy outcomes in pregnant women with gestational diabetes mellitus (GDM). This study aimed to assess the effect of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with GDM during pregnancy and at birth.
Methods: this was a randomized controlled trial conducted in a university hospital in the centre of Tunisia, from October 2020 to May 2021. The intervention group (n=61) received a self-care education programme with the usual care plan for GDM, while the control group received only the usual care plan (n=60). This trial was registered in the Pan African Clinical Trials Registry under the registration number PACTR202106591503674.
Results: at baseline, there was no significant difference between groups in terms of sociodemographic and clinical characteristics. The findings showed that the intervention significantly reduced maternal and neonatal hospitalizations (p=0.000), caesarean section (p=0.002), preterm labour (p=0.002), macrosomia (p=0.000), foetal distress (p=0.001), newborn respiratory complication (p=0.01) and hypoglycaemia (p=0.000).
Conclusion: implementing a tailored-care education for pregnant women with GDM had a positive impact on mother and infant clinical outcomes. Midwives and endocrinologists should use this programme to reduce maternal and neonatal complications during and after pregnancy.
Gestational diabetes mellitus (GDM) is a severe but neglected threat to maternal and child health [1]. It is defined as any degree of hyperglycaemia that is recognized for the first time during pregnancy [2]. In 2019, it was estimated that 1 in 6 births worldwide was affected by GDM [1]. Most cases of hyperglycaemia in pregnancy were in low- and middle-income countries, due to limited access to maternal healthcare. A recent systematic review revealed that pregnant women in the Middle East and North Africa region are burdened with a substantial prevalence of GDM, particularly in the Gulf Cooperation Council and North African countries [3]. In Tunisia, it was estimated that 9.3% of pregnant women developed GDM [4].
This condition is associated with multiple adverse pregnancy outcomes. Women with GDM experience high blood pressure, large birth weight babies, obstructed labour, and caesarean section [1,5]. Infants of diabetic mothers are exposed to metabolic and hematologic disorders, respiratory distress, cardiac disorders, and neurologic impairment due to perinatal asphyxia and birth traumas [6]. Additionally, the mother and baby are both at subsequent high risk of type 2 diabetes [1]. Therefore, it is substantial for women with GDM to carefully control and monitor their glycemia with their healthcare provider to reduce the risk of complications.
It is established that an early diagnosis associated with an adapted treatment is not enough to guarantee an optimal GDM control [7,8]. There is a strong connection between screening, diagnosis, treatment, and education. Indeed, recent studies revealed that self-care education using information and communication technologies was effective for blood sugar control, GDM knowledge, and a healthy lifestyle [9-12]. Nevertheless, pregnant women with low socioeconomic status and from low- and middle-income countries often have limited access to e-health programmes [1]. Besides, evidence showed that healthcare professionals and family support are key elements to empower women with GDM. Therefore, it is crucial to implement face-to-face education programmes for this population. According to the literature, different interventions were carried out to manage GDM [9,13-18]. Most of them were information technology (IT) programmes such as mobile health using smartphone applications [18,19], educational digital optical disc (DVD) [11], and web-based programmes [9,17,18]. Few studies implemented self-care education programmes for women with GDM. An Iranian clinical trial reported that self-care education had a positive effect on 2-h postprandial plasma glucose, APGAR scores, and neonatal hospitalization [14]. Another Iranian trial revealed that self-care education for pregnant women with GDM reduced caesarean section and macrosomia [15].
An Egyptian quasi-experimental study showed that self-care education was effective for GDM knowledge, attitudes, and self-care behaviour, and significantly reduced preterm and caesarean section [16]. The impact of self-care education on significant neonatal outcomes such as neonatal hypoglycaemia, foetal distress, birth defect, respiratory complications, and intra-uterine foetal death was poorly addressed. Besides, scant information exists about the effect of this programme on blood glucose control before birth and GDM-related hospitalization in low-middle income countries, such as Tunisia. Therefore, this study aimed to assess the effectiveness of a tailored self-care education programme on maternal and neonatal outcomes in pregnant women with GDM during pregnancy and at birth.
The hypothesis was that women with GDM who received the tailored self-care educational programme will develop fewer maternal and neonatal complications during pregnancy and at birth in comparison with the control group who received usual care education.
Study design: this was a randomized controlled trial carried out in the Department of Endocrinology and the Department of Gynaecology at a University Hospital in the centre of Tunisia. The trial period was 8 months, from October 2020 to May 2021.
Study population: the target population was pregnant women with a diagnosed GDM by the obstetrician-gynaecologist in the first trimester, aged 19 to 44 years. Patients with chronic diabetes mellitus or other chronic diseases were not included. Participants were recruited from the endocrinology and gynaecology departments. The oral glucose tolerance test was used from 24 to 28 weeks for GDM screening in patients without any risk factors, such as advanced maternal age (≥35 years) [20], overweight or obesity, personal and family history of any form of diabetes [21]. In patients with risk factors of GDM, fasting blood glucose was used for GDM screening from the beginning of pregnancy up to 24 weeks.
Sample size: the sample size was calculated using the BiostaTGV with a power 1 - β = 0.8, two-tail α = 0.05, and a reduction with 0.6 in the mean of fasting blood glucose in the intervention group at follow-up. The required sample size was 72 pregnant women with GDM (36 women in each group). Considering a 20% of dropout and failure to consent, 50 participants in each group are needed.
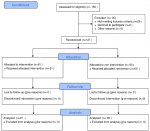
Recruitment procedure: Figure 1 shows the patients´ recruitment procedure. One hundred and fifty-six pregnant women who visited the gynaecology and the endocrinology departments in the first trimester of pregnancy were assessed for eligibility. Thirty-five women were excluded for the following reasons: 29 did not meet inclusion criteria, and 6 declined to participate. One hundred twenty-one were randomly allocated to the intervention and control groups by the principal investigator using sealed opaque envelopes. Each participant had an identification number (ID) (from 1 to 121) allocated by the research assistant. This latter submitted each ID in an individual sealed opaque envelope. The principal investigator randomly chooses from the envelopes to allocate the participant in the intervention or the control group. Using a coin, the first participant was allocated to the intervention group, the second to the control group, and the third to the intervention group, etc. The experimental group (n = 61) received an education programme according to a pre-established programme based on self-care with the usual care plan for GDM. The control group (n = 60) received only the usual care plan for GDM.
Tailored self-care education programme: the intervention aimed to enhance the self-capacity of women in GDM monitoring and management on daily basis through understanding the dietetic and hygiene measures. Self-care education was delivered by a multidisciplinary team composed of a midwife (principal investigator), an endocrinologist, a nurse, and a dietician. The intervention was carried out in the conference room of the Endocrinology Department of Farhat Hashed University Hospital for six consecutive days. The content was developed by the research team. It was based on international recommendations [1,22]. It included the pathophysiology of GDM and its complications, the technique of injecting insulin by syringe, the equipment and technique of self-monitoring by glucometer, and the nutrition and diet of patients with GDM. The nutritional and dietary model was based on the recommendations of the British Diabetic Association [22]. These recommendations included eating healthier carbohydrates like whole-grain bread, brown rice, vegetables (e.g. 1 sweet potato per day) 5 types of fruits a day (150ml of fruit juice, one handful of grapes, seven strawberries, two tinned pineapple rings), cutting down sugar and replacing it with low or zero-calorie sweeteners, perfecting portion sizes (2 heaped tablespoons of cooked rice, 3 tablespoons of breakfast cereal, 3 heaped tablespoons of boiled pasta or cooked noodles, 60g of reduced or low-fat cream cheese, 125g of low-sugar yogurt), understanding the glycaemic index, eating more fish (a portion of 140g per day). The utilization of the educational programme was described in detail in Table 1. On the first day, the physician and the midwife conducted an individual preliminary interview with participants to discuss their current health status, to present the programme, to define their objectives, and to identify their expectations.
The intervention group was divided into 8 subgroups of 6 to 8 women homogeneous in term of gestational age. This group division helped to build strong communication between participants in the same gestational age and to exchange their experiences. For example, group one was composed of women with 17 to 20 weeks of pregnancy (n=19). It was then divided into 2 subgroups of 6 and one subgroup of 7 participants.
Women in the intervention group self-monitor their blood glucose control daily at home using a glucometer provided by the research team. This glucometer helped women to test their blood glucose on daily basis. Basing on the test results, they can self-regulate their diet according to the recommendations provided during the education program. The participants received a brief reminder of the education programme during GDM consultation. The women were followed-up monthly during GDM consultations to ensure compliance with the education program using the daily booklet of GDM self-monitoring. In a glycaemic cycle using glucometer (test before meals and 2 hours after meals 3 times per day), if the fasting blood glucose was ≥5.3 mmol/L and/or the blood glucose 2 hours after meal was ≥6.7 mmol/L, this means that the woman did not respect the recommendations.
Usual care plan: at the Obstetrics and gynaecology Department of Farhat Hashed University Hospital, women with gestational diabetes systematically and regularly benefit from specialized prenatal care plans with a monitoring protocol that meets international standards, to optimize the management of the pregnancy.
This care plan consisted of: a first consultation with the endocrinologist for general information about healthy eating, diet, and insulin therapy technique if needed, as well as the aims and the benefits of the follow-up consultations; providing all patients with usual care in the high-risk pregnancy unit of the maternity and neonatal centre of the hospital; an obstetrical consultation every 15 days for all women with GDM from 32 weeks until hospitalization; hospitalization from 38 weeks allows closer monitoring of the patient and a recording of the foetal heart rate twice daily. Pre-aesthetic consultation is planned for hospitalized patients and corticosteroid therapy is prescribed when indicated. A concertation with the neonatologist is systematic before any decision of planned caesarean section. All new-borns were examined at birth by a paediatrician.
Data collection: sociodemographic data, family history with diabetes mellitus, personal history (GDM, fetal macrosomia, intrauterine fetal demise (IUFD), pregnancy toxemia, prematurity) and clinical information related to the current pregnancy and GDM were collected from patients´ records using a data collection sheet. The primary outcomes were the number of hospitalizations due to GDM, the number of hospitalization days, and blood glucose control. This later was recorded based on the number of blood glucose cycles during hospitalizations, fasting blood glucose, and hypoglycaemia. A glycemic cycle consists of repetitive blood glucose tests 6 times a day: before meals and 2 hours after meals 3 times per day.
The secondary outcomes were the birth term (premature ≤36 weeks, full-term ≥37 weeks), the birth mode (vaginal, caesarean section), and the neonatal outcomes at birth (foetal distress, Appearance, Pulse, Grimace, Activity and Respiration (APGAR) score at 1 and 5 minutes, birth weight, hospitalization, respiratory complications, glycemia, foetal infection, birth defect, and intra-uterine foetal death).
Statistical analysis: data were analysed using the IBM SPSS Statistics version 20.0. The normal distribution of continuous variables was assessed using the Kolmogorov-Smirnov test. If the p value was significant (p = 0.05) the hypothesis that the respective distribution is normal was rejected. Continuous variables were described as means and standard deviation. The Kolmogorov-Smirnov test showed that continuous variables in this study were not normally distributed. Therefore, we used Mann-Whitney to assess the difference between groups. Categorical variables (age group, socioeconomic status, family, and personal histories, etc.) were described as frequencies and percentages. To compare the difference between groups in terms of categorical variables, Chi square test was used. A significance level of 0.05 was considered.
Ethics: this study was approved by a local research ethics committee under the reference code CEFMS 79/2021. It was registered to the Pan African Clinical Trials Registry under the registration number PACTR202106591503674. Participants were informed and they signed written consent before their enrolment in the study. The obtained data were confidential and anonymous.
Table 2 shows that there was no significant difference between the intervention and the control groups at baseline in terms of sociodemographic and clinical data. In the intervention group, the mean term of the current pregnancy at GDM diagnosis was 15.82 ± 1.86 weeks of gestation, and in the control group, it was 15.41 ± 2.20. The mean of gesture in the intervention group was 2.51 ± 1.44 and in the control group 2.82 ± 1.58.
Comparison of primary outcomes during pregnancy follow-up: Table 3 shows that the number of hospitalizations due to GDM, the number of hospitalization days, and the number of blood glucose cycles during all hospitalizations were significantly higher in the control group in comparison with the intervention group (p= 0.000). At baseline, there was not a significant difference between groups in term of fasting blood glucose (p=0.07).
In the intervention group, the mean of fasting blood glucose was significantly reduced at follow-up (p=0.000). The rate of women with hypoglycaemia at admission and discharge was significantly higher in the control group than in the intervention group (respectively at admission: 36.7% and 9.8%, p=0.000; respectively at discharge: 21.7% and 8.2%, p=0.03).
Comparison of secondary outcomes after pregnancy follow-up: Table 4 reports that 3 women in the intervention group and 15 women in the control group had premature labour, with a significant difference (p=0.002). The rate of caesarean section in the control group was significantly higher than in the intervention group (p=0.002). Regarding new born health outcomes, there was a significant difference between groups in terms of foetal distress (p=0.001), APGAR score at 1 and 5 minutes (p=0.002; p=0.001, respectively), neonatal hospitalization (p=0.000), respiratory complication (p=0.01), and hypoglycaemia (p=0.000).
Education programmes for pregnant women with GDM aiming to strengthen self-care are needed [13]. The present study aimed to assess the effectiveness of a tailored-care education programme on maternal and neonatal outcomes in pregnant women with GDM. The findings showed that women who received a tailored-care education programme with a multidisciplinary team including a dietitian, a midwife, a nurse, and an endocrinologist had significantly a lower number of hospitalizations due to GDM, a lower rate of hypoglycaemia, premature labour, and caesarean section in comparison to the control group. Additionally, new-borns of women in the intervention group had significantly greater APGAR scores at 1 and 5 minutes, a normal birth weight, and less foetal distress, neonatal complications including neonatal hospitalization, respiratory complication, and hypoglycaemia in comparison to the control group.
The current findings are consistent with the results of the current literature. Mirghafourvand et al. found that self-care training on physical activity and nutrition reduced the rate of macrosomia and caesarean section in Iranian pregnant women with GDM [15]. Kolivand et al. reported that a self-care guide package delivered in face-to-face educational sessions had a positive effect on maternal self-efficacy and 2-h postprandial plasma glucose, APGAR scores, and neonatal hospitalization in Iranian pregnant women with GDM [14]. Ibrahim and Saber revealed that Egyptian pregnant women with GDM who received self-care education programmes had significantly better GDM knowledge, attitudes, and self-care behaviour and fewer neonatal complications including hypoglycaemia, macrosomia, and hospitalization at the neonatal intensive care unit immediately after birth [16]. In the same study, the authors found that a self-care education programme did not significantly reduce prematurity and respiratory distress. Likewise, Mirghafourvand et al. revealed that there were no significant differences between groups in terms of preterm labour [15].
What the current study adds is that a tailored-care education programme with a multidisciplinary team significantly reduced preterm labour, respiratory complications, foetal distress, neonatal hospitalization, macrosomia, and hypoglycaemia immediately after birth. However, there was no significant difference between the intervention and the control group in terms of foetal infection, birth defect, and intra-uterine foetal death. Regarding maternal complications, this intervention improved blood glucose control and reduced the rate of mother hospitalizations due to GDM, hypoglycaemia during pregnancy, vaginal specimen, glycosuria, and caesarean section in comparison to the control group.
Limitations: the present study had some limitations. First, double blinding was not possible to use. Second, this study was monocentric, which makes the results not generalizable. However, monocentric studies conserve the homogeneity of the sample. As well, it would be interesting if this study assessed the impact of tailored-care education on anxiety, depression, and stress in pregnant women with GDM.
Implications for practice: the current findings are beneficial for midwives and nurses practicing in women´s health centres. It is highly recommended to implement face-to-face and tailored-care education programmes for pregnant women with GDM from low- and middle-income countries. This fact can strengthen the connection between patients and healthcare providers and improve access to maternal and child healthcare centres. Tailored-care education programmes for pregnant women with GDM should be an urgent project of maternal and child health policy since it can improve healthcare coverage and reduce health inequalities in low- and middle-income countries. Besides, this intervention reduced hospitalization due to GDM, which helps to limit the overload of patients in the endocrinology department and the economic burden of GDM.
The present study revealed that a tailored-care education programme with a multidisciplinary team including a midwife, a nurse, a dietitian, and an endocrinologist reduced mother hospitalizations due to GDM, hypoglycaemia during pregnancy, preterm labour, caesarean section, macrosomia, and neonatal complications including neonatal hospitalization, respiratory complication, foetal distress, and hypoglycaemia. The need for making tailored-care education programmes and maternal and child healthcare policy for pregnant women with GDM in low- and middle-income countries should be considered. This fact can improve healthcare coverage and reduce health inequalities in low- and middle-income countries. Tailored-care education help to limit the overload of patients in the endocrinology department and the economic burden of GDM.
What is known about this topic
- Good antenatal care and patient education are crucial for a positive pregnancy and childbearing experience, particularly in pregnant women with gestational diabetes;
- Few studies implemented a tailored-care education programme and explore its effects on maternal and neonatal outcomes in low- and middle-income countries;
- Self-care education in pregnant women with gestational diabetes improved glycaemic control and APGAR scores and reduced caesarean section and neonatal hospitalizations.
What this study adds
- Implementing a tailed self-care education for pregnant women with GDM had a positive and broad impact on mother and new born clinical outcomes;
- For mothers, it reduced mother hospitalizations due to GDM, hypoglycaemia during pregnancy, preterm labour, caesarean section;
- For babies, this intervention reduced the rate of macrosomia, respiratory complication, foetal distress, and hypoglycaemia.
The authors declare no competing interests.
Conception and design: Sihem Chahed, Latifa Lassouad, Amel Maaroufi; acquisition of data: Sihem Chahed, Maha Dardouri; statistical analysis: Chahed Sihem, Latifa Lassouad, Maha Dardouri; analysis and interpretation of data: Sihem Chahed, Ali Mtiraoui, Hedi Khairi; drafting of the manuscript: Sihem Chahed, Latifa Lassouad, Maha Dardouri; critical revision: Latifa Lassouad, Amel Maaroufi, Ali Mtiraoui, Hedi Khairi. All the authors read and approved the final version of this manuscript.
Table 1: self-care education programme
Table 2: sociodemographic and clinical data of participants in the intervention and the control groups at baseline
Table 3: comparison of primary outcomes between groups at baseline and follow-up
Table 4: comparison of secondary outcomes between groups at follow-up
Figure 1: patients´ recruitment procedure
- International Diabetes Federation. Care and prevention: Gestational diabetes. 2020. Accessed 14th December, 2021.
- Dirar AM, Doupis J. Gestational diabetes from A to Z. World J Diabetes. 2017;8(12):489-511. PubMed | Google Scholar
- Al-Rifai RH, Abdo NM, Paulo MS, Saha S, Ahmed LA. Prevalence of Gestational Diabetes Mellitus in the Middle East and North Africa, 2000-2019: A Systematic Review, Meta-Analysis, and Meta-Regression. Front Endocrinol (Lausanne). 2021;12:668447. PubMed | Google Scholar
- Mallouli M, Derbel M, Ingrid A, Sahli J, Zedini C, Ajmi T et al. Associated outcomes to fetal macrosomia: effect of maternal diabetes. Tunis Med. 2017;95(2):120-125. PubMed | Google Scholar
- Centers for Disease Control and Prevention. Gestational Diabetes and Pregnancy. Updated July 14, 2022. Accessed 14th December, 2021.
- Mitanchez D, Yzydorczyk C, Simeoni U. What neonatal complications should the pediatrician be aware of in case of maternal gestational diabetes? World J Diabetes. 2015;6(5):734-43. PubMed | Google Scholar
- Carolan M. Women´s experiences of gestational diabetes self-management: a qualitative study. Midwifery. 2013;29(6):637-45. PubMed | Google Scholar
- Han S, Middleton P, Shepherd E, Van Ryswyk E, Crowther CA. Different types of dietary advice for women with gestational diabetes mellitus. Cochrane Database Syst Rev. 2017 Feb 25;2(2):CD009275. PubMed | Google Scholar
- Kim YS, Kim HS, Kim YL. Effects of a Web-Based Self-Management Programme on the Behavior and Blood Glucose Levels of Women with Gestational Diabetes Mellitus. Telemed J E Health. 2019;25(5):407-414. PubMed | Google Scholar
- Sayakhot P, Carolan-Olah M, Steele C. Use of a web-based educational intervention to improve knowledge of healthy diet and lifestyle in women with Gestational Diabetes Mellitus compared to standard clinic-based education. BMC Pregnancy Childbirth. 2016 Aug 5;16(1):208. PubMed | Google Scholar
- Draffin CR, Alderdice FA, McCance DR, Maresh M, Harper R, Patterson CC et al. Impact of an educational DVD on anxiety and glycaemic control in women diagnosed with gestational diabetes mellitus (GDM): A randomised controlled trial. Diabetes Res Clin Pract. 2017;126:164-171. PubMed | Google Scholar
- Yew TW, Chi C, Chan SY, van Dam RM, Whitton C, Lim CS et al. A Randomized Controlled Trial to Evaluate the Effects of a Smartphone Application-Based Lifestyle Coaching Programme on Gestational Weight Gain, Glycemic Control, and Maternal and Neonatal Outcomes in Women With Gestational Diabetes Mellitus: The SMART-GDM Study. Diabetes Care. 2021;44(2):456-463. PubMed | Google Scholar
- Park SJ, Lee J. The effects of health care programmes for gestational diabetes mellitus in South Korea: a systematic review. Korean J Women Health Nurs. 2020 Dec 31;26(4):274-284. PubMed | Google Scholar
- Kolivand M, Rahimi MA, Keramat A, Shariati M, Emamian MH. Effect of a new self-care guide package on maternal and neonatal outcomes in gestational diabetes: A randomized control trial. J Diabetes. 2019 Feb;11(2):139-147. PubMed | Google Scholar
- Mirghafourvand M, Zandinava H, Shafaei FS, Mohammad-Alizadeh-Charandabi S, Ghanbari-Homayi S. Effectiveness of self-care training on pregnancy consequences in gestational diabetes: a randomized controlled clinical trial. Shiraz E-Medical Journal. 2019 Jun 30;20(6). Google Scholar
- El Sayed Ibrahim R, Mousa Saber N. Impact of Self-care Programme for Gestational Diabetic Women on Pregnancy Outcomes. Am J Nurs Res. 2020;8(1):122-131. Google Scholar
- Ming WK, Ding W, Zhang CJP, Zhong L, Long Y, Li Z et al. The effect of exercise during pregnancy on gestational diabetes mellitus in normal-weight women: A systematic review and meta-analysis. BMC Pregnancy Childbirth. 2018 Nov 12;18(1):440. PubMed | Google Scholar
- Jeon YK, Kim HJ, Yang MY, Jung D, Young YK, Noh GO et al. Effects of a postnatal care programme on self-efficacy, self-management, and glycemic control in women with gestational diabetes mellitus. Korean J Women Heal Nurs. 2018;24(4):367-378. Google Scholar
- Huang F, Zhang S, Tian Y, Li L, Li Y, Chen X et al. Effect of mobile health based peripartum management of gestational diabetes mellitus on postpartum diabetes: A randomized controlled trial. Diabetes Res Clin Pract. 2021 May;175:108775. PubMed | Google Scholar
- Lao TT, Ho LF, Chan BC, Leung WC. Maternal Age and Prevalence of Gestational Diabetes Mellitus. Diabetes Care. 2006 Apr;29(4):948-9. PubMed | Google Scholar
- Plows JF, Stanley JL, Baker PN, Reynolds CM, Vickers MH. The Pathophysiology of Gestational Diabetes Mellitus. Int J Mol Sci. 2018 Oct 26;19(11):3342. PubMed | Google Scholar
- The British Diabetic Association. What can I eat with gestational diabetes? United Kingdom. Accessed 16th June, 2022.