Sexual maturation of HIV-infected and uninfected male children in Abakaliki, South-East, Nigeria: a cross-sectional study
Chijioke Ogodo Ogeh, Maria-Lauretta Orji, Kenechukwu Kosisochukwu Iloh, Chijioke Joel Nweke, Blessed Uzochukwuamaka Ogeh, Ogochukwu Nneka Iloh, Ifeoma Josephine Emodi
Corresponding author: Kenechukwu Kosisochukwu Iloh, Department of Paediatrics, University of Nigeria Teaching Hospital, Ituku-Ozalla, Enugu, Nigeria 
Received: 11 Apr 2021 - Accepted: 03 Jun 2022 - Published: 17 Jun 2022
Domain: Pediatrics (general)
Keywords: Sexual maturation, adolescents, males, HIV-infected
©Chijioke Ogodo Ogeh et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Chijioke Ogodo Ogeh et al. Sexual maturation of HIV-infected and uninfected male children in Abakaliki, South-East, Nigeria: a cross-sectional study. Pan African Medical Journal. 2022;42:133. [doi: 10.11604/pamj.2022.42.133.29305]
Available online at: https://www.panafrican-med-journal.com//content/article/42/133/full
Research 
Sexual maturation of HIV-infected and uninfected male children in Abakaliki, South-East, Nigeria: a cross-sectional study
Sexual maturation of HIV-infected and uninfected male children in Abakaliki, South-East, Nigeria: a cross-sectional study
Chijioke Ogodo Ogeh1, ![]() Maria-Lauretta Orji1, Kenechukwu Kosisochukwu Iloh2,&, Chijioke Joel Nweke3, Blessed Uzochukwuamaka Ogeh1,
Maria-Lauretta Orji1, Kenechukwu Kosisochukwu Iloh2,&, Chijioke Joel Nweke3, Blessed Uzochukwuamaka Ogeh1, ![]() Ogochukwu Nneka Iloh2, Ifeoma Josephine Emodi2
Ogochukwu Nneka Iloh2, Ifeoma Josephine Emodi2
&Corresponding author
Introduction: HIV infection, through various mechanisms causes a derangement in sexual maturation. This study compared the Marshal and Tanner staging of HIV-infected and uninfected males. The aim of the study was to determine the sexual maturation in male children infected with HIV on HAART in Abakaliki.
Methods: this was a cross-sectional and comparative study involving 80 HIV-infected boys aged 8-17 years and 80 uninfected counterparts matched for age and socio-economic class. Stages of sexual maturation (testicular size and pubic hair) were determined according to the method proposed by Marshall and Tanner. The testicular size was measured using an orchidometer. Data analysis was done with SPSS version 20. Structured questionnaire was used to collect information on socio-demographics.
Results: assessment of pubic hair development, showed that 45 (56.2%) of the subjects were in the pre-pubertal stage compared to 27 (33.8%) among the controls, this relationship was statistically significant (p=0.005, OR = 2.5, C.I=1.3-4.8). The mean testicular volume among subjects was found to be 8.29 ± 8.26mls compared to 11.57 ± 8.26mls found in controls. This relationship was also statistically significant. There were significant statistical relationships between duration on HAART and clinical stages of disease with both pubic hair development and testicular volume of subjects and controls.
Conclusion: HIV-infected males had significantly delayed onset and progression of sexual maturation. Routine assessment of the sexual maturation of HIV-infected children as well as addressing the modifiable variables influencing sexual maturity is recommended.
Amongst all the developmental changes that occur in adolescence, sexual maturation is the most significant in its influence on the behaviour of boys and girls [1]. The physical changes that occur during the development of secondary sexual characteristics were staged by Marshall and Tanner [2]. They devised a method of classifying the adolescent based on the level of sexual maturation into five stages [2]. This classification utilizes size of the scrotum and testes, and the penile shafts as well as the pattern of distribution and quantity of the pubic hair [2]. The age at initiation, sequence and period of puberty in males differ among persons and between children from different ethnic groups, countries, and races and also differ among developed and developing nations [3]. Normal initiation of sexual maturation in males may range from nine years to 14 years and may take an average of 1.5-6 years to be completed [3]. The earliest age at which the physical signs of puberty occurs is 8 years (SMR 2) in females and 9.5 years (SMR 2) in males [4]. Factors that influence sexual maturation could be hormonal, physical activity level, chromosomal, ethnic, constitutional, socio-economic status, nutritional and chronic illnesses like HIV [5-7]. Hormonal control of sexual maturation depends primarily on the activation of the pituitary gland with subsequent release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) [8]. HIV infection may affect metabolic and endocrine functions and thus alter hormonal systems involved in the control of growth and development [9]. Reported endocrine problems related to HIV infection in children include growth deceleration and delayed onset of puberty [10]. A perceived or actual delay in sexual maturation and biological development can lead to the development of poor body image and low self-esteem which may result in psychosocial problems such as eating disorders, depression, social withdrawal, isolation, declining academic performance, school avoidance, teasing and bullying by peers [11].
Assessment of sexual maturation using testicular size and pubic hair development in boys can be done by two methods, namely; the adolescent self-report and the physician assessment [12]. The disadvantage of adolescent self-report method is that a bias could occur resulting in over or under reporting. The self-report method is also easier to perform and subjects are not exposed to physical examinations [12]. In the physician assessment method, the physician physically examines the respondents to decide their maturation stage and grades them according to the criteria of Tanner [12]. This study therefore, aimed to compare the sexual maturation of perinatally HIV-infected males aged 8-17 years and their uninfected counterparts at the Alex Ekwueme Federal University Teaching Hospital and Mile Four Mission Hospital, Abakaliki, Ebonyi State, Nigeria.
The study was conducted at the Alex Ekwueme Federal University Teaching Hospital and Mile Four Mission Hospital, Abakaliki, Ebonyi State, Nigeria using physician assessment.
Study site: the study was carried out in the paediatric HIV clinics of both hospitals. The clinics run once a week in each hospital and has an average attendance of 20 and 15 patients respectively. A total of 200 children were registered in the paediatric HIV clinic, (105 males were aged between 8 and 17 years) as at August 2018 from both hospitals. The controls were recruited from the Children Outpatient Clinic (CHOP). This outpatient clinic runs daily, except on weekends with an average attendance of 350 patients per week.
Study design: this was a cross-sectional and comparative study. HIV-infected males aged 8-17 years who met the inclusion criteria were consecutively enrolled from the HIV clinic until the desired sample size was achieved.
Study population: the study subjects were HIV-infected males aged 8-17 years who were born to women with documented HIV-1 infection during pregnancy or at the time of delivery and in whom HIV infection had been diagnosed through detection of viral markers (DNA PCR) or the persistence of HIV-1 antibodies (investigated by means of enzyme linked immunosorbent assay) after 18 months of life. The controls were HIV-uninfected males who were matched for age and social class and without chronic diseases such as sickle cell anemia, chronic kidney disease or asthma attending the children outpatient clinic.
Sample size determination: the sample size required was determined using the method as shown:
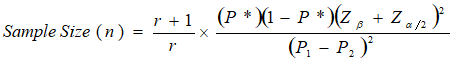
Where r = ratio of control to cases (subjects), 1 for equal number of cases and control.
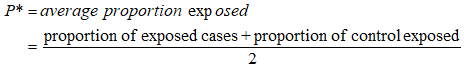
Fifty percent (50% (0.5)) was used when there was no recognizable estimate. Zβ = normal standard variate for significant level as identified in previous section. i.e normal standard variate for power of 80% which is equal to 0.84. Zα/2 = normal standard variate for significant level as identified in previous section i.e normal standard variate at 95% confidence level = 1.96. P1-P2 = different in proportions or effect size identified from previous studies. P1 = is proportion in cases (subjects) (0.17) [13]. P2 = is proportion in controls (0.04) [13]. n = minimum sample size.
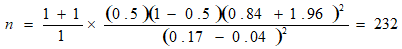
Furthermore, a second formula correcting the sample size for a finite population was used because the first formula was drawn from a finite population of HIV-infected children numbering less than 10,000 population, thus:
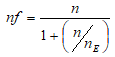
Where, nf = represent sample size for a finite population n = represent a population greater than 10,000 participants. nE = estimated total number of HIV-infected males aged 8-17 years in FETHA and Mile Four Hospital were approximately 105 (61+44). Hence;
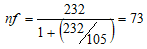
The minimum sample size was thus 73.
Non-responders rate
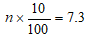
Therefore, the actual minimum sample size of cases (subjects) was 80. However, the sample size is allocated proportionally to the two hospitals under study thus: let N1 be the number of HIV patient aged 8-17 years in FETHA and N2 be the number of HIV patient aged 8-17 years mile four, n = minimum sample size = 80 and N = total number of HIV patient aged 8-17 years in the two hospital = 105. Therefore, the sample size for each hospital is given by: for FETHA;
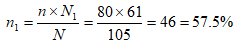
For mile four;
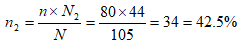
The same proportion was used for selection of the control.
Sampling method: perinatally HIV-infected males (subjects) who met the inclusion criteria were enrolled consecutively during the weekly paediatric HIV clinics. On enrollment, informed consent from the parents/caregivers and assent from the subjects were obtained by the researchers. Oyedeji social classification system was used to determine the social class of each participants. It was classified into high, medium and low based on the occupation and level of education of the parents. The social class is graded on a score of 1 to 5, where 1 being the highest and 5 being the lowest. Each score has two variables, occupation and level of education. The social class was further categorised into low (scores 4-5), middle (scores 2-3) and upper (scores 1-2) socio-economic class. Baseline World Health Organization (WHO) clinical staging and immunological classification of subjects on enrollment into the paediatric HIV clinic was obtained from the children´s case record forms. The WHO clinical staging was based on the WHO case definitions for HIV surveillance and clinical staging in children [14]. Standardized clinical parameters based solely on patients´ clinical features were used to delinate patients into different stages from stage 1 (asymptomatic) to stage 4 (AIDS) [14]. The HIV-uninfected males (controls) were enrolled from the Children Outpatient clinics after informed consent from caregivers and assent from the controls had been obtained by the researchers. The controls received HIV counseling and testing (HCT), according to national algorithm. The HIV negative controls, were subsequently enrolled and matched for age and social class with the subjects. A case record form was then completed for them by the researchers.
The subjects and the controls were examined by one of the researchers in a well-lit private room with a male chaperone. When private examination rooms were unavailable, privacy was ensured by using portable screening curtains to provide privacy for the participants. Tanner staging was identified, using the standardized method proposed by Marshall and Tanner for staging of sexual maturation [2]. The staging was done by examination of testicular volume and pubic hair growth. Pubic hair development was also staged from one to five. In the pre-pubertal stage 1, there was no pubic hair. Stage 2 was characterized by scanty, long and slightly pigmented hair while in stage 3, the hair is darker, starting to curl but in small amounts. In stage 4, pubic hair resembled adult pubic hair but less in quantity, coarse and curly and finally in stage 5, the pubic hair increases further in volume, spreads onto the medial thighs Estimation of testicular volume was done using an orchidometer. The right testis was first examined before the left testis for consistency and uniformity of data collection. Placing the participants in supine and cross-legged position, the testis was gently isolated. Then without squeezing the testis, scrotal skin was stretched. A manual side by side comparison between the testis and beads was made to establish the bead whose size matches that of the testis using a Prader orchidometer. Stage 1 is when testicular volume is less than 4 ml, stage 2 is 4-8 ml, stage 3 is 9-12 ml, stage 4 is 15-20 ml and stage 5 is when testicular volume is greater than 20 ml [13]. Puberty were classified as having started if they were at Tanner stage II or greater for pubic hair development [2]. Pubic hair development was classified as delayed puberty if pubic hair had not started by age 14 years [5,15]. All information gathered were documented in the case record form.
Ethical consideration: ethical approval was obtained from the Health Research and Ethics Committee of the Alex Ekwueme Federal University Teaching Hospital (FETHA/REC/VOL1/2017/597) and permission letter from Mile Four Mission Hospital (RE/M4H/30/18). The confidentiality of information and data retrieved from the study participants were emphasized to all study participants and were kept throughout and after the study. All the essential data relating to the survey were kept safe by the principal investigator. The study participants were assigned identification numbers and their data coded using their initials and identification numbers.
Data analysis: data were analyzed, using the Statistical Package for Social Sciences (SPSS) version 20 for windows (IBM Corp., Armonk, NY, USA). Descriptive statistics (frequencies, percentages, means and standard deviations) were generated for the variables. Means of continuous variables were compared, using the Student´s t-test, while effect of HIV status on testicular volume/pubic hair development were evaluated using logistic regression. All tests were two-tailed and conducted at 5% level of significance.
A total of 80 HIV-infected subjects and 80 HIV uninfected controls within the same age bracket of 8-17 years were enrolled for the study and matched for age and socio-economic class. Majority of the subjects and controls (70%) were pre/early adolescent (8-14 years) stage of life while 30% of the study participants were in middle adolescent (15-17 years). Forty-eight of the subjects (60%) and 48 controls (60%) respectively were in the lower socio-economic class while 32 (40%) were in the upper socio-economic class but none of the participants were from middle class. Table 1 is the clinical characteristics of the subjects. It shows that more of the subjects 64 (80%) were on first line of HAART. The table also depicts that 3 (3.75%) subjects were on HAART for less than 6 months while majority of them had been on these drugs for more than 5 years. In clinical staging of the subjects, 22 (27.5%) of them were in stage 3 while 1 (1.25%) subject was in stage 4. Additionally, 43 subjects (55.8%) had unsuppressed viral load while 34 (44.2%) had suppressed viral load. Three (3) subjects were on HAART for less than 6 months and were not classified as suppressed or unsuppressed viral load.
Secondary sexual characteristics of the study groups: the age distribution and testicular development of the study participant are shown in Table 2. Thirty-seven (46.25%) subjects compared to 7 (8.75%) controls were still in pre-pubertal stage while seventy-three (91.25%) controls compared to forty-three (53.75%) subjects were in pubertal stage of testicular development. The mean testicular volume for the subjects (8.288±8.259) was significantly lower than the controls (11.575±8.717) with p-value of 0.015. Table 3 depicts the age distribution and pubic hair development of the study participants. Forty-five (56.25%) of the subjects were in the pre-pubertal stage of pubic hair development, when compared with 27 (33.75%) of the controls, (p-value <0.001). For pubertal pubic hair development, 53 (66.25%) of the controls were in pubertal growth while only 35 (43.75%) of the subjects were pubic hair development with p-value of <0.001 as shown in Table 3.
Assessment of the pattern of secondary sexual maturation of the study groups: most of the subjects were in pre-pubertal stages for testicular development and pubic hair development [TSI=37 (46.25%) and PH1=45 (56.25%)] compared to the controls [TS1=7 (8.75%) and PHI=27 (33.75%)] as shown in Table 4. The controls were about twice more likely to attain puberty than subjects (p = 0.005, OR=2.524, CI=1.330-4.787). Controls were more likely to achieve puberty by testicular development than subjects with p-value <0.001, OR=8.973, CI=3.680-21.882.
Examining relationship between sexual maturation and some modifiable factors: Table 5 is the result of the logistic regression of sexual maturation (testicular size and pubic hair) on some modifiable factors (CD count, clinical stages and duration on HAART). The result showed a significant relationship between sexual maturation and clinical stages. It further showed an odd ratio of less than 1 in all the categories of the clinical stages, indicating a better sexual maturation in each of those stages compare to the reference category (stage IV). Further, the result showed a significant relationship between pubic hair growth with duration on HAART.
HIV infection was noted to negatively affect the total number of males that attained sexual maturation in the index study. More of the subjects were in pre-pubertal stage of testicular development 37 (46.25%) than controls 7 (8.75%). In subsequent testicular development (TS II), (21.25%) of the subjects were in stage II compared with controls (47.5%) on the same stage. Therefore, HIV uninfected controls were observed to develop faster from testicular stage II to stage V more than the HIV-infected subjects. This implies that the subjects were more likely to be in the pre-pubertal stage using testicular size development, which leads to delayed pubertal development in the subjects. This may be due to disruption of the gonadotropin-releasing hormone axis. Ebling FJ and Ojeda SR et al. [16,17], observed that pulsatile secretion of GnRH helps in the pituitary secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) in males, which helps in testicular development and spermatogenesis respectively. This pathway is disrupted in HIV infection and subsequently affects pubertal development. Puberty begins with testicular enlargement to greater than 2.5cm in length or greater than 4mls in volume initiated by pulsatile secretion of GnRH from its neuron and therefore delay in pubertal development is often a function of testicular development. A study by Majaliwa ES et al. [10] found that there is delay in pubertal development in prenatally HIV-infected children. This study agrees with the findings by Majaliwa ES et al. [10].
However, Kessler M et al. [18] found no difference in testicular development between subject and control. Also, the age of onset of tanner stage 4 was similar in both groups in their study. Furthermore, in this study, more subjects were also noted to be in the pre-pubertal stage of pubic hair development 45 (56.25%) than the controls 27 (33.75%). This is to be expected since in majority of adolescents testicular development preceded pubic hair. Therefore, delay in testicular development will likely lead to delay in development of pubic hair. Williams PL et al. [19] noted that for both subjects and controls testicular development preceded pubic hair development. These findings imply that HIV infection is associated with delayed initiation and subsequent progression through pubertal stages of testicular and pubic hair development.
Similar findings were found on the pubic hair development indicating that subjects were more likely to remain in the pre/early adolescent period than the controls. The difference in age compared favorably with the findings made by Mbwile GR [20] in Tanzania and Williams PL et al. [19] in the United States. The explanation for the age difference in HIV-infected versus HIV uninfected children may be due to the general effects of chronic illness as demonstrated by Pozo J and Argente J [21]. The study by Buchacz K et al. [11] corroborated well with this study and showed that there was delayed onset of pubertal development in children and adolescent with HIV infection. In addition, the studies by Buchacz K et al., Mbwile GR, Pozo J & Argente J and Iloh ON et al. [11,20-22] found that HIV infection affected the onset of puberty and progression into different pubertal stages. This suggests that HIV infection is associated with delayed onset and progression through the pubertal stages of testicular size and pubic hair development in HIV-infected children and adolescents. HIV-infected males were also observed to be less likely to reach PH2-PH5 which indicates the delaying progression to full sexual maturity and initiation than the HIV uninfected controls. These observations further reiterate the fact that HIV infection may lead to delay in both the timing of initiation of puberty and the progression through other subsequent pubertal stages [4]. Similar findings were documented by Mbwile GR [20] and Szubert AJ et al. [23] in Africa and de Martino M et al. [24] in Italy. These buttress the fact that HIV infection probably because of the chronicity of the disease and opportunistic infections (OIs) that they are prone to may negatively affects the age of pubertal development.
There was significant relationship between CD4 count and development of pubic hair. This suggests that subjects in advanced or in a severe immunologically depressed stage may have delay in attainment of puberty in HIV-infected children. The reason for the observation may be because subjects who have low CD4 count are prone to opportunistic infection which may lead to poor growth. Bellavia A et al. [25] observed that there was delay in sexual maturation in HIV-infected children which resulted from poor growth. The observation of delayed pubic hair development in subjects who have low CD4 count is supported by de Martino M et al. [24] and Buchacz K et al. [11] who in their studies noted that delayed puberty is more common in children with low CD4 count.
The logistic regression analysis of the present study showed that clinical stages was significantly related to the sexual maturation. This may be explained by the facts that opportunistic infections may affect the pubertal development and sexual maturation. Furthermore, there was a positive significant relationship between duration on HAART and sexual maturation. This may be explained by the facts that ARV drugs may lead to restoration of immunity and subsequently improvement in sexual maturation. The study by Mbwile GR [20] supported this finding and noted that ARVs helps in improvement in sexual development and maturation. Szubert AJ et al. [23] observed that early initiation of HAART will lead to males attaining sexual maturity in earlier age than those who started ARV drugs at an older age. Williams PL et al. [19] also supported the fact that ART may result in more normal pubertal onset and development.
There was a significant delay in both the age at onset and progression of sexual maturation in HIV-infected subjects compared to their HIV-uninfected counterparts.
Limitation: the study was a case-control hospital-based study with only one assessment of physical growth and sexual maturation. A prospective cohort study with multiple interval assessments would have given more information on the age at onset of testicular enlargement and pubic hair development as well as the subsequent pubertal development of HIV-infected males.
What is known about this topic
- Adolescents infected vertically by HIV are at higher risk of developmental impairment, growth alteration, wasting, delayed- puberty and impaired neuro-cognitive function;
- An actual pubertal delay in sexual maturation in adolescent can lead to the development of poor body image and low self-esteem which may result in psychosocial problems like school avoidance, poor academic achievement, isolation, eating disorders, bullying/teasing by peers, depression, and social withdrawal.
What this study adds
- This study affirms that there is a significant delay in both the age at onset and progression of sexual maturation in HIV-infected subjects compared to their HIV-uninfected counterparts;
- There was an association between HIV disease severity and delay in sexual maturation.
The authors declare no competing interests.
All the authors have read and agreed to the final manuscript.
We acknowledge all the adolescents who participated in the study.
Table 1: clinical characteristics of the subjects
Table 2: age distribution and testicular size development of the study participants
Table 3: age distribution and pubic hair development of the study participants
Table 4: sexual maturation of the study participants
Table 5: regression analysis of sexual maturation on social class, immunological stage, clinical stage and duration on HAART
- Gross RT, Duke PM. The effect of early versus late physical maturation on adolescent behaviour. Paediat clin North AM. 1980;27(1):71-77. PubMed | Google Scholar
- Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Arch Dis Child. 1969;44(235):291-303. PubMed | Google Scholar
- Nwokocha, ARC. Adolescence and associated problems. In: Azubuike JC, Nkanginieme KEO, editors. Pediatrics and child health in a tropical region. 2nd ed. Owerri, 2007:91-100.
- Cromer B. Adolescence, physical and social development. In: Kliegman RM, Behrman RE, Schor NF, Stanton BF, St Geme JW (eds). Nelson Textbook of Paediatrics.19th edition. Philadelphia. Elsevier Saunders. 2011:649-654.
- Bikle DD, Wang Y. Insulin like growth factor-I: a critical mediator of the skeletal response to parathyroid hormone. Curr Mol Pharmacol. 2012;5(2):135-142. PubMed | Google Scholar
- Gajdos ZK, Henderson KD, Hirschhorn JN, Palmert MR. Genetic determinants of pubertal timing in the general population. Mol Cell Endocrinol. 2010;324(1-2):21-29. PubMed | Google Scholar
- Rogol AD. Sex steroids, growth hormone, leptin and the pubertal growth spurt. Endocr Dev. 2010;17:77-85. PubMed | Google Scholar
- Mamatha ML, Merchant RH, Muntaz S. Clinical manifestations of HIV infections in children In: Mamatha ML, Merchant RH (eds) Principles of perinatal and pediatric HIV/AIDS. Ist edition, Mumbai Jaypee Brothers. 2011;71-84.
- Richmond EJ, Rogol AD. Male pubertal development and the role of androgen therapy. Nat Clin Pract Endocrinol Metab. 2007;3(4):338-44. PubMed | Google Scholar
- Majaliwa ES, Mohn A, Chiarelli F. Growth and puberty in children with human immunodeficiency virus infection. J Endocrinol Invest. 2009;32(1):85-90. PubMed | Google Scholar
- Buchacz K, Rogol AD, Lindsey JC, Wilson CM, Hughes MD, Seage GR 3rd et al. Pediatric AIDS Clinical Trials Group 219 Study Team. Delayed onset of pubertal development in children and adolescents with perinatally acquired HIV infection. J Acquir Immune Defic Syndr. 2003;33(1):56-65. PubMed | Google Scholar
- Azevedo JC, Brasil LM, Macedo TB, Pedrosa LF, Arrais RF. Comparison between objective assessment and self-assessment of sexual maturation in children and adolescents. J Pediatr (Rio J). 2009;85(2):135-42. PubMed | Google Scholar
- Emmanuel M, Bokor BR. Tanner Stages. In: StatPearls. Treasure Island (FL): StatPearls Publishing. Jan 2021.
- Tindyebwa D, Kayita J, Musoke P, Eley B, Nduati R, Coovadia H et al. Handbook on Paediatric AIDS in Africa. Africa network for the care of children affected by AIDS. 2nd edition. Uganda: Family Health International. 2001;3-26.
- Rosen DS, Foster C. Delayed puberty. Pediatr Rev. 2001 Sep;22(9):309-15. PubMed
- Ebling FJ. The neuroendocrine timing of puberty. Reproduction. 2005;129(6):675-83. PubMed | Google Scholar
- Ojeda SR, Lomniczi A. Puberty in 2013: unravelling the mystery of puberty. Nat Rev Endocrinol. 2014;10(2):67-9. PubMed | Google Scholar
- Kessler M, Kaul A, Santos-Malavé C, Borkowsky W, Kessler J, Shah B. Growth patterns in pubertal HIV-infected adolescents and their correlation with cytokines, IGF-1, IGFBP-1, and IGFBP-3. J Pediatr Endocrinol Metab. 2013;26(7-8):639-44. PubMed | Google Scholar
- Williams PL, Abzug MJ, Jacobson DL, Wang J, Van Dyke RB, Hazra R et al. Pubertal onset in children with perinatal HIV infection in the era of combination antiretroviral treatment. AIDS. 2013 Jul 31;27(12):1959-70. PubMed
- Mbwile GR. Growth and pubertal development among children aged 8-18 years in Dar es Salaam. Dissertation. Muhimbili University of Health and Allied Sciences. 2012.
- Pozo J, Argente J. Delayed puberty in chronic illness. Best Pract Res Clin Endocrinol Metab. 2002 Mar;16(1):73-90. PubMed | Google Scholar
- Iloh ON, Iloh KK, Ubesie AC, Emodi IJ, Ikefuna AN, Ibeziako NS. Comparison of Tanner staging of HIV-infected and uninfected girls at the University of Nigeria Teaching Hospital, Ituku/Ozalla, Enugu, Nigeria. J Pediatr Endocrinol Metab. 2017 Jul 26;30(7):725-729. PubMed | Google Scholar
- Szubert AJ, Musiime V, Bwakura-Dangarembizi M, Nahirya-Ntege P, Kekitiinwa A, Gibb DM et al. Pubertal development in HIV-infected African children on first-line antiretroviral therapy. AIDS. 2015 Mar 13;29(5):609-18. PubMed | Google Scholar
- de Martino M, Tovo PA, Galli L, Gabiano C, Chiarelli F, Zappa M. Puberty in perinatal HIV 1 infection: a multicenter longitudinal study of 212 children. AIDS 2001;15(12):1527-34. PubMed | Google Scholar
- Bellavia A, Williams PL, Di Meglio LA, Hazra R, Abzug MJ, Patel K et al. Delay in sexual maturation in perinatally HIV-infected youths is mediated by poor growth. International Maternal Pediatric Adolescent AIDS Clinical Trials (IMPAACT) P219/219C study, and the pediatric HIV/AIDS cohort study (PHACS). AIDS. 2017;31(9):1333-41. PubMed | Google Scholar











