Prevalence and correlates of hypothyroidism in pregnancy: a cross-sectional study at Bouget General Hospital, Ivory Coast
Valéry Katché Adoueni, Auguste Jean-Claude Azoh, Ethmonia Kouame, David Guanga Meless, Pascal Sibailly, Augustin Koudou Derbe, Marie-Chantal N´Guessan, Koffi Benjamin Dzade, Simplice Koffi, Théodore Kouakou, Lydie Viviane Arra, Yolande Ouattara
Corresponding author: Auguste Jean-Claude Azoh, Programme National de Lutte contre les Maladies Métaboliques et de Prévention des Maladies non Transmissibles (PNLMM /PMNT) Ministère de la Santé et de l'Hygiène Publique de la République de Côte d'Ivoire (MSHP), Abidjan, Côte d'Ivoire 
Received: 24 Nov 2021 - Accepted: 03 Jan 2022 - Published: 13 Jan 2022
Domain: Endocrinology
Keywords: Thyroid disorders, hypothyroidism, Black women, pregnancy
©Valéry Katché Adoueni et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Valéry Katché Adoueni et al. Prevalence and correlates of hypothyroidism in pregnancy: a cross-sectional study at Bouget General Hospital, Ivory Coast. Pan African Medical Journal. 2022;41:37. [doi: 10.11604/pamj.2022.41.37.32553]
Available online at: https://www.panafrican-med-journal.com//content/article/41/37/full
Research 
Prevalence and correlates of hypothyroidism in pregnancy: a cross-sectional study at Bouget General Hospital, Ivory Coast
Prevalence and correlates of hypothyroidism in pregnancy: a cross-sectional study at Bouget General Hospital, Ivory Coast
![]() Valéry Katché Adoueni1, Auguste Jean-Claude Azoh1,&, Ethmonia Kouame1, David Guanga Meless2,3, Pascal Sibailly1, Augustin Koudou Derbe1, Marie-Chantal N´Guessan1, Koffi Benjamin Dzade1, Simplice Koffi1, Théodore Kouakou1, Lydie Viviane Arra1, Yolande Ouattara1
Valéry Katché Adoueni1, Auguste Jean-Claude Azoh1,&, Ethmonia Kouame1, David Guanga Meless2,3, Pascal Sibailly1, Augustin Koudou Derbe1, Marie-Chantal N´Guessan1, Koffi Benjamin Dzade1, Simplice Koffi1, Théodore Kouakou1, Lydie Viviane Arra1, Yolande Ouattara1
&Corresponding author
Introduction: several adverse pregnancy outcomes have been reported in gestations with associated hypothyroidism. The prevalence of hypothyroidism has not been frequently reported in Black Africans. This study sorts to report the prevalence and associated factors of hypothyroidism in black African pregnant women.
Methods: this was a hospital-based cross-sectional study, including all pregnant women attending the gynecologic unit of Bouget General Hospital Abidjan. Serum thyroid-stimulating hormone and T4 were obtained from all participants and analyzed using a fluorescent Immunochemistry assay. Data were analyzed using R version 4.05. Univariable and multivariable logistic regression was used to assess factors associated with hypothyroidism and statistical significance considered as p < 0.05.
Results: overall there were 693 participants, mean age of 28.1(SD 6.4) years with an average gestational age of 24.1 (SD 8) weeks, and a majority of study participants were in the second trimester of gestation. The prevalence of hypothyroidism was 12.1% (n = 84) (10.8% subclinical hypothyroidism and 1.3% clinical hypothyroidism) whereas 1.9% (n = 13) had hyperthyroidism. In addition, patients with reported type 1 diabetes mellitus had an increased risk of hypothyroidism (aOR: 12.6, 95% CI 1.9-100.8; p ≤ 0.01).
Conclusion: this study revealed a high prevalence of hypothyroidism, though mostly in the subclinical form. Further research is warranted to confirm these findings which may have implications on early screening of hypothyroidism in Black African women.
Thyroid disorders are of relevance in public health and rank second to diabetes as the most common endocrinological disorder globally [1, 2]. Adverse maternal and fetal outcomes are reported in gestations with associated hypothyroidism [3-5]. It is thus recommended to screen for thyroid disorders during gestation [1, 6]. In addition, pregnancy is marked by several changes including physiological changes in the thyroid gland [7]. The first trimester of pregnancy is accommodated by high levels of human chorionic gonadotropin (HCG) levels which contains a similar alpha (α) unit as thyrotropin stimulating hormone (TSH), thus acting on the TSH receptors, leading to a significant decrease in TSH levels during the early phase of pregnancy [7]. However, higher levels of TSH are reported in the later phase of pregnancy due to the progressive decline of HCG levels during gestation [8, 9]. The fetal thyroid gland has been reported to be underproductive in the early half of pregnancy thus there is fetal dependence on adequate levels of maternal thyroid hormones for proper fetal development [5].
The prevalence of hypothyroidism in pregnancy varies across different ethnic groups [10]. In a low to middle-income setting, the prevalence is reported between 5 - 18% [11-13]. Although subclinical hypothyroidism is more prevalent than overt hypothyroidism in pregnancy, studies still report poor neurologic development in children born to mothers with any form of hypothyroidism. In sub-Saharan Africa and specifically the Ivory Coast and to our knowledge, data is scarce on the prevalence of hypothyroidism in pregnancy. Abdulslam et al. reported a prevalence of 18% in a group of hypertensive pregnant women in Nigeria [12]. Given the dearth of available literature on hypothyroidism amongst pregnant women in Africa, the study sorts to describe the prevalence and features of hypothyroidism amongst pregnant women in the Ivory Coast.
Study design and study setting: this was a hospital-based descriptive cross-sectional study conducted from the 6th to the 21st of February 2019 at Port-Bouët General Hospital in Abidjan is the most populated city in the Ivory Coast with a population of 5,355,000 inhabitants. The Port-Bouët General Hospital, located in the commune of the same name in the southeast of the city of Abidjan, is a level 1 reference hospital in the health system pyramid. It receives a population of 75505 people including 13000 pregnant women each year. This hospital provides curative and preventive health care and has several departments, including one dedicated to the consultation of pregnant women. The prenatal consultation service is run by six midwives working under the supervision of gynecologists and obstetricians. Approximately 50 patients receive care here every day.
Study participants: eligible study participants were all pregnant women attending the gynecologic unit of the study site. All participants who consented were included in the study. Non-consenting patients were excluded. We calculated a minimum sample size of 55 participants at a 95% confidence interval using the Lorentz formula, assuming a prevalence of hypothyroidism of 16% as reported by Abdulslam et al. in Nigeria [12].
Study variables: a team of investigators was trained on how to fill in the structured case report form on an electronic tablet. Baseline characteristics including age, parity and gravidity, height, weight, body mass index, and resting blood pressure were collected. A blood sample was drawn from consenting participants and stored. Fluorescent immunochemistry assay mainly Cobas E411 instrument from Roche Diagnostics was used to quantify the level of TSH for all study participants. Thyroid status for all participants was based on the guidelines of the American Thyroid Association (ATA) for diagnosing thyroid diseases in pregnancy [1]. Free T4 levels were assessed for all patients presenting with hypothyroidism.
Definitions: forms of hypothyroidism were considered in two stages: subclinical hypothyroidism for TSH between 2.5 and 10 mIU/L and normal free T4 12-22 pmol/L in the first trimester and TSH between 3 and 10 mIU/L and free T4 12-22 pmol/L during the second and third trimester; Clinical hypothyroidism when TSH was between 2.5 and 10mIU/L at first trimester and between 3 and 10mIU/L at the second and third trimester, and free T4 < 12 pmol/L; Hyperthyroidism: TSH < 0.1 mIU/L.
Statistical analysis: data were analyzed using R version 4.05, categorical variables are presented as frequency and percentages, and continuous variables as mean and standard deviation. The Chi-squared test was used to compare categorical variables, whereas the independent student T-test was used to compare continuous variables. Univariable and multivariable logistic regression models were used to evaluate patient characteristics associated with hypothyroidism in pregnancy. The selection of variables included in the model was based on current literature on prevalence and risk factors of hypothyroidism in pregnancy. All variables with a p-value of less than 0.2 in the univariable model were included in the multivariable model. Statistical significance was considered as p < 0.05.
Ethical consideration: the study protocol was approved by the ethical review boards of the National Ethics and Research Committee of Côte d'Ivoire (CNER-CI) and conducted in compliance with principles laid down in the Declaration of Helsinki and as well as with Côte d'Ivoire´s national ethics principles and regulations. All participants gave their informed consent before joining the study.
General characteristics
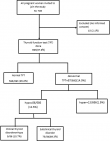
Of 708 pregnant women eligible for study inclusion, 2.1% (n = 15) were excluded due to refusal to participate (Figure 1). The remaining 693 participants were included in this study. Their age ranged from 12 to 45 years, with a mean age of 28.1 (SD 6.4) years. The mean age was comparable between patients with hypothyroid, euthyroid, and hyperthyroid presentations. The mean gestational age was 24.1 weeks and a majority of the study participants (407/693) were in the second trimester of gestation. More than half of the women were multigravida (55%). Previous miscarriage was found in 30.20% (n = 209) of participants. Also, 7.0% (n = 49) of study participants resided in goiter endemic zones and very few reported current type 1 diabetes mellitus. Table 1 represents the general characteristics of study participants.
Prevalence and correlates of hypothyroidism in pregnancy
The mean serum TSH was 1.80 (SD 1.02) mIU/L with a linear increase in mean TSH from the first through the third trimester (1.29, 1.81, 2.05 respectively). Of the 693 participants, 84 (12.1%) had hypothyroidism (Figure 1). In addition, subclinical hypothyroidism was the most represented form of hypothyroidism (75/84, 89.3%). There was no statistically significant difference in the presentation of symptoms of hypothyroidism (weight gain, constipation, cold intolerance, muscle cramps) between patients with or without hypothyroidism. Also, there was a slight increase in crude odds (OR: 1.3, 95% CI 0.7-2.6; p = 0.5) of hypothyroidism amongst participants in the third trimester of gestation though not significant. After adjusting for age and period of gestation, patients with a history of type 1 diabetes mellitus had an increased association with hypothyroidism (aOR: 12.6, 95% CI 1.9-100.8; p = < 0.01) (Table 2).
Several adverse outcomes have been described in pregnancies with associated hypothyroidism amongst which poor fetal neurological development [4, 14]. In this study, we aimed to determine the prevalence and associated factors of hypothyroidism in pregnancy. The prevalence of hypothyroidism in this study was 12.1% and there was a strong association with hypothyroidism in participants with concomitant type 1 diabetes mellitus. These results portray the burden of hypothyroidism in pregnancy and highlight the importance of further research, which may have some implications, including an early screening of hypothyroidism in pregnancy in an African context to prevent poor maternal and fetal outcomes during pregnancy and after delivery.
The prevalence of hypothyroidism in our study population is similar to the reports from other low to middle-income settings [13, 15-17]. A higher prevalence (16%) was however reported in Nigeria by Abdulslam K et al. [12]. This difference in prevalence might be explained by different populations and setting as well as the difference in TSH cutoffs. But it should be noted that more than half of the participants in the Nigerian study had associated hypertension contrary to 11% of participants in this study with systolic blood pressure above 140 mmHg. Contrarily, a study in India reported a lower prevalence (6.3%) of hypothyroidism [18]. This discrepancy is explained by the higher trimester-dependent cutoffs of TSH (at least 5IU/ml) used in the Indian study. On the contrary, a lower prevalence of hypothyroidism in pregnancy has been reported in high-income settings [19, 20] which is explained by differences in cutoff values used for diagnosis of hypothyroidism and secondly by the geographical differences in presentation of thyroid diseases [10]. In our study, there was no statistically significant difference in symptoms between those with or without hypothyroidism. Similar findings have been reported by Goel P et al. in India [18]. However, it should be noted that thyroid disorders can be hard to diagnose more precisely in pregnancy due to physiological rising levels of TSH but also because pregnancy-related symptoms due to high human chorionic gonadotropin and estrogen can overlap with symptoms of hyperthyroidism.
In our study, multivariable analysis showed that patients with a reported history of type 1 diabetes mellitus had an increased risk of hypothyroidism. This finding has been previously reported by Shahbazian et al. who demonstrated in their study that the risk of thyroid dysfunction was more common in participants with pre-gestational or gestational diabetes [21]. Potential explanations include the fact that type 1 diabetes is an autoimmune disorder, patients with type 1 diabetes mellitus could also present with concomitant or cross-reacting autoimmune activity in other endocrine glands including the thyroid [22]. Although the sample for our study was non randomly selected women attending a reference hospital and therefore not nationally representative of African Black pregnant women, and which may therefore present some biases, the potential beneficial impact of this study as first published data cannot be overemphasized. Our findings will serve as a base to design further studies in other settings to better precise the burden of thyroid disorders in African pregnant Black women.
This study revealed a higher prevalence of hypothyroidism in Black African pregnant women compared to that reported in Caucasians. These results raise awareness of the increased burden of this endocrine disorder and thus substantiate existing literature on the importance of regular screening of thyroid disorders in pregnancy in Africa, consequently avoiding certain adverse pregnancy outcomes.
What is known about this topic
- Adverse maternal and fetal outcomes are reported in gestations with associated hypothyroidism;
- Hypothyroidism might be prevalent in African pregnant women but has not been well established.
What this study adds
- The prevalence of hypothyroidism in African pregnant women is high, up to 12.1%;
- Most of these African pregnant women with hypothyroidism present with subclinical forms of the disease;
- Screening of hypothyroidism in Black African women might be relevant to reduce associated adverse maternal and fetal outcomes.
The authors declare no competing interests.
Valéry Katché Adoueni, Auguste Jean-Claude Azoh, and Ethmonia Kouame conceived the study and drafted the protocol. David Guanga Meless, Pascal Sibailly, Augustin Koudou Derbe, Marie-Chantal N´Guessan, and Koffi Benjamin Dzade collected the data. Valéry Katché Adoueni, Auguste Jean-Claude Azoh, and Yolande Ouattara did the statistical analysis, interpreted the results, and drafted the initial version of the manuscript. All authors read and approved the final version of the manuscript.
Our sincere thanks go to: Dr. Cissé, Director of the Port-Bouët General Hospital for his commitment to the smooth running of the survey. Dr Chenal and his collaborators at Laboratoire Chenal. Dr. Meless David for his availability and professionalism, and to all the staff of the gynaecologic-obstetrics department of the Port-Bouët General Hospital for their assistance and sound advice to the investigators, and all investigators for their commitment to the study. We also thank the staff of the PNLMM/PMNT. Prof Anastase Dzudie and the Clinical Research Education, Networking and Consultancy (CRENC) team for their support all through this publication.
Table 1: general characteristics of study participants
Table 2: factors associated with hypothyroidism
Figure 1: flow chart of the study participants
- Alexander EK, Pearce EN, Brent GA, Brown RS, Chen H, Dosiou C et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease during Pregnancy and the Postpartum. Thyroid. Mary Ann Liebert Inc. 2017 Mar;27(3):315-389. PubMed | Google Scholar
- Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99:39-51. PubMed | Google Scholar
- Mahadik K, Choudhary P, Roy PK. Study of thyroid function in pregnancy, its feto-maternal outcome; a prospective observational study. BMC Pregnancy Childbirth. BMC Pregnancy and Childbirth. 2020 Dec 10;20(1):769. PubMed | Google Scholar
- Haddow JE, Palomaki GE, Allan WC, Williams JR, Knight GJ, Gagnon J et al. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med. 1999 Aug 19;341(8):549-55. PubMed | Google Scholar
- Moog NK, Entringer S, Heim C, Wadhwa PD, Kathmann N, Buss C. Influence of maternal thyroid hormones during gestation on fetal brain development. Neuroscience. 2017 Feb 7;342:68-100. PubMed | Google Scholar
- Spencer L, Bubner T, Bain E, Middleton P. Screening and subsequent management for thyroid dysfunction pre-pregnancy, during pregnancy and in the immediate postpartum period. Cochrane Database Syst Rev. 2015 Sep 21;(9):CD011263. PubMed | Google Scholar
- Cignini P, Cafà EV, Giorlandino C, Capriglione S, Spata A, Dugo N. Thyroid physiology and common diseases in pregnancy: review of literature. J Prenat Med. 2012 Oct;6(4):64-71. PubMed | Google Scholar
- Zubair Baloch, Pierre Carayon, Bernard Conte-Devolx, Laurence Demers M, Ulla Feldt-Rasmussen, Jean Francois Henry et al. Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid [Internet]. Thyroid. 2003 Jan;13(1):3-126. PubMed | Google Scholar
- Glinoer D. The regulation of thyroid function in pregnancy: Pathways of endocrine adaptation from physiology to pathology. Endocr Rev. 1997 Jun;18(3):404-33. PubMed | Google Scholar
- Sletner L, Jenum AK, Qvigstad E, Hammerstad SS. Thyroid Function during Pregnancy in A Multiethnic Population in Norway. J Endocr Soc. 2021 May 4;5(7):bvab078. PubMed | Google Scholar
- Yassaee F, Farahani M, Abadi AR. Prevalence of Subclinical Hypothyroidism in Pregnant Women in Tehran-Iran. Int J Fertil Steril. Royan Institute; 2014;8:163. Google Scholar
- Abdulslam K, Yahaya I. Prevalence of thyroid dysfunction in gestational hypertensive Nigerians. Sub-Saharan African J Med. 2015;2:19. Google Scholar
- Gupta P, Jain M, Verma V, Gupta NK. The Study of Prevalence and Pattern of Thyroid Disorder in Pregnant Women: A Prospective Study. Cureus. 2021 Jul 18;13(7):e16457. PubMed | Google Scholar
- Nandi-Munshi D, Taplin CE. Thyroid-related neurological disorders and complications in children. Pediatr Neurol [Internet]. Elsevier Inc; 2015;52:373-82. PubMed | Google Scholar
- Dhanwal DK, Bajaj S, Rajput R, Subramaniam KAV, Chowdhury S, Bhandari R et al. Prevalence of hypothyroidism in pregnancy: An epidemiological study from 11 cities in 9 states of India. Indian J Endocrinol Metab. 2016;20:387. PubMed | Google Scholar
- Dülek H. The Prevalance of Thyroid Dysfunction and Relationship with Perinatal Outcomes in Third Trimester Pregnants Who Apply to Tertiary Center. North Clin Istanbul. 2019 Sep 2;6(3):267-272. PubMed | Google Scholar
- Sepasi F, Rashidian T, Shokri M, Badfar G, Kazemi F, Azami M. Thyroid dysfunction in Iranian pregnant women: A systematic review and meta-analysis. BMC Pregnancy and Childbirth. 2020 Jul 14;20(1):405. PubMed | Google Scholar
- Goel P, Kaur J, Saha PK, Tandon R, Devi L. Prevalence, associated risk factors and effects of hypothyroidism in pregnancy: A study from north India. Gynecol Obstet Invest. 2012;74(2):89-94. PubMed | Google Scholar
- Diéguez M, Herrero A, Avello N, Suárez P, Delgado E, Menéndez E. Prevalence of thyroid dysfunction in women in early pregnancy: Does it increase with maternal age? Clin Endocrinol (Oxf). 2016 Jan;84(1):121-6. Google Scholar
- Casey BM, Dashe JS, Wells CE, McIntire DD, Leveno KJ, Cunningham FG. Subclinical Hyperthyroidism and Pregnancy Outcomes. Obstet Gynecol [Internet]. 2006 Feb;107(2 Pt 1):337-41. PubMed | Google Scholar
- Shahbazian H, Shahbazian N, Rahimi Baniani M, Yazdanpanah L, Latifi SM. Evaluation of thyroid dysfunction in pregnant women with gestational and pre-gestational diabetes. Pakistan J Med Sci [Internet]. 2013;29(2):638-41. PubMed | Google Scholar
- Shun CB, Donaghue KC, Phelan H, Twigg SM, Craig ME. Thyroid autoimmunity in Type 1 diabetes: Systematic review and meta-analysis. Diabet Med. 2014 Feb;31(2):126-35. PubMed | Google Scholar