Speciation and antifungal susceptibility of Candida isolates from diabetic foot ulcer patients in a tertiary hospital in Kenya
Victor Moses Musyoki, Winnie Mutai, Nancy Ngugi, Fredrick Otieno, Moses Muia Masika
Corresponding author: Victor Moses Musyoki, Department of Medical Microbiology, School of Medicine, College of Health Sciences, University of Nairobi, Nairobi, Kenya 
Received: 16 Jul 2021 - Accepted: 06 Jan 2022 - Published: 13 Jan 2022
Domain: Microbiology,Diabetes care,Infectious disease
Keywords: Diabetic foot ulcer, Candida species, antifungal resistance, susceptibility, fungi
©Victor Moses Musyoki et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Victor Moses Musyoki et al. Speciation and antifungal susceptibility of Candida isolates from diabetic foot ulcer patients in a tertiary hospital in Kenya. Pan African Medical Journal. 2022;41:34. [doi: 10.11604/pamj.2022.41.34.30815]
Available online at: https://www.panafrican-med-journal.com//content/article/41/34/full
Research 
Speciation and antifungal susceptibility of Candida isolates from diabetic foot ulcer patients in a tertiary hospital in Kenya
Speciation and antifungal susceptibility of Candida isolates from diabetic foot ulcer patients in a tertiary hospital in Kenya
![]() Victor Moses Musyoki1,&, Winnie Mutai1, Nancy Ngugi2, Fredrick Otieno3,
Victor Moses Musyoki1,&, Winnie Mutai1, Nancy Ngugi2, Fredrick Otieno3, ![]() Moses Muia Masika1,4
Moses Muia Masika1,4
&Corresponding author
Introduction: diabetic foot ulcer is the leading cause of hospital admissions, lower limb amputation and death among diabetic patients. Little information is available on fungal isolation in diabetic foot ulcer patients, especially in sub-Saharan Africa. This study aimed to describe Candida species infecting diabetic foot ulcers in patients receiving clinical care at Kenyatta National Hospital and assess their antifungal susceptibility profile.
Methods: this was a cross-sectional study carried out at Kenyatta National Hospital among adult diabetic foot ulcer patients over a three-month period. Species identification of Candida was performed using VITEK - 2 System and further confirmed by Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry. Antifungal susceptibility testing was determined using VITEK-2 System. Data were analysed using WHONET and SPSS.
Results: among the 152 study patients recruited, 98% (n=149) had type 2 diabetes. Sixty one percent of the participants were male. The mean age of the study participants was 50.7 years (SD 12.9). A total of 36 Candida species were isolated, of which 75% (n=27) were Candida albicans. Candida lusitaniae (8%, n=3) and C. dubliniensis (5%, n=2) were the predominant non-albicans Candida species. The overall prevalence of diabetic foot ulcer candidiasis was 20% (n=31). C. albicans isolates (26%) were resistant to caspofungin, fluconazole, micafungin, and voriconazole but highly susceptible to amphotericin B and flucytosine (81-96%). Non-albicans Candida species isolated were susceptible (90-100%) to a majority of the antifungal agents tested.
Conclusion: Candida albicans was the predominant species isolated and showed low resistance rates to the commonly administered antifungal agents. There is need to include fungal diagnosis in the investigation of diabetic foot ulcer infection.
Diabetes mellitus is a metabolic disorder characterized by chronic hyperglycemia [1-3] and severe complications [3]. Globally, 1.6 million deaths occur every year due to diabetes and diabetes-related complications, with more than 80% occurring in low and middle-income countries [4,5].
Diabetic foot ulcer (DFU) is the most common complication and leading cause of hospitalization and non-traumatic lower limb amputations. It´s estimated that 10-15% of diabetic patients will develop DFU during their diabetic life [5-7]. According to a recent meta-analysis, the overall prevalence of DFUs is about 13%, while in Kenya, Nyamu et al. reported a prevalence of 4.6%, slightly lower than what was previously reported in Tanzania (7.3%) and Egypt (6.2%) [8,9]. The foot ulcer is a significant predisposing factor for microbial infections [10] and early diagnosis and treatment of microbial agents with appropriate antimicrobial therapy is essential. In most instances, the infected wounds are polymicrobial in nature, and information on infecting bacterial agents is available from studies done in both developed and developing countries [11]. Although culture dependent techniques have been widely utilized in isolating infecting microorganisms, major focus has been on Staphylococcus aureus and gas gangrene associated Clostridium perfringens creating isolation biasness [10,12].
Lack of guidelines on diagnosis and treatment of DFU for microorganisms including fungi, especially in sub-Saharan Africa has contributed largely to the paucity of data on fungal agents infecting DFU. Fungal DFU infection poses a major health concern with antifungal resistance complicating management of these infections and significantly increasing incidence rates of lower limb amputation despite the proper surgical and antimicrobial therapy [13,14]. Candida species is the principal fungal pathogen with non-albicans Candida (NAC) species emerging as important pathogens isolated from DFU [15,16]. Multidrug resistant Candida albicans and pandrug resistant C. auris cases have been reported in Asia, South Africa and United States and have led to increased length of hospital stay and healthcare costs [17]. Antimicrobial resistance remains a global health problem and a threat to management of diabetic foot infections [18,19]. Identification of Candida species and the primary antifungal drugs to use for treatment in diabetic foot infection is essential in reducing the cost of managing diabetic foot ulcers, amputation and monitoring of antifungal drug resistance.
This study was conducted to determine the species and antifungal susceptibility pattern of Candida isolated from diabetic foot ulcers in patients receiving clinical services at Kenyatta National Hospital in Nairobi, Kenya.
Study design and subjects: this was a cross sectional study carried out in Kenyatta National Hospital (KNH), a tertiary teaching and referral hospital in Nairobi, Kenya. Using Cochran sample size calculation for finite population correction and KNH DFU records (180) for three months, we consecutively recruited adult diabetic patients, both outpatient and inpatient with any type of diabetes presenting with acute and chronic non-healing foot ulcers. Chronic foot ulcers were defined as wounds that did not heal within 3 (three) months. Patients who were on immunosuppressive drugs/state, systemic or topical antimicrobial agents for more than one week at the time of enrolment were excluded.
Data and sample collection: clinical and socio-demographic data was collected using a structured questionnaire after obtaining written informed consent. A total of 152 non-repetitive deep tissue samples were collected over a three-month period from each patient using sterile cotton swabs (levine technique) after debriding and cleansing the wound with normal saline (NaCl, 0.9%). The swabs were taken to University of Nairobi Microbiology Laboratory within 2 hours for analysis.
Microbiological methods: two smears from each sample were prepared and examined microscopically in 10% potassium hydroxide (KOH) and gram staining. The specimens were inoculated on Sabouraud Dextrose Agar (SDA) media supplemented with chloramphenicol and gentamicin and incubated under aerobic conditions at 37°C for 24-48 hours. Growth on SDA was evaluated for colonial morphology and the yeast identified by conventional methods including germ tube test, microscopic morphology on KOH and gram staining. Further identification including speciation was done using VITEK-2 System (YST card) and Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS). Antifungal susceptibility testing of Candida isolates was done using the VITEK� 2 System (AST-YS08) (BioMérieux, France) and analyzed according to the 2017 Clinical and Laboratory Standards Institute guideline (CLSI M60). The panel of antifungal agents tested included amphotericin B, caspofungin, fluconazole, flucytosine, micafungin and voriconazole. C. albicans ATCC 10231 and C. parapsilosis ATCC 22019 were used as controls during the laboratory procedure.
Statistical analysis: identification and antifungal susceptibility data were retrieved from the VITEK� 2 System and imported to WHONET (version 5.6) through BACLINK software. Analysis was done using WHONET and IBM SPSS Statistics version 21. Frequency distribution and proportions was done for categorical variables such as gender, type of diabetes and measures of central tendency for numerical variables such as age. Chi-square was done in bivariate analysis to assess any association between categorical variables. Confidence intervals were calculated using the Agresti-Coull interval as recommended in the CLSI M60 document which details analysis and presentation of cumulative antimicrobial susceptibility test data.
Ethics statement: this study was approved by the Kenyatta National Hospital-University of Nairobi Ethics and Research Committee (P290/04/2019). Permission to conduct the study in Kenyatta National Hospital was granted by the Head of Department, Medicine and the Head of Diabetes and Endocrine Clinic, Kenyatta National Hospital. Patients were enrolled in the study after written informed consent was obtained. Data was collected using anonymous questionnaires and no personal identifiers were analyzed.
Patient demographics: a total of 152 patients met the inclusion criteria with nearly all patients presenting with type 2 diabetes (98%, n=149). Majority of these patients were male (61%, n=93) and urban dwellers (74%, n=113). The mean age of the study participants was 50.7 years (SD, 12.9). The median duration of diabetes and diabetic foot ulcer was 11 years (IQR 5.25-11.0) and 2 months (IQR 1.0-3.0) respectively. Median random blood sugar level was 7.10 mmol/L (IQR 5.93-8.19). Approximately 20% (n=30) of the patients were on antibiotic treatment mainly metronidazole (80%, n=24), as prophylaxis while none was on antifungal drug (Table 1).
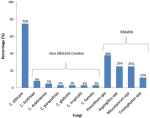
Isolation of Candida, other fungi and bacteria: out of the 152 samples collected from the foot ulcers, 31 had yeast cells which were confirmed by germ tube test (GTT) and further by VITEK - 2 System and MALDI-TOF MS. Of the 31 samples presenting with yeast cells, we isolated and differentiated 36 Candida species. C. albicans (75%, n=27) was the most frequently encountered species while C. lusitaniae (8%, n=3) was the predominant non-albicans Candida isolate followed by C. dubliniensis (5%, n=2). Other non-albicans Candida species identified included C. glabrata, C. tropicalis, C. famata and C. parapsilosis (each 2%, n=1). Majority of the Candida isolates were from male (24%, n=22), patients above 40 years (20%, n=23), acute wounds (21%, n=20) and ulcers of Wagner grade I and II (20%, n=26). However, this association was not statistically significant (p>0.05). Additionally, moulds were isolated from eight culture plates after 7-14 days aerobic incubation at 19-25°C. Based on colonial morphology on SDA and Lactophenol Cotton Blue (LPCB) staining technique, the moulds were identified and differentiated as Penicillium spp (38%, n=3), Aspergillus spp (25%, n=2), Microsporum spp (25%, n=2) and Trichophyton mentagrophytes (12%, n=1) (Figure 1).
Other than fungi we also identified bacteria species from 59 samples using gram staining microscopy method. Nine (15%) of these had gram positive cocci in clusters, 30 (51%) had gram negative rods while 20 (34%) of these samples had mixed bacterial infection of both gram positive cocci in clusters and gram negative rods. We further noted monomicrobial etiology (C. albicans) in only 4 (2.6%) of the samples compared to 27 (17.8%) of polymicrobial existence. The predominant polymicrobial etiology comprised of C. albicans, gram positive cocci in clusters and gram negative rods.
Antifungal susceptibility profile: overall, Candida species (n=35) were susceptible to voriconazole, flucytosine, micafungin, caspofungin, amphotericin B and fluconazole (77-97%). Among the six antifungal agents, the highest level of susceptibility was noted in flucytosine and amphotericin B while highest level of resistance was observed in caspofungin. Although the number of C. albicans and non-albicans Candida species isolated in this study did not meet the CLSI AST reporting threshold (>30 isolates each), the results were presented due to the mycological significance. C. albicans was resistant to caspofungin, fluconazole, micafungin and voriconazole (26%) but susceptible to amphotericin B and flucytosine (81-96%). Non-albicans Candida species isolated were susceptible (90-100%) to a majority of the antifungal agents tested (Table 2).
The aim of this study was to determine the Candida species infecting DFU and assess their antifungal susceptibility pattern. Candida albicans was the most common species isolated while among the NAC species, C. lusitaniae and C. dubliniensis were the most prevalent. Overall, Candida species isolated were susceptible to voriconazole, flucytosine, micafungin, caspofungin, amphotericin B and fluconazole. The highest level of susceptibility was noted in flucytosine and amphotericin B while highest level of resistance was observed in caspofungin.
The prevalence of DFU candidiasis (20.4%) and predominance of C. albicans in this study is consistent with previous similar studies carried out in Turkey, India and Iran that reported prevalence ranging from 16-30% and isolated C. albicans as the major species [20-22]. Contrary to these findings, other studies carried out in other parts of the world reported C. parapsilosis as the predominant yeast in DFUs suggestive of emergence of NAC as significant pathogens [14,15]. The high frequency of Candida species in DFU may be attributed to immunological imbalances and increased glucose concentration in tissues and body fluids that predispose diabetic patients to fungal infections. In addition, immunomodulating action of antibiotics may support yeast survival and replication [23-25]. While we identified C. lusitaniae and C. dubliniensis as the most prevalent NAC species, these have not been previously reported in DFU [14,15].
The diversity of etiology seen in this study comprising mostly of a mixture of Candida species, gram negative rods, gram positive cocci and moulds may contribute to the chronic state of the wounds. This findings concur with previous studies in other parts of the world that have shown nearly all cases of DFU infections are polymicrobial [26-28]. Although the polymicrobial nature of DFU infection is not clear, it may be related to impaired immune system, gene regulation in biofilm formation and the non-fastidious nature of most of the organisms [25,26,29]. Earlier reports from India [8] and China [30] have reported monomicrobial infections comprising mostly of gram negative bacteria among acute DFU patients [14].
The emergence of multidrug and pandrug-resistant C. albicans and NAC noted in several parts of the world necessitates continuous antifungal susceptibility testing and monitoring. The NAC isolates in this study were 100% susceptible to voriconazole, micafungin, fluconazole and flucytosine. C. albicans showed high rate of resistance to the antifungals tested than NAC, similar to observations noted in earlier studies in Europe, India, and Kenya that reported a comparable resistance rate (20-48%) of C. albicans to triazoles [16,31,32]. Additionally, resistance to both triazole (fluconazole and voriconazole) and echinocandins (caspofungin and micafungin) group of antifungals was relatively high compared to amphotericin B and flucytosine. This observation is contrary to what has been documented in Saudi Arabia, Tunisia, and South Africa, where susceptibility rates of 96-100% to triazoles and echinocandins to both C. albicans and NAC species were recorded [33-35]. The differences in resistance rates noted in C. albicans and NAC species to triazole and echinocandins could be explained by the high clinical usage especially the fluconazole in immunocompromised patients as prophylactic drug, molecular activation of the efflux pump and mutation of ERG11 and ERG3 genes involved in azole target binding and accumulation of the toxic sterol 14-α-methyl-3, 6 diol [36]. Resistance in echinocandins may be attributed to mutations of FK1 and FK2 genes encoding for the enzyme glucan synthetase [37].
Candida albicans and NAC species isolated in this study showed low rates of resistance to amphotericin B similar to results of studies done in India and South Africa that reported resistance rate of 4-10% to amphotericin B [14,34]. The low incidence of resistance to amphotericin B in our study and previous similar studies could be explained by the fact that the agent is not commonly used among diabetic patients due to hypokalemia-associated nephrotoxicity. Our findings contrasts with those from other studies done in different parts of the world that reported up to 100% susceptibility rate of amphotericin B to Candida species [38-41]. Resistance to polyenes in Candida species may be associated with defective C5, 6-desaturase functionality in C. albicans and mutation of ERG 2, 3, 5, 6 and 11 genes involved in ergosterol cell membrane synthesis. Different studies have shown C. lusitaniae to be intrinsically resistant to amphotericin B which could also explain the resistance noted among the NAC species in our study [36,42].
Generally, Candida species exhibit susceptibility activity to flucytosine which is used in combination with other antifungal agents in treatment of yeast infections. Resistance to this drug is slowly emerging as demonstrated in this study where C. albicans and NAC species showed low resistance and high susceptibility rates, respectively, to flucytosine. These findings are comparable to the low resistance rate (4-10%) and high susceptibility to flucytosine in both C. albicans and NAC isolates documented in studies from different tertiary hospitals in Europe, India, Iran and South Africa [14,34,43,44]. The low resistance to flucytosine observed in these studies may be due to the synergistic combination of the drug with other antifungal agents for clinical use. Resistance noted in monotherapy as shown in similar studies may be attributed to mutations of FCY1, 2 and FUR1 genes associated with actively transportation of the drug into the fungal cell and enzymatic conversion of the drug into 5-fluorouracil or 5-fluorouridine monophosphate [45]. It is also worth noting that the overall resistance noted to the different classes of antifungal agents in Candida species may also be due to biofilms formed by the organisms present in these wounds [46,47].
The main limitation in our study was the small sample size, lack of clinical information, particularly HBA1C to correlate with Candida infection, and the VITEK-2 AST panel of antifungal agents which excluded other agents. Identification of bacteria to genus and species level would have further supported our findings on polymicrobial infections; however, the scope of this study was to highlight fungal infecting agents. We would also have wished to detect the genes coding for resistance to support the resistance pattern observed.
This study shows both C. albicans and NAC species are important etiological agents infecting diabetic foot ulcers. The study therefore provides evidence for the need to include fungal diagnosis including species identification in the investigation of diabetic foot ulcer infection. Additionally, the low level of resistance to antifungal drugs reported in this study should not be ignored because they can gradually progress to high levels, hence continuous antifungal resistance surveillance and strengthening of antifungal stewardship programmes is imperative to enhance patient care and management. In future, further research should use advanced molecular approaches to explore the diverse group of microbes infecting DFU and establish their clinical implications.
Funding: VMM received partial funding from Kenyatta National Hospital Research and Programs Department (Award reference number: KNH/R&P/23H/99/13) to conduct this study. The funding program or funders had no role in study design, data collection, analysis and decision in preparation and publishing this manuscript.
What is known about this topic
- Diabetic foot infection is polymicrobial in nature increasing the risk of lower limb amputation;
- Antifungal resistance, especially Candida species to azole antifungal agents is an emerging healthcare concern.
What this study adds
- Candida albicans was the most frequent species isolated and showed low resistance rates to the commonly used antifungal agents;
- Non-albicans Candida species isolated were highly susceptible to antifungal agents tested;
- Isolation of yeast and moulds as fungal etiological agents infecting diabetic foot ulcers highlight the significance of including fungal diagnosis in microbiological analysis of diabetic foot infection.
The authors declare no competing interests.
All the authors were involved in conception and designing of the project. VMM and WM performed the experiments. VMM and MMM analyzed the data. All the authors read and approved the final manuscript.
The authors would like to acknowledge Isaac Miruka and all the members of staff, Department of Medicine, KNH for their time and expertise; the research assistant Mary Margaret and the patients. The authors would also like to acknowledge the Research Coordinator and Head of Department, Laboratory Medicine, Kenyatta National Hospital. Our appreciation also goes to laboratory staff, Medical Microbiology for sample analysis.
Table 1: socio-demographic and clinical characteristics of diabetic foot ulcer patients receiving clinical care at Kenyatta National Hospital (N=152)
Table 2: antifungal susceptibility profile of Candida species isolated
Figure 1: distribution of fungi isolated from diabetic foot ulcers
- Raza M, Anurshetru B. Clinical study of coexistence of fungal infections in diabetic foot ulcers and its management. Int Surg J. 2017;4(1):3943-50. Google Scholar
- Kalshetti VT, Wadile R, Bothikar ST, Ambade V, Bhate VM. Study of fungal infections in diabetic foot ulcer. Indian J Microbiol Res. 2017;4(1):87-9. Google Scholar
- Brownrigg J. RW, Apelqvist J, Bakker K, Schaper NC, Hinchliffe RJ. Evidence - Based Management of PAD & the Diabetic Foot. Eur J Vasc Endovasc Surg. 2013 Jun;45(6):673-81. PubMed | Google Scholar
- World Health Organization. Global Report on Diabetes. 2018;1-84.
- IDF. International Diabetes Federation: Diabetes Atlas. 2017;1-2.
- Zhang P, Lu J, Jing Y, Tang S, Zhu D, Bi Y. Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis. Ann Med. 2017;49(2):106-16. PubMed | Google Scholar
- Desalu OO, Salawu FK, Jimoh AK, Adekoya AO, Busari OA, Olokoba AB. Diabetic foot care: self reported knowledge and practice among patients attending three tertiary hospital in Nigeria. Ghana Med J. 2011;45(2):60-5. PubMed | Google Scholar
- Rastogi A, Sukumar S,Hajela A Mukherjee S. The microbiology of diabetic foot infections in patients recently treated with antibiotic therapy: a prospective study from India. J Diabetes Complications. 2017;31(2):407-12. PubMed | Google Scholar
- Nyamu PN, Otieno CF, Amayo EO, Mcligeyo SO. Risk factors and prevalence of diabetic foot ulcers at Kenyatta National Hospital, Nairobi. East Afr Med J. 2003;80(1):36-43. PubMed | Google Scholar
- Karmaker M, Sanyal S, Sultana M, Hossain M. Association of bacteria in diabetic and non-diabetic foot infection - an investigation in patients from Bangladesh. Journal Infect Public Health. 2016;9(3):267-77. PubMed | Google Scholar
- Saseedharan S, Sahu M, Chaddha R, Pathrose E, Bal A, Bhalekar P. Epidemiology of diabetic foot infections in a reference tertiary hospital in India. Braz J Microbiol. 2018;49(2):401-6. PubMed | Google Scholar
- Al-hegami MA, Alghalibi SM, Al-Mamari A. Microorganisms Responsible of Diabetic Foot Infection in Taiz City, Yemen. Int J Curr Microbiol Appl Sci. 2016;5(7):431-41. PubMed | Google Scholar
- Peters, Edgar J. Pitfalls in diagnosing diabetic foot infections. Diabetes Metab Res Rev. 2016;32(1):254-60. PubMed | Google Scholar
- Chellan G, Shivaprakash S, Ramaiyar S, Karimassery V, Varma N, Kumar H. Spectrum and prevalence of fungi infecting deep tissues of lower-limb wounds in patients with type 2 diabetes. J Clin Microbiol. 2010;48(6):2097-102. PubMed | Google Scholar
- Gitau A, Ng´ang´a Z, Sigilai W, Bii C, Mwangi M. Fungal infections among diabetic foot ulcer patients attending diabetic clinic in Kenyatta National Hospital, Kenya. East Afr Med J. 2011;88(1):9-17. PubMed | Google Scholar
- Khadka S, Sherchand JB, Pokhrel BM, Parajuli K, Mishra SK. Isolation, speciation and antifungal susceptibility testing of Candida isolates from various clinical specimens at a tertiary care hospital, Nepal. BMC Res Notes. 2017 Jun 24;10(1):218. PubMed | Google Scholar
- Centre of Disease Control. Candida auris: a drug-resistant germ that spreads in healthcare facilities. 2019. PubMed | Google Scholar
- Vandeputte P, Ferrari S, Coste AT. Antifungal resistance and new strategies to control fungal infections. Int J Microbiol. 2012;2012:26. PubMed | Google Scholar
- Kean R, Delaney C, Rajendran R, Sherry L, Metcalfe R, Thomas R et al. Gaining insights from Candida biofilm heterogeneity: One Size Does Not Fit All. J Fungi. 2018 Jan 15;4(1):12. PubMed | Google Scholar
- Fata S, Modaghegh M, Naseri A, Mohammadian M, Ghasemi M, Meshkat M et al. Mycotic infections in diabetic foot ulcers in Emam Reza Hospital, Mashhad, 2006-2008. Jundishapur J Microbiol. 2011;4(1):11-6. Google Scholar
- Abilah S, Kannan N, Rajan K, Pramodhini M, Ramanathan M. Clinical study on the prevalence of fungal infections in diabetic foot ulcers. Int J Curr Res Rev. 2015;7(23):8-13. Google Scholar
- Öztürk AM, Tasbakan MI, Metin DY, Yener C, Uysal S. A neglected causative agent in diabetic foot infection: a retrospective evaluation of 13 patients with fungal etiology. Turk J Med Sci. 2019 Feb 11;49(1):81-86. PubMed | Google Scholar
- Ali EM. Ozone application for preventing fungal infection. Diabetol Croat. 2013;42(1):3-22. Google Scholar
- Mlinaric-Missoni E, Kalenic S, Vukelic M, Syo D, Belicza M, Vazic-Babic V. Candida infections of diabetic foot ulcers. Diabet Med. 2005 Aug;22(8):1124-5. PubMed | Google Scholar
- Malone M. The Microbiome of Diabetic Foot Ulcers and the Role of Biofilms: a thesis submitted in partial fulfilment of the requirements for the degree of Doctor of Philosophy (PhD) [thesis]. [Australia]: Western Sydney University; 2018. Google Scholar
- Wu M, Pan H, Leng W, Lei X, Chen L, Liang Z. Distribution of microbes and drug susceptibility in patients with diabetic foot infections in Southwest China. J Diabetes Research. 2018 Aug 5;2018:9817308. PubMed | Google Scholar
- Kareliya H, Bichile L, Bal A, Varaiya A, Bhalekar P. Fungal infection in diabetic foot a clinico-microbiological study. Acta Sci Microbiology. 2019;2(7):49-55. Google Scholar
- Jouhar L, Minhem M, Akl E, Rizk N, Hoballah J. Microbiological profile of diabetic foot infection in the Middle East and North Africa: a systematic review. Diabet Foot J Middle East. 2019;6(1):43-50. Google Scholar
- Mutonga D, Mureithi M, Ngugi N, Otieno F. Bacterial isolation and antibiotic susceptibility from diabetic foot ulcers in Kenya using microbiological tests and comparison with RT PCR in detection of S. aureus and MRSA. BMC Res Notes. 2019;12(1):244. PubMed | Google Scholar
- Huang Y, Cao Y, Zou M, Luo X, Jiang Y, Xue Y et al. A comparison of tissue versus swab culturing of infected diabetic foot wounds. Int J Endocrinol. 2016;2016:8198714. PubMed | Google Scholar
- Minea B, Nastasa V, Moraru R, Kolecka A, Flonta M, Marincu I et al. Species distribution and susceptibility profile to fluconazole, voriconazole and MXP-4509 of 551 clinical yeast isolates from a Romanian multi-centre study. Eur J Clin Microbiol Infect Dis. 2015 Feb;34(2):367-83. PubMed | Google Scholar
- Ooga V, Bii C, Gikunju J. Characterization and antifungal drug susceptibility of clinical isolates of Candida species. Afr J Health Sci. 2015;19(3):84-92. Google Scholar
- Johargy A. Antimicrobial susceptibility of bacterial and fungal infections among infected diabetic patients. J Pak Med Assoc. 2016 Oct;66(10):1291-1295. PubMed | Google Scholar
- Mnge P, Okeleye B, Vasaikar S, Apalata T. Species distribution and antifungal susceptibility patterns of Candida isolates from a public tertiary teaching hospital in the Eastern Cape Province, South Africa. Braz J Med Biol Res. 2017 May 15;50(6):e5797. PubMed | Google Scholar
- Eddouzi J, Lohberger A, Vogne C, Manai M, Sanglard D. Identification and antifungal susceptibility of a large collection of yeast strains isolated in Tunisia hospitals. Med Mycol. 2013 Oct;51(7):737-46. PubMed | Google Scholar
- Wiederhold NP. Antifungal resistance: current trends and future strategies to combat. Infect Drug Resist. 2017 Aug 29;10:249-259. PubMed | Google Scholar
- Sanguinetti M, Posteraro B, Lass-Florl C. Antifungal drug resistance among Candida species: mechanisms and clinical impact. Mycoses. 2015 Jun;58 Suppl 2:2-13. PubMed | Google Scholar
- Sugandhi P, Prasanth DA. Prevalence of yeast in diabetic foot infections. Int J Diabetes Dev Ctries. 2016;1(1):491-8. Google Scholar
- Munguia-perez R, Remigio-alvarado N, Hernandez-arroyo MM, Castañeda-roldan E. Antifungal susceptibility of yeasts isolated from clinical samples from a tertairy hospital from State of Puebla. Int J Med Health Sci. 2017;11(6)
- Tasneem U, Siddiqui MT, Faryal R, Shah AA. Prevalence and antifungal susceptibility of Candida species in a tertiary care hospital in Islamabad, Pakistan. J Pak Med Assoc. 2017 Jul;67(7):986-991. PubMed | Google Scholar
- Zaidi KU, Mani A, Parmar R, Thawani V. Antifungal susceptibility pattern of candida albicans in human infections. Open Biol Sci J. 2018;4(3):1-6. Google Scholar
- Cowen L, Sanglard D, Howard S, Rogers D, Perlin D. Mechanisms of antifungal drug resistance. Cold Spring Harb Lab Press. 2014 Nov 10;5(7):a019752. PubMed | Google Scholar
- Schmalreck A, Willinger B, Haase G, Blum G, Fegeler W, Becker K. Species and susceptibility distribution of 1062 clinical yeast isolates to azoles, echinocandins, flucytosine and amphotericin B from a multi-centre study. Mycoses. 2012 May;55(3):e124-37. PubMed | Google Scholar
- Sadeghi G, Zeinali E, Alirezaee M, Amani A, Mirahmadi R, Tolouei R. Species distribution and antifungal susceptibility of Candida species isolated from superficial candidiasis in outpatients in Iran. J Mycol Medicale. 2014;24(2):e43-50. PubMed | Google Scholar
- Arendrup MC, Patterson T. Multidrug-resistant candida: epidemiology, molecular mechanisms and treatment. J Infect Dis. 2017;216(Suppl 3):445-51. PubMed | Google Scholar
- Silva S, Rodrigues C, Ara D, Rodrigues ME, Henriques M. Candida species biofilms´ antifungal resistance. J Fungi. 2017 Feb 21;3(1):8. PubMed | Google Scholar
- Bruder-nascimento A, Camargo CH, Mondelli AL, Sugizaki MF, Sadatsune T, Bagagli E. Candida species biofilm and Candida albicans ALS3 polymorphisms in clinical isolates. Braz J Microbiol. 2015 Mar 4;45(4):1371-7. PubMed | Google Scholar