Undiagnosed apical hypertrophic cardiomyopathy in an old amateur soccer player: a case report
Mahassine El Harras, Ilham Bensahi, Salma Abdeladim, Fatimazahra Merzouk, Amal Elouarradi, Sara Oualim, Mohamed Sabry
Corresponding author: Mahassine El Harras, Department of Cardiology, Mohammed VI University of Health Sciences Cheikh Khalifa Hospital, Casablanca, Morocco 
Received: 26 Feb 2021 - Accepted: 03 Nov 2021 - Published: 25 Nov 2021
Domain: Cardiology
Keywords: Hypertrophic cardiomyopathy, apical hypertrophic cardiomyopathy, sport cardiology, case report
©Mahassine El Harras et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Mahassine El Harras et al. Undiagnosed apical hypertrophic cardiomyopathy in an old amateur soccer player: a case report. Pan African Medical Journal. 2021;40:182. [doi: 10.11604/pamj.2021.40.182.28588]
Available online at: https://www.panafrican-med-journal.com//content/article/40/182/full
Case report 
Undiagnosed apical hypertrophic cardiomyopathy in an old amateur soccer player: a case report
Undiagnosed apical hypertrophic cardiomyopathy in an old amateur soccer player: a case report
![]() Mahassine El Harras1,&,
Mahassine El Harras1,&, ![]() Ilham Bensahi1, Salma Abdeladim1, Fatimazahra Merzouk1,
Ilham Bensahi1, Salma Abdeladim1, Fatimazahra Merzouk1, ![]() Amal Elouarradi1,
Amal Elouarradi1, ![]() Sara Oualim1, Mohamed Sabry1
Sara Oualim1, Mohamed Sabry1
&Corresponding author
Hypertrophic cardiomyopathy is a primary muscle disorder characterized by an abnormal thickness of the left ventricular wall. It is often going undiagnosed because many patients have few symptoms and can lead normal lives. This is a case report about an apical cardiomyopathy diagnosed at a very late stage in an old amateur soccer player. He was hospitalized due to acute chest pain; neurologic disorder related to a hypertensive emergency, he underwent successful percutaneous coronary intervention, echocardiography and CMR revealed Apical hypertrophic cardiomyopathy. The development of sports cardiology has major importance in the detection of cardiac disease which may have poor prognosis. Our patient had the chance to achieve his entire career without rhythmic complications.
Hypertrophic cardiomyopathy is a muscle disorder with several morphological manifestations, characterized by a natural history, and specific prognosis, it´s a complex, primary, and inherited cardiac disease [1,2]. Patients with Hypertrophic cardiomyopathy (HCM) can present one or more of non-pathognomonic symptoms from shortness of breath to arrythmias causing sudden death, hence the interest of early diagnosis, good management and family screening. We report a case of an apical cardiomyopathy none diagnosed in an old amateur soccer player.
Patient information: a 72-year-old man, high level athlete who exercised as an amateur soccer player during 12 years, with a 1-year history of hypertension, diabetes mellitus since 2010.
Clinical findings: he was hospitalized due to a hypertensive emergency.
Timeline of current episode: the patient presented an acute chest pain, neurologic disorder, a left sided weakness related to a hypertensive emergency.
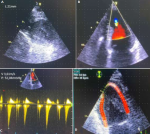
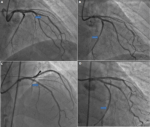
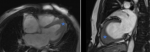
Diagnostic assessment: he presented a high blood pressure up to 220/110mmHg, the electrocardiogram showed an atrial fibrillation with deep negative T waves. Cerebral Magnetic resonance imaging (MRI) revealed a recent left sylvien stroke. Further, echocardiography (Figure 1) revealed normal systolic function with left ventricular ejection fraction (LVEF) as 65%, apical hypertrophic cardiomyopathy the wall thickness was up to 21mm with left ventricular obstruction, he underwent a coronarography, it revealed a double stenosis in the proximal and middle left anterior descending artery, and sever stenosis in the distal circumflex artery, he was taken for successful percutaneous coronary intervention (PCI) through femoral route (Figure 2). Cardiac magnetic resonance imaging (CMR) was performed, it detected apical LV hypertrophy up to 17mm in the apical anterior wall, 12mm in the apical inferior wall, 18mm in the apical septal and lateral walls, with fibrosis in the apex and apical segment of the LV (Figure 3). The Holter electrocardiogram (EKG) showed sustained ventricular tachycardia. The results were consistent with an apical hypertrophic cardiomyopathy corresponding to the fourth type of Marron classification.
Therapeutic interventions: the patient was treated with propranolol, ischemic cardiopathy medication and anticoagulant.
Follow-up and outcome of interventions: the sustained ventricular tachycardia persisted under medical treatment, according to the European society of cardiology (ESC) recommendation the risk of cardiac sudden death was up to 7.74%, an International classification of diseases (ICD) was placed. A family screening was performed, including medical history, clinical examination, EKG, and a transthoracic echography in the first-degree relatives.
Patient perspective: the patient hopes to lead a normal life, and hopes his descendants are free from the disease.
Informed consent: the patient was informed, and consent was obtained.
Hypertrophic cardiomyopathy is defined by “the presence of increased left ventricular wall thickness that is not explained by abnormal loading conditions” [3]. Maron described four types, type I corresponds to the basal septum´s hypertrophy, type II corresponds to the whole septum´s hypertrophy, type III the hypertrophy interest the septum, anterior and anterolateral wall and type IV is represented by a LV apical hypertrophy [4]. This fourth type is a rare morphologic variant and an atypical form of HCM along with mid-ventricular obstruction, appointed as Apical HCM, spade shaped, Japanese variety, Yamaguchi syndrome [3,5]. Sakamoto described a cardiac disorder manifested by negative T-waves on electrocardiography, associated with apical hypertrophy of the left ventricle [6]. The prevalence is high in Japan and Asian countries. It´s around 15% in the Japanese population as compared to 3% of a United states cohort [7]. Apical HCM has been recognized as familial disease, implicating the role of genetics in the development of this morphological pattern of hypertrophy [8]. More than half of patients are asymptomatic, they are male and are diagnosed during their 4th decade [9]. The most common symptom is atypical chest pain, dyspnea, exercise intolerance, palpitations, atrial fibrillation and syncope [10]. The wave inversions in precordial leads are present in 93% of the cases, the giant and deep T waves are present only in 47%. Furthermore, electrocardiographic left ventricular hypertrophy is found in 65% of the cases [6,11]. The Holter EKG can show the presence of ventricular ectopies, which is a risk factor of sudden death [12]. Typical echocardiographic finding includes increased thickness of the left ventricular wall in the apical region. The cut-off value is fixed at 15mm although in the case of typical clinical symptoms and apex morphology, the wall thickness value is between 13 and 15mm. In the four-chamber view the echocardiographic shape of the left ventricle is compared to an “ace of spades”. The echocardiographic contrast is used to improve the visualization of left ventricle´s morphology and objectify the presence of apical aneurysm [10,13]. The diastolic gradient must be determined; it´s a potential additional risk factor of sudden death [14]. Cardiovascular magnetic resonance (CMR) provide a high-resolution image, complete covering of left ventricle morphology including the apex. It shows a typical “ace-of-spades” silhouette of LV, apex wall width > 15mm with basal/apex wall thickness ratio > 1.5 [15].
The extent of late gadolinium enhancement can be used to predict cardiovascular mortality, but current data do not support its use in prediction of Sickle cell disease (SCD) risk [16]. Making the difference between hypertensive heart disease and HCM is a real challenge, there is some clinical features favoring HCM with systemic hypertension such as family history of HCM, marked repolarization abnormalities, conduction disease or Q-waves on EKG, right ventricular hypertrophy, severe diastolic dysfunction, late gadolinium enhancement, maximum LV wall thickness [3]. The use of a β-blockers or calcium channel blockers is recommended to prolong diastole time in patients with preserved ejection fraction. In patients with reduced ejection fraction typical heart failure medication should be used. All patients with atrial fibrillation should receive anticoagulation Therapy. Surgical myotomy and alcohol ablation are the recommended method of treatment when patients are refractory to optimal medical treatment. The aim of surgical treatment is to increase the volume of the left ventricle [10]. Data about the use of surgical myotomy or alcohol septal ablation in AHCM are limited; a few cases have been reported [17]. The Association for healthcare communications and marketing (AHCM) prognosis is better than other form of HCM, cardiovascular mortality is up to 1.9%, the most frequent morbid event are atrial fibrillation and myocardial infarction [11]. Recent studies suggest that the risk of sudden cardiac death, morbid events, is com�parable to patients with other variants of hypertrophic cardiomyopathy [18]. According to American guidelines patients who have one of the following risk factors as family history of SCD, syncope, asymptomatic non-sustained ventricular tachycardia, an abnormal blood pressure response to exercise and a left ventricular wall thickness > 30mm, must have an implantable defibrillator for primary prevention [19]. In 2014, a hypertrophic cardiomyopathy risk-SCD was elaborated to indicate the ICD implantation [3]. As an emerging country the sport cardiology was not developed in the sixties. Soccer players hadn´t systematically an echocardiographic imaging, some of them died on the grass, even emergency medicine was not developed. Our patient was a regional team player, he never experienced any cardiac symptom and fortunately he didn´t have any complication during his matches.
Our patient had the luck to achieve his professional career as a soccer player without any rhythmic complication but in the other side he developed morbid events. Sport cardiology must be improved in our country. Even if the AHCM´s prognosis can be better all patients must have an assessment of the risk of sudden death and the family screening should be considered.
The authors declare no competing interests.
Mahassine El Harras: conception, literature review, drafting of the manuscript. Ilham Bensahi: conception, literature review, reviewing manuscript. Salma Abdeladim, Fatimazahra Merzouk, Amal Elouarradi, Sara Oualim: literature review, reviewing manuscript, patient�s medical management. Mohamed Sabry: visualization and supervision. All authors have read and agreed to the final manuscript.
Figure 1: transthoracic echocardiography; A) apical hypertrophic cardiomyopathy with a wall thickness up to 21mm; B, C) left ventricular obstruction; D) altered apical strain
Figure 2: coronarography; A, B) double stenosis in the proximal and middle left anterior descending artery, severe stenosis in the distal circumflex artery; C, D) successful percutaneous coronary intervention interesting left anterior descending artery and circumflex artery
Figure 3: cardiac MRI demonstrating focal apical left ventricular hypertrophy (star), confirming the diagnosis of apical hypertrophic cardiomyopathy
- Roberts R, Sigwart U. New concepts in hypertrophic cardiomyopathies, part I. Circulation. 2001 Oct 23;104(17):2113-6. PubMed | Google Scholar
- Maron BJ. Hypertrophic cardiomyopathy: a systematic review. JAMA. 2002 Mar 13;287(10):1308-20. PubMed | Google Scholar
- Authors/Task Force members, Elliott PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the task force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J. 2014 Oct 14;35(39):2733-79. PubMed | Google Scholar
- Maron BJ, Gottdiener JS, Epstein SE. Patterns and significance of distribution of left ventricular hypertrophy in hypertrophic cardiomyopathy. A wide angle, two dimensional echocardiographic study of 125 patients. Am J Cardiol. 1981 Sep;48(3):418-28. PubMed | Google Scholar
- Arad M, Penas-Lado M, Monserrat L, Maron BJ, Sherrid M, Carolyn Y Ho et al. Gene mutations in apical hypertrophic cardiomyopathy. Circulation. 2005 Nov 1;112(18):2805-11. PubMed | Google Scholar
- Sakamoto T, Tei C, Murayama M, Ichiyasu H, Hada Y. Giant T wave inversion as a manifestation of asymmetrical apical hypertrophy (AAH) of the left ventricle. Echocardiographic and ultrasono-cardiotomographic study. Jpn Heart J. 1976 Sep;17(5):611-29. PubMed | Google Scholar
- Kitaoka H, Doi Y, Casey SA, Hitomi N, Furuno T, Maron BJ. Comparison of prevalence of apical hypertrophic cardiomyopathy in Japan and the United States. Am J Cardiol. 2003 Nov 15;92(10):1183-6. PubMed | Google Scholar
- Moro E, D'Angelo G, Nicolosi GL, Mimo R, Zanuttini D. Long-term evaluation of patients with apical hypertrophic cardiomyopathy. Correlation between quantitative echocardiographic assessment of apical hypertrophy and clinical-electrocardiographic findings. Eur Heart J. 1995 Feb;16(2):210-7. PubMed | Google Scholar
- Mirabbasi SA, Khalighi K, Mukkamala S, Kodali A. A rare case of apical hypertrophic cardiomyopathy (AHCM). J Community Hosp Intern Med Perspect. 2017 Jun 6;7(2):122-125. PubMed | Google Scholar
- Paluszkiewicz J, Krasinska B, Milting H, Gummert J, Pyda M. Apical hypertrophic cardiomyopathy: diagnosis, medical and surgical treatment. Kardiochir Torakochirurgia Pol. 2018 Dec;15(4):246-253. PubMed | Google Scholar
- Eriksson MJ, Sonnenberg B, Woo A, Rakowski P, Parker TG, Wigle ED et al. Long-term outcome in patients with apical hypertrophic cardiomyopathy. J Am Coll Cardiol. 2002 Feb 20;39(4):638-45. PubMed | Google Scholar
- Rubinshtein R, Glockner JF, Ommen SR, Araoz PA, Ackerman MJ, Sorajja P et al. Characteristics and clinical significance of late gadolinium enhancement by contrast-enhanced magnetic resonance imaging in patients with hypertrophic cardiomyopathy. Circ Heart Fail. 2010 Jan;3(1):51-8. PubMed | Google Scholar
- Klarich KW, Attenhofer Jost CH, Binder J, Connolly HM, Scott CG, Freeman WK et al. Risk of death in long-term follow-up of patients with apical hypertrophic cardiomyopathy. Am J Cardiol. 2013 Jun 15;111(12):1784-91. PubMed | Google Scholar
- Nakamura T, Matsubara K, Furukawa K, Azuma A, Sugihara H, Katsume H et al. Diastolic paradoxic jet flow in patients with hypertrophic cardiomyopathy: evidence of concealed apical asynergy with cavity obliteration.J Am Coll Cardiol. 1992 Mar 1;19(3):516-24. PubMed | Google Scholar
- Brenes JC, Doltra A, Prat S. Cardiac magnetic resonance imaging in the evaluation of patients with hypertrophic cardiomyopathy. Glob Cardiol Sci Pract. 2018 Aug 12;2018(3):22. PubMed | Google Scholar
- Priori SG, Blomstr�m-Lundqvist C. 2015 European society of cardiology guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death summarized by co-chairs. Eur Heart J. 2015 Nov 1;36(41):2757-9. PubMed | Google Scholar
- Soler R, Rodríguez E, Rodríguez JA, Pérez ML, Penas M. Magnetic resonance imaging of apical hypertrophic cardiomyopathy. J Thorac Imaging. 1997 Jul;12(3):221-5. PubMed | Google Scholar
- Jan MF, Todaro MC, Oreto L, Tajik AJ. Apical hypertrophic cardiomyopathy: present status. Int J Cardiol. 2016 Nov 1;222:745-759. PubMed | Google Scholar
- Bernard JG, Barry JM, Robert OB, Joseph AD, Michael AF, Mark SL et al. 2011 ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: executive summary: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. Circulation. 2011 Dec 13;124(24):2761-96 PubMed | Google Scholar