Multidrug resistance to commonly prescribed antibiotics in Escherichia coli isolated from barbecued beef (Suya) sold in a Nigerian City
Danladi Walong Datok, David Ishaleku, Paul Alumbugu Tsaku, Elisha Obakas Agya, Moses Peter Adoga
Corresponding author: Danladi Walong Datok, Department of Microbiology, Nasarawa State University, Keffi, Nigeria 
Received: 11 Aug 2020 - Accepted: 15 May 2021 - Published: 19 May 2021
Domain: Bacteriology,Microbiology
Keywords: Drug resistance, Escherichia coli, hygiene, public health, Suya, Nigeria
©Danladi Walong Datok et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Danladi Walong Datok et al. Multidrug resistance to commonly prescribed antibiotics in Escherichia coli isolated from barbecued beef (Suya) sold in a Nigerian City. Pan African Medical Journal. 2021;39:50. [doi: 10.11604/pamj.2021.39.50.25502]
Available online at: https://www.panafrican-med-journal.com//content/article/39/50/full
Research 
Multidrug resistance to commonly prescribed antibiotics in Escherichia coli isolated from barbecued beef (Suya) sold in a Nigerian City
Multidrug resistance to commonly prescribed antibiotics in Escherichia coli isolated from barbecued beef (Suya) sold in a Nigerian City
Danladi Walong Datok1,2,&, David Ishaleku1, Paul Alumbugu Tsaku1, Elisha Obakas Agya1, ![]() Moses Peter Adoga1
Moses Peter Adoga1
&Corresponding author
Introduction: Suya, a form of barbecued meat widely consumed in Nigeria is a rising source of concern for the dissemination of pathogens and antibiotic resistance.
Methods: this study was carried out to determine the antibiotic resistance profile of Escherichia coli (E. coli) isolated from Suya sold in Karu Local Government Area, Nasarawa State, Nigeria. A total of three hundred (300) Suya samples were collected and screened for the presence of E. coli. An antibiotic susceptibility study was carried out on the isolated bacteria to determine their resistance profiles.
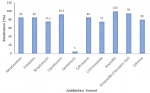
Results: the overall isolation and occurrence of E. coli was 13.3%. The isolated organisms were most resistant to Ampicillin (100%) followed by Amoxicillin/Clavulanic acid (95%), Ciprofloxacin (92.5%), Nitrofurantoin, Imipenem and Ceftriaxone (85%), Cefixime (80%), Streptomycin (77.5%), and Cotrimoxazole (77%), with a low level of resistance recorded against Gentamycin (5%). Most of the E. coli isolates had multiple resistance (MAR) to at least five antibiotics (MAR Index of ≥ 0.5) and the most frequent MAR Index was 0.8 with 37.5% occurrence. The most frequently encountered resistance phenotype was Nitrofurantoin-Imipenem-Streptomycin-Ciprofloxacin-Ceftriaxone-Amoxicillin/clavulanic acid-Cefixime-Ampicillin. The E. coli isolates categorised based on drug resistance classes were Multi-Drug Resistance, MDR (97.5%), >Pan Drug Resistance, PDR (2.5%), >Non-Multi-Drug Resistance, NMDR (0.0%) and Extensive Drug Resistance, XDR (0.0%).
Conclusion: these findings indicate a potential danger of multidrug resistant organisms in circulation. Antibiotics stewardship and drug resistance surveillance is strongly recommended for all stakeholders
Suya is a popular traditionally processed, ready-to-eat barbecued meat product, which is usually served or sold along the streets and served at hospitality industries such as social functions, club houses, picnics and restaurants. It is an ideal source of energy and animal protein. The practice of processing Suya is known from time immemorial [1] and it is consumed and perceived by consumers to be hygienic and uncontaminated. It is so named by the Hausa speaking natives of the northern Nigeria but its consumption transcends borders of ethnicity, tribe or religion across Nigeria, West Africa and beyond [2, 3].
Despite the wide distribution of this delicacy, little or no attention is paid to its safety, quality and hygiene; hence the possible occurrence of food borne disease [4, 5]. It is fairly recent that studies probing the microbiological and public health integrity of Suya began to emerge [2, 3, 6, 7].
The presence of E. coli in foods that are ready for consumption is indicative of poor hygiene and contamination could have arisen from human or animal faecal sources [2]. Most non-pathogenic but virulent strains of E. coli which causes several kinds of ailments, including food-borne infections have been identified [8]. E. coli strains that are widely resistant to commonly prescribed antibiotics have been reported [6, 9]. This study examined the antibiotic resistance profile of E. coli isolated from Suya sold in Karu Local Government Area of Nasarawa State, Nigeria.
Study Area
This study was carried out at Karu Local Government Area of Nasarawa State, Nigeria in May, 2020. It is located between latitudes 8� 5´ N and 10� 42´ N and longitudes 9� 25´E and 7�54´E of the Greenwich Meridian covering a spatial extent of about 2,640 km² with a population of 205,477 [10]. It shares its western, northern, eastern and southern boundaries with Abuja - the Federal Capital Territory, Kaduna state, Keffi and Nasarawa local government areas of Nasarawa state respectively [11]. The major towns in Karu LGA are; Mararaba, One Man Village, Ado, New Nyanya, Nyanya Gwandara and Masaka.
Sample collection
Three hundred (300) Suya samples were randomly purchased at Masaka, Ado, Mararaba, New Nyanya, Nyanya Gwandara and One Man Village (50 samples from each location). The samples were aseptically wrapped and labelled with date, time of sampling, area of sampling in a foil paper and transported to the Microbiology Laboratory of Nasarawa State University, Keffi, Nigeria for analysis.
Isolation and identification of Escherichia coli
Primary culture was prepared by aseptically inoculating 1 g of the Suya sample in 10 ml of nutrient broth and incubated at 37°C for 24 hrs. To obtain pure cultures, samples from the primary culture were sub-cultured on Levine Eosin Methylene Blue (EMB) Agar (HiMedia, India) plates by streaking and incubated at 37°C. The plates were observed after 24 hrs incubation; greenish metallic sheen indicates the presence of Escherichia spp [12]. API 20E (BiomerieuxTM) kit was used for biochemical identification of E. coli following manufacturer´s instructions.
Antibiotics susceptibility test
The antibiotics susceptibility test of the E. coli isolates was carried out using Kirby-Baeur disk diffusion method. The antibiotic disks were firmly placed on the sterile Mueller Hinton Agar (MHA) plates, seeded with test organisms standardized to 0.5 McFarland´s turbidity and incubated at 37 oC for 24 hrs. Diameter of zones of inhibition was then measured to the nearest millimetre and reported in accordance with the antimicrobial susceptibility breakpoint of CLSI [13].
Determination of Multiple Antibiotic Resistance (MAR) Index
The MAR Index was determined according to the method of Krumperman [14] and Paul et al. [15]. From the result of the antibiotic susceptibility test, MARI was calculated as:
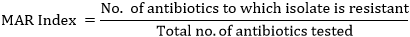
Data analysis
Data obtained was entered into Microsoft ExcelTM 2016 and subsequently SPSS v25 for statistical computation. The difference in the number of the bacterial isolates was compared from the different locations using p < 0.05 as the threshold for statistical significance.
This study determined the presence of E. coli in Suya sold in Karu local government area of Nasarawa state, Nigeria. Out of 300 samples obtained for this study, E. coli was isolated from 40 out of the 300 samples which is equivalent to a 13.3% isolation rate. The isolation rate from major towns within the study area is outlined in Table 1. The antibiotic resistance profile in this study indicates that all 40 E. coli isolated from Suya within Karu, Nasarawa state was resistant to ampicillin. Out of the ten antibiotics tested (i.e. Nitrofurantoin, Imipenem, Streptomycin, Ciprofloxacin, Gentamycin, Ceftriaxone, Cotrimoxazole, Ampicillin, Amoxicillin/Clavulanic acid and Cefixime), resistance levels = 75% was observed against all but one of the antibiotics. Only 5% resistance was recorded against gentamycin (Figure 1).
The antibiotics resistance phenotypes and the multiple antibiotics resistance index (MARI) observed in this study are presented in Table 2. MARI = 0.5 was observed which implies that; all the isolated E. coli from Suya sold in Karu local government area of Nasarawa state, Nigeria are resistant to at least five out of the 10 antibiotics tested in this study. The resistance phenotypes with the highest rate of occurrence were F Ipm S Cip Cro Sxt Amc Cfm Amp 7(17.5%) and F Ipm S Cip Cro Amc Cfm Amp 7(17.5%). Thirty-nine (97.5%) of the E. coli isolates were of the multidrug resistance (MDR) class, while 1(2.5%) of the isolates was pan-drug resistant (PDR) (Table 3).
In this study, 40 (13.3%) E. coli isolates were recovered from 300 Suya sampled at Karu Local Government Area of Nasarawa. The occurrence of E. coli from major towns of the local government area was Mararaba (16.1%) followed by New Nyanya (16.0%), Masaka (15.4%), Ado (12.5%), One Man Village (10.9%), and Nyanya Gwandara (8.0%). The isolation of E. coli from the Suya samples in this area suggests poor handling the meat, poor sanitation practices and lack of education among food handlers which if not controlled might constitute risk for food borne illness. Other possible factors could be due to the dusty nature of the area where meat shops are located or Suya displayed on tables with no wire mesh or net protecting it from flies.
This finding corroborates previous reports which found spiced, ready-to-eat meat products to be bacterial-laden [2, 3, 6, 7, 16, 17]. Magwira et al. [18] in Botswana reported that E. coli isolated from meat had occurrences of 2.3% whereas, 3.8% occurrence of E. coli was reported by of Abd El-Atty and Meshref [19] in Egypt, with a prevalence of 2.0% in sausage which was attributed to contamination from faeces of infected animals as well as unsatisfactory hygienic measures during handling. The lowest occurrence recorded at Nyanya Gwandara might be due to the lower population in that area as theorized by Ologhobo et al. [20], or it may be that most Suya vendors warm the product before serving their customers.
In this study, the E. coli had high rates of resistance to most of the tested antibiotics with the exception of Gentamycin which had only 5.0% resistance. The high antibiotic resistance observed could be that they are the most commonly available antibiotics used for treatment as well as growth promoters and in routine chemoprophylaxis among livestock as speculated by Olatoye [21]. This portends a major challenge in both human and animal medicine because these drugs are commonly used in the treatment of common human ailments.
All the isolates in this study were Multiple Antibiotic Resistant (MAR) with MAR Index of = 0.5 and the most frequent MAR Index was 0.8 with occurrence of 37.5%. Most of the E. coli isolates were Multi Drug Resistance (MDR) with the order of occurrence as: MDR (97.5%) > PDR (2.5%) >XDR (0.0%) and NMDR (0. 0%). This shows that the indiscriminate use of antibiotics in this environment may eventually supersede the drug sensitive microorganisms from an antibiotic saturated environment [22]. This is worrisome as Mishra et al. [23] asserted that MAR index of 0.4 and above is associated with human faecal source contamination and therefore sensitivity patterns and treatment should be guided by laboratory investigations.
This study evaluated a total of 300 Suya samples and 40 E. coli were isolated and identified using standard microbiological techniques. The overall isolation and occurrence of E. coli was 13.3% with the highest from Mararaba (16.1%) while Nyanya Gwandara had the least with 8.0%. The isolates were most resistant to Ampicillin (100%) while Gentamycin had the least with 5% resistance. The multiple antibiotic resistance pattern of the isolates showed that most of the E. coli were resistant to more than five antibiotics, hence exhibited Multi-Drug Resistance (MDR) strains with the order of occurrence as: Multi-Drug Resistance, MDR (97.5%) >Pan Drug Resistance, PDR (2.5%) > Non-Multi-Drug Resistance, NMDR (0.0) and Extensive Drug Resistance, XDR (0.0). All the isolates showed Multiple Antibiotic Resistance (MAR) with MAR Index of ≥ 0.5 and the most frequent MAR Index was 0.8 with occurrence of 37.5%. These findings indicate the possibility of the dissemination of multidrug resistant food-borne pathogens such as diarrhoeagenic E. coli within the studied locality. It is recommended that proactive measures should be taken by the government and civil society organizations to educate the meat vendors and the general populace on proper hygienic handling Suya. The public should also be educated on the proper usage of antibiotics in order to reduce the continuous distribution of multidrug resistance.
What is known about this topic
- Escherichia coli is a regular foodborne pathogen;
- Antibiotics resistance by E. coli to frequently prescribed antibiotics is common phenomenon.
What this study adds
- In this study, 13% occurrence of E. coli was found in barbecued beef (Suya) which is ready for consumption in Karu area of Nasarawa state, central Nigeria;
- Ninety-seven-point five percent (97.5%) of the E. coli isolated from the Suya are multidrug resistant.
The authors declare no competing interests.
Danladi Walong Datok: conceived and designed the experiments, performed the experiments, analyzed and interpreted the data. David Ishaleku: analyzed and interpreted the data, contributed reagents, materials, analysis tools or data. Paul Alumbugu Tsaku: wrote the manuscript, analyzed and interpreted the data. Elisha Obakas Agya: performed the experiments. Moses Peter Adoga: wrote the initial draft of manuscript, contributed reagents, materials, analysis tools or data. All the authors have read and approved the final version of the manuscript.
We acknowledge the valuable contributions by the entire laboratory staff of the Department of Microbiology, Nasarawa state university, Keffi, Nigeria.
Table 1: occurrence of Escherichia coli from Suya samples in relation to locations
Table 2: antibiotic resistance phenotypes of Escherichia coli isolated from Suya sold in Karu Local Government Area of Nasarawa State, Nigeria
Table 3: antibiotic resistance classes of Escherichia coli isolated from Suya sold in Karu Local Government Area of Nasarawa State, Nigeria
Figure 1: antibiotic resistance profile of Escherichia coli isolated from Suya sold in Karu Local Government Area of Nasarawa State, Nigeria
- CooksInfo. Roasting Meat. CooksInfo. Accessed 30 April 202.
- Adesoji AT, Onuh JP, Musa AO, Akinrosoye PF. Bacteriological qualities and antibiogram study of bacteria from. The Pan African Medical Journal. 2019;33(219). doi:10.11604/pamj.2019.33.219.17729. PubMed | Google Scholar
- Jonathan SG, Adeniyi MA, Asemoloye MD. Fungal Biodeterioration, Aflatoxin Contamination, and Nutrient Value of “Suya Spices”. Scientifica. 2016;2016:4602036. PubMed | Google Scholar
- Okonkwo I, Sunday M, Emeka C. Microbiological Safety and Proximate Composition of Suya Stored at Ambient Temperature for Six Hours from Maiduguri, Northern Nigeria. Internet Journal of Food Safety. 2012;14:11-16. PubMed | Google Scholar
- Nyenje ME, Odjadjare CE, Tanih NF, Green E, Ndip RN. Foodborne Pathogens Recovered from Ready-to-Eat Foods from Roadside Cafeterias and Retail Outlets in Alice, Eastern Cape Province, South Africa: Public Health Implications. Int J Environ Res Public Health. 2012;9(8):2608-2619. PubMed | Google Scholar
- Eze AE, Eze NC, Amaeze OV, Eze CN. Fishes and smoked meat delicacies as sources of multidrug resistant bacteria and parasitic worms. AJAR. 2013;8(22):2799-2805. PubMed | Google Scholar
- Unachukwu MN, Momoh OR, Nwakanma C. Bacteriological examination of Suya meat sold in Enugu Metropolis. World Journal of Pharmaceutical Research. 2018;4(12):61-70. Google Scholar
- Yun Z, Zeng L, Huang W, Wu Q, Fan Y, Zheng S et al. Detection and Categorization of Diarrheagenic Escherichia coli with Auto-microfluidic Thin-film Chip Method. Scientific Reports. 2018;8(1):12926. PubMed | Google Scholar
- Tsaku PA, Ngwai YB, Pennap GRI, Ishaleku D, Ibrahim T, Nkene IH et al. Extended-Spectrum Beta-Lactamase-production in Escherichia coli isolated from door handles in Nasarawa State University, Keffi, Nigeria. Heliyon. 2019;5(8). doi:10.1016/j.heliyon.201e02177. PubMed | Google Scholar
- National Bureau Of Statistics. National Bureau Of Statistics. Accessed 1 May 2021.
- NAGIS. MAPS. Accessed 1 May 2021.
- Cheesbrough M. District Laboratory Practice in Tropical Countries. Leiden. Cambridge University Press. 2006. Accessed 1 May 2020.
- Patel JB. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. 2017.
- Krumperman PH. Multiple antibiotic resistance indexing of Escherichia coli to identify high-risk sources of fecal contamination of foods. Appl Environ Microbiol. 1983;46(1):165-170. PubMed | Google Scholar
- Paul S, Bezbaruah RL, Roy MK, Ghosh AC. Multiple antibiotic resistance (MAR) index and its reversion in Pseudomonas aeruginosa. Letters in Applied Microbiology. 1997;24(3):169-171. PubMed | Google Scholar
- Tambekar DH, Jaiswal VJ, Dhanorkar DV, Gulhane PB, Dudhane MN. Identification of microbiological hazards and safety of ready-to-eat food vended in streets of Amravati City, India. Journal of Applied Biosciences. 2008;7:7. Google Scholar
- Egbebi AO, Seidu KT. Microbiological evaluation of Suya (dried smoked meat) sold in Ado and Akure, South West Nigeria. European Journal of Experimental Biology. 2011;1(4).
- Magwira CA, Gashe BA, Collison EK. Prevalence and Antibiotic Resistance Profiles of Escherichia coli O157:H7 in Beef Products from Retail Outlets in Gaborone, Botswana. Journal of Food Protection. 2005;68(2):403-406. PubMed | Google Scholar
- Abd El-Atty NS, Meshref AMS. Prevalence of Salmonella and E.coli O157 in some foods. Journal of Veterinary Medical Research. 2008;18(1):73-78.
- Ologhobo AD, Omojola AB, Ofongo ST, Moiforay S, Jibir M. Safety of street vended meat products - chicken and beef Suya. African Journal of Biotechnology. 2010;9(26):4091-4095. Google Scholar
- Olatoye, Isaac Olufemi. The incidence and antibiotics susceptibility of Escherichia coli O157:H7 from beef in Ibadan Municipal, Nigeria. Afr J Biotechnol. 2010;9(8):1196-1199. Google Scholar
- Al-Kobaisi MF Jawetz. Melnick & Adelberg´s Medical Microbiology. Sultan Qaboos Univ Med J. 2007;7(3):273-275.
- Mishra M, Patel AK, Behera N. Prevalence of Multidrug Resistant E. coli in the river Mahanadi of Sambalpur. current Research in Microbiology and Biotechnology. 2013;5: 239-244.