Anti-glomerular basement membrane disease with intense nephrotic syndrome: a new case report
Ikram Mami, Emna Ghzel, Rym Abida, Fadwa Hlaoui, Hela jebali, Lamia Rais, Soumaya Beji, Fethi Ben Hamida, Lilia Ben Fatma, Karim Zouaghi
Corresponding author: Ikram Mami, Department of Nephrology, Dialysis and Kidney Transplantation, La Rabta Hospital, Tunis, Tunisia 
Received: 21 Jun 2021 - Accepted: 05 Aug 2021 - Published: 16 Aug 2021
Domain: Nephrology
Keywords: Anti-glomerular, basement membrane disease, nephrotic syndrome, proteinuria, case report
©Ikram Mami et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Ikram Mami et al. Anti-glomerular basement membrane disease with intense nephrotic syndrome: a new case report. Pan African Medical Journal. 2021;39:243. [doi: 10.11604/pamj.2021.39.243.30461]
Available online at: https://www.panafrican-med-journal.com//content/article/39/243/full
Case report 
Anti-glomerular basement membrane disease with intense nephrotic syndrome: a new case report
Anti-glomerular basement membrane disease with intense nephrotic syndrome: a new case report
![]() Ikram Mami1,2,&, Emna Ghzel1,2,
Ikram Mami1,2,&, Emna Ghzel1,2,![]() Rym Abida2,3, Fadwa Hlaoui1,2, Hela Jebali1,2, Lamia Rais1,2, Soumaya Beji1,2,
Rym Abida2,3, Fadwa Hlaoui1,2, Hela Jebali1,2, Lamia Rais1,2, Soumaya Beji1,2, ![]() Fethi Ben Hamida2,4, Lilia Ben Fatma1,2, Karim Zouaghi1,2
Fethi Ben Hamida2,4, Lilia Ben Fatma1,2, Karim Zouaghi1,2
&Corresponding author
Anti-glomerular basement membrane (anti-GBM) disease was usually described as a small vessel vasculitis presenting with acute kidney injury, haematuria and non-nephrotic proteinuria. We report a case of anti-GBM disease revealed by an intense nephrotic syndrome. The urinary protein level was 12g/day. Renal biopsy only showed crescent glomerulonephritis with linear staining of IgG in direct immunofluorescence without other glomerulonephritis. Immunoglobulin G (IgG) anti-GBM antibody titer was elevated.
Anti-glomerular basement membrane (anti-GBM) disease is a rare condition causing renal and/or alveolar damage [1]. It is an auto-immune condition triggered by immune complexes formed by immunoglobulin G (IgG) auto-antibodies against the alpha 3 chain of type IV collagen [1]. Renal manifestations of this disease typically include hematuria, non-nephrotic proteinuria and rapidly progressive renal failure [1]. Clinical presentation of anti-GBM disease by nephrotic syndrome is rare. We hereby present a case of anti-GBM disease revealed by an intense nephrotic syndrome.
Patient information: a 25-year-old male patient, smoker, without previous exposure to toxic substances, coming from a rural area, was admitted to the nephrology department for a nephrotic syndrome that was discovered after a pulmonary infection.
Clinical findings: clinical examination found a general state without fever. Edema was important. Arterial pressure was 160/90mmHg. Urine dipstick analysis showed positive hematuria and 3 marks of proteinuria. Pulmonary examination was normal and so was the rest of the clinical assessment.
Diagnostic assessment: laboratory tests revealed a renal failure with a creatinine level at 274μmol/l, an intense nephrotic syndrome with serum protein level at 38g/l, serum albumin level at 18g/l and urinary protein level of 12g/24 hours, as well as a microcytic anemia at 8.7g/dl. Chest radiography was normal. Renal ultrasonography found kidney measurements within normal range with preserved corticomedullary differentiation and contours. Considering all the information above, our patient had an impure nephrotic syndrome with an anemia inadequately proportional to the level of the renal failure. Perinuclea anti-neutrophil cytoplamic (p ANCA), cytoplasmic anti-neutrophil cytoplasmic (c ANCA) antibodies and antinuclear antibodies (AAN) were negative. Serum complement is normal. However, an Immunoglobulin (IgG) anti-GBM antibody titer was elevated. Renal pathology of 25 glomerulus found large cellular crescents in 17 glomeruli: circumferential in 11 glomeruli and circumscribed in 7 glomeruli, without rupture of Bowman´s membrane (Figure 1). There were also segmental sclerotic lesions with flocculo-capsular apposition in 2 glomeruli alongside with extensive tubular necrosis lesions. Linear staining of IgG were found in direct immunofluorescence. Moreover, anti-GBM antibodies were positive. Diagnosis of anti-GBM disease was confirmed. Computed Tomography showed alveolar hemorrhage.
Therapeutic intervention: treatment was immediately initiated: one gram per day of intravenous bolus methylprednisolone for 3 days relayed by oral prednisone intake at a dosage of 1mg/kg/d, associated with plasmapheresis as 6 intravenous boli of 500mg of cyclophosphamide: every 2 weeks for a month then every 3 weeks.
Follow-up and outcomes: pulmonary outcome was favorable. Anti-GBM antibodies were negative after 12 sessions of plasmapheresis. However, renal function had dramatically decreased leading to a terminal chronic kidney disease and periodic hemodialysis within one month.
Anti-GBM disease is a small vessel vasculitis involving capillaries of the kidneys and the lungs. It classically characterized by rapidly progressive glomerulonephritis, associated or not with intra-alveolar hemorrhage (Goodpasture syndrome) [2]. The disease has a bimodal distribution as it is predominately noted within young males and older females. Smoking seems to be the main risk factor [3]. Our observation is distinctive by the existence of an intensive nephrotic syndrome with extensive proteinuria ranging at 12g/24 hours. Such nephrotic syndrome had rarely been described in the course of good pasture disease. Few cases were reported in the literature with documented nephrotic range proteinuria [4-7] (Table 1). Moreover, larger studies had confirmed the low frequency of nephrotic syndrome in goodpasture [8,9]. The various studies sought to find an explanation for this association between the GoodPasture syndrome and nephrotic syndrome. It has been previously described that anti-GBM disease with nephrotic syndrom can be associated to others glomerulonephritis such as membranous nephropathy (MN) and minimal change disease [10]. However, simultaneous anti-GBM disease and MN were the association the most described in the literature [10]. Association of anti-GBM glomerulonephritis and MN was explained by of immune complex deposits in the sub-epithelial space [8]. In our presentation, nephrotic syndrome could not be explained by renal biopsy results subject to an electron microscopy study. Standard treatment for anti-GBM disease is aggressive, including plasmapheresis along with cyclophosphamide and corticosteroids [2]. Zhong et al. also reported a case report of anti-GBM disease with nephrotic syndrome treated by Tacrolimus with partial remission [6]. Renal progression was unfavourable and the diagnosis of the terminal stage was retained with initiation of hemodialysis after one month.
Goodpasture syndrome is to be called to mind, even in cases of heavy proteinuria. This highlights the important role of renal biopsy in the differential diagnosis of such unusual clinical presentations and the importance of histological study of the electron microscope to reveal podocytopathies associated with Goodpasture syndrome.
The authors declared no competing interests.
Data collection: Rym Abida, Fadwa Hlaoui. Writing of manuscript: Ikram Mami, Emna Ghzel. Interpretation of analysis and renal biopsy: Lamia Rais, Hela jebali, Karim Zouaghi. Revision of manuscript: Lilia Ben Fatma, Fethi Ben Hamida, Soumaya Beji. All the authors have read and agreed to the final manuscript.
Table 1: anti-glomerular basement membrane disease with nephrotic syndrome: summary of literature
Figure 1: A) renal biopsy, Masson's trichrome (x200), circumferential cellular crescent; B) renal biopsy, Masson's trichrome (x200), capillary fibrinoid necrosis; C) renal biopsy, immunofluorescence (x200); linear anti-IgG antibody staining along the GBM; D) computed tomography showed diffuse alveolar hemorrhage
- Greco A, Rizzo MI, De Virgilio A, Gallo A, Fusconi M, Pagliuca G et al. Goodpasture's syndrome: a clinical update. Autoimmun Rev. 2015 Mar;14(3):246-53. PubMed | Google Scholar
- McAdoo SP, Pusey CD. Anti-Glomerular basement membrane disease. Clin J Am Soc Nephrol. Clin J Am Soc Nephrol. 2017 Jul 7;12(7):1162-1172. PubMed | Google Scholar
- Hellmark T, Segelmark M. Diagnosis and classification of Goodpasture's disease (anti-GBM). J Autoimmun. Feb-Mar 2014;48-49:108-12. PubMed | Google Scholar
- Qunibi WY, Taylor K, Knight TF, Senekjian HO, Györkey F, Weinman EJ. Nephrotic syndrome in anti-GBM antibody mediated glomerulonephritis. South Med J. 1979 Nov;72(11):1396-8. PubMed | Google Scholar
- Okafor CC, Balogun RA, Bourne DT, Alhussain TO, Abdel-Rahman EM. An unusual case of anti-glomerular basement membrane disease presenting with nephrotic syndrome. Int Urol Nephrol. 2011 Dec;43(4):1249-53. PubMed | Google Scholar
- Zhong Z, Tan J, Tang Y, Li Z, Qin W. Goodpasture syndrome manifesting as nephrotic-range proteinuria with anti-glomerular basement membrane antibody seronegativity: a case report. Medicine (Baltimore). 2020 Sep 25;99(39):e22341. PubMed | Google Scholar
- Shibata Y, Fukuoka K, Yokota R, Lee H, Sayo H, Ikegaya N et al. Nephrotic syndrome due to minimal-change disease superimposed on anti-glomerular basement membrane antibody positive glomerulonephritis; a case report. BMC Nephrol. 2020 Jul 17;21(1):283. PubMed | Google Scholar
- Nasr SH, Collins AB, Alexander MP, Schraith DF, Herrera Hernandez L, Fidler ME et al. The clinicopathologic characteristics and outcome of atypical anti-glomerular basement membrane nephritis. Kidney Int. 2016 Apr;89(4):897-908. PubMed | Google Scholar
- Shen CR, Jia XY, Cui Z, Yu XJ, Zhao MH. Clinical-pathological features and outcome of atypical anti-glomerular basement membrane disease in a large single cohort. Front Immunol. 2020 Sep 3;11:2035. PubMed | Google Scholar
- Ogawara A, Harada M, Ichikawa T, Fujii K, Ehara T, Kobayashi M. Co-existence of Anti-Glomerular basement membrane glomerulonephritis and membranous nephropathy in a female patient with preserved renal function. Tohoku J Exp Med. 2017 Dec;243(4):335-341. PubMed | Google Scholar
Search
This article authors
On Pubmed
On Google Scholar
Citation [Download]
Navigate this article
Similar articles in
Key words
Tables and figures
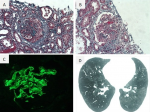 Figure 1: A) renal biopsy, Masson's trichrome (x200), circumferential cellular crescent; B) renal biopsy, Masson´s trichrome (x200), capillary fibrinoid necrosis; C) renal biopsy, immunofluorescence (x200); linear anti-IgG antibody staining along the GBM; D) computed tomography showed diffuse alveolar hemorrhage
Figure 1: A) renal biopsy, Masson's trichrome (x200), circumferential cellular crescent; B) renal biopsy, Masson´s trichrome (x200), capillary fibrinoid necrosis; C) renal biopsy, immunofluorescence (x200); linear anti-IgG antibody staining along the GBM; D) computed tomography showed diffuse alveolar hemorrhage











