Reactive mass vaccination campaign against cholera in the COVID-19 context in Cameroon: challenges, best practices and lessons learned
Adidja Amani, André Arsène Bita Fouda, Armanda Jeanne Nangmo, Solange Ngo Bama, Collins Asaah Tatang, Marie Angèle Mbang, Emmanuel Christian Epee Douba, Aimé Gilbert Mbonda Noula, Mariette Dia, Malika Bouhenia
Corresponding author: Adidja Amani, Sub-Directorate of Vaccination, Directorate of Family Health, Ministry of Public Health, Yaoundé, Cameroon 
Received: 08 Jan 2021 - Accepted: 05 Apr 2021 - Published: 22 Apr 2021
Domain: Infectious diseases epidemiology,Vaccine cold chain,Vaccine logistic
Keywords: Challenges, practices, lessons learned, cholera, vaccination, Cameroon, COVID-19
© Adidja Amani et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Adidja Amani et al. Reactive mass vaccination campaign against cholera in the COVID-19 context in Cameroon: challenges, best practices and lessons learned. Pan African Medical Journal. 2021;38:392. [doi: 10.11604/pamj.2021.38.392.27754]
Available online at: https://www.panafrican-med-journal.com//content/article/38/392/full
Research 
Reactive mass vaccination campaign against cholera in the COVID-19 context in Cameroon: challenges, best practices and lessons learned
Reactive mass vaccination campaign against cholera in the COVID-19 context in Cameroon: challenges, best practices and lessons learned
Adidja Amani1,&, André Arsène Bita Fouda2, ![]() Armanda Jeanne Nangmo3, Solange Ngo Bama4,
Armanda Jeanne Nangmo3, Solange Ngo Bama4, ![]() Collins Asaah Tatang1, Marie Angèle Mbang1, Emmanuel Christian Epee Douba5, Aimé Gilbert Mbonda Noula6, Mariette Dia7, Malika Bouhenia8
Collins Asaah Tatang1, Marie Angèle Mbang1, Emmanuel Christian Epee Douba5, Aimé Gilbert Mbonda Noula6, Mariette Dia7, Malika Bouhenia8
&Corresponding author
Introduction: since 1971, Cameroon is facing a growing series of cholera epidemics despite all the efforts made by the government to address this huge public health threat. In 2020, in addition to the COVID-19 pandemic, Cameroon recorded a high cholera case fatality rate of 4.3% following epidemics noted in the South, Littoral and South-West regions. The Cameroon Ministry of Public Health, has thus organized a reactive vaccination campaign against cholera to address the high mortality rate in the affected health districts of those regions. The objective of this study was to describe the challenges, best practices and lessons learned drawing from daily experiences from this reactive vaccination campaign against cholera.
Methods: we conducted a cross-sectional study drawn from the results of the campaign. We had a target population of 631,109 participants aged 1 year and above resident of the targeted health areas.
Results: the overall vaccination coverage was 64.4% with a refusal rate ranging from 0-10% according to health districts. Vaccination coverage was the lowest among people aged 20 years and above. The main challenge was difficulty maintaining physical distanciation, the main best practice was the screening of all actors taking part at the vaccination against COVID-19 and we found that emphasizing on thorough population sensitization through quarter heads and social mobilizers and adequately programming the campaign during a good climate season is crucial to achieving good vaccination coverage.
Conclusion: lessons learned from this study could serve to inform various agencies in the event of planning rapid mass vaccination programs during pandemics.
Cholera is a human diarrheal disease endemic in Africa, caused by the toxins of the bacteria called Vibrio cholera [1-3]. It is manifested by watery diarrhea with a rice-white appearance emitting a peculiar odor which dehydrates the patient and is lethal if proper care is not provided promptly [4,5]. This disease continues to wreak havoc in poor countries with limited resources, including Cameroon, where access to drinking water remains a real and growing challenge for some populations.
The spread of cholera is dependent on numerous environmental and biological variables, including seasonal environmental drivers, host immunity, the infectivity of the bacteria and lytic bacteriophages [6]. According to Andrew Azman´s systematic review that was carried out in 2013, toxigenic cholera median incubation period is 1.4 days with 5% of cases symptoms by 0.5 days and 95% by 4.4 days [7]. Cholera epidemics are expected to kick off during episodes of prevailing warm air temperature with stagnant river flows, creating favorable environmental conditions for the growth of cholera bacteria [8]. Heavy rainfall, through floods and/or breakdown of sanitary infrastructures, accelerates interaction between contaminated water and human activities, resulting in an epidemic [8]. Also, in some contexts like Cameroon, there are risk factors like extreme poverty and promiscuity where transmission occurs through foods, water and close contact which favour an epidemic to set in [1].
Despite inadequate cholera cases reporting due to surveillance systems failings [9-11], in 2015, the global burden of cholera was estimated at 2.86 million (ranging from 1.4 to 4 million) of cases resulting in 95000 deaths (with significant differences in estimates ranging from 21,000 to 143,000) [10]. Even though all regions of the world are affected by cholera, in 2014, sub-Saharan Africa (SSA) remained the most affected continent with approximately 90% of cholera cases [12]. In 2019, 13 African countries reported a total of 55,087 cases and 872 deaths, corresponding to a case fatality rate (CFR) of 1.6% with Cameroon bearing the highest CFR [13]. Cameroon reported its worst outbreak in 2011 (22,762 suspected cases and 786 deaths) [14] since the first case of cholera was registered on February 4, 1971 [15,16]. The annual case fatality rates are exhibiting steep upward trend and remain consistently well above the acceptable World Health Organization (WHO) value of 1%. In 2018, Cameroon had 997 suspected cases and 58 deaths yielding a CFR of 5.8% [17]. In 2019, 478 suspected cases and 19 deaths (CFR of 4.0%) [18] and during the 40th epidemiological week of 2020, there were 1,848 suspected cases and 80 deaths yielding a CFR of 4.3%.
Based on the cholera risk assessment conducted by the WHO in 2018 in Cameroon, these regions fulfil all the necessary conditions for a reactive vaccination campaign. First, some health areas in these regions face a serious lack of safe water and poor hygienic conditions, as well as poor access to health care services. Second, as the South-West region has suffered a socio-political crisis for four consecutive years, life insecurity another prominent factor. Third, the Londji Health Area (South region) comprises mainly islets with no access to latrines and its population consists primarily of fishermen. This area is also characterized by a large influx of surrounding population in need of fish. Thus, poor hygiene coupled with unfavourable living conditions, poor drinking water coverage and life insecurity pose a serious risk to the local population. Moreover, owing to the contiguity between the Littoral and the South-West region, residents of these three regions are particularly vulnerable to the spreading of a cholera epidemic.
Since the beginning of COVID-19 pandemic, the WHO has issued guidelines indicating that mass vaccination campaigns should cease until the COVID-19 situation resolves [19,20]. However, given the sanitary risks to which some Cameroonians localities are exposed and considering the decision-making framework for the implementation of mass vaccination campaigns in the context of COVID-19 [21], a reactive mass vaccination campaign against cholera was deemed necessary in the Littoral, South and South-West regions. The organization of this campaign is part of an integrated approach, complementary to contributing to ending cholera global 2030 roadmap strategy [22] and to all other cholera interventions implemented in the COVID-19 context by the ministry of public health, in collaboration with technical and financial partners.
Besides, even though prompted by the current COVID-19 situation in the world, the WHO and the Cameroon Ministry of Public Health prescribed hygienic measures and restricted movement, the resurgence of cholera outbreaks represents a double dividend of health risks to certain populations in Cameroon. Against this backdrop, the objective of this article is to describe vaccination coverage and discuss challenges, lessons learned and best practices for carrying out a mass vaccination campaign against cholera during COVID-19 with the overarching goal of informing global health organizations and leaders on the campaign outcomes.
Study design: this was a cross-sectional study employing the results of the reactive mass vaccination campaign against cholera carried out from August to September 2020. It was conducted in some health areas (HAs) of cholera affected health districts (HDs) of the Littoral, South and South-West regions of Cameroon which recorded at least one cholera case from March to August 2020.
Study setting: the study was conducted in the following HDs and HAs. In the Littoral region, we had the HD of Bonassama (Mabanda and Bonassama HAs), New-Bell (Nkolouloun, Sebenjongo, Mbam Ewondo, New-Bell Bamiléké, Makea and Youpwe HAs), Nylon (Soboum) and Japoma (Bwang). In the South region, we had the Kribi HD (Londji, Adjap, Grand-Batanga, Hevecam and Kribi HAs). In the South-West region Limbe HD (Mabeta HA) and Tiko HD (Holforth, Kange, Likomba, Missellele, Mondoni, Mudeka, Mutegene, Tiko Town HAs). It is worth noting that, a vaccination campaign was also carried out in Manoka HD (Littoral region) in February 2020 but these data are not included in this paper as it was not carried out during the COVID-19 pandemic in Cameroon.
Study target population: this study was conducted among people of both sexes, aged one year and above, including pregnant women. Health areas target population was drawn from the Cameroon Ministry of Public Health 2020 target populations (Table 1).
Leadership and planning: to carry out this vaccination campaign, a preparation plan was put in place. Preparatory meetings at the central and regional level were held after approval by the Cameroon Ministry of Public Health. The WHO has declared its intention to fund the operational costs of the campaign. At the regional level, the following activities were conducted: advocacy with the administration, traditional and religious authorities; raising awareness of worshippers in places of prayer, messages to key sectors and community organizations; production of sensitization materials such as banners and posters; briefing meetings between the public health regional delegates and their staff, central supervisors, district executive teams and partners on the organization of the campaign. A coordinating committee at the regional level and in the Health districts was put together.
Oral cholera vaccines (OCV) procurement: Shanchol™ and Euvichol-PlusR were the two bivalent OCV used during this campaign. A total of 1,168,996 doses of Schanchol™ and Euvichol-PlusR were made available by the International Coordinating Group (ICG) emergency stockpile. The WHO facilitated the availability of operational costs for the campaign. Besides, doctors without borders helped with logistics in Kribi HD. Shanchol™ was used in the Littoral region and Euvichol-PlusR in the South and South-West regions.
Cold chain and waste management: vaccines were transported from the central cold room to the 3 regional cold rooms because many of these HDs/HAs did not have an adequate cold chain system. Arrangements were made for adequate storage of vaccines at the local level between +2°C to +8°C until they were administered as recommended by the manufacturers.
Staff training: after briefing the district senior teams at the public health regional delegations, social mobilizers and vaccinators were trained. The training was conducted by pools with the support of central supervisors and WHO partners to respect physical distanciation. In total, 2271 people were trained for the campaign; 1482 persons, 489 persons and 300 persons in the Littoral, South-West and South regions respectively.
COVID-19 screening: before being enrolled as a social mobilizer or a vaccinator, each volunteer had to be screened for COVID-19 using the Rapid Diagnostic Test SD BIOSENSOR COVID-19 Ag to avoid infecting others during the vaccination campaign against cholera.
Data analysis: data were collected through vaccination tally sheets. The treatment and analysis of data was done through Excel version 2013.
Social mobilization: as part of community sensitization, the objective was to inform at least 95% of household´s heads before the visit of vaccination teams.
Littoral region: we can observe that refusal cases were so many in the Littoral region (Table 2). The average was 7% ranging from 3% in the Nylon Health District to 10.28% in the Japoma Health District. The number of refusal cases we were not able to manage in this region was also high. In the Littoral region, the main refusal reasons were “undetermined causes” (66%) followed by “not being the person who decides” (13%) and then a dangerous vaccine (11%). According to the independent monitoring carried out by WHO partners the main information pathway was through social mobilization (71%).
South-West region: in the South-West region for instance, the main refusal causes were “being completely vaccinated” (51%) and “undetermined causes” (46%). The main pathway of information was griots (43%) followed by social mobilization (37%).
South region: the refusal rate in the South region is 1.2%. The main population information pathway was through social mobilization (26%) followed by the health personnel (19%).
Administrative vaccine coverage
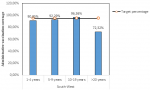
South-West region: these results reveal the different oral cholera vaccination coverage in the affected health areas of the South-West region (Table 3). The overall vaccination coverage in the South-West region was 81.97% but greatly varying across age ranges and health areas. Only two health areas in the South-West region achieved an acceptable vaccination coverage >95%. There was a high vaccination coverage in Mabeta (Limbe Health District) which is a small health area of 6899 people because of ease of geographic access through roads as compared to health areas of Tiko District where you have to take canoe sometimes to reach certain health areas. Besides, in Limbe, they were seeing fresh cases of cholera dying around them, this also increased their fear of the disease and vaccine acceptance. In Figure 1, we can observe there was less than 80% vaccination coverage among populations aged 20 years and above. This might be due to the belief that vaccination is linked to children with some parents vaccinating their children and don´t get vaccinated themselves. Only the adolescent´s population achieved the target vaccination rate of at least 95%.
Littoral region: the overall coverage rate in the Littoral region was the lowest despite the huge target population. This might be due to the poor perception of cholera risk or to the inherent behaviour (doubtful character) of people living in the Littoral region. Secondly, this campaign was conducted entirely during August when there are torrential rainfalls in the Littoral region with trees falling on roads and preventing the passage of teams or cars. The latter prevented vaccination teams from going into the field. The initial vaccination schedule of 2 days was prolonged for 4 more days. Also according to the independent monitoring that was carried out by WHO, we also had a great number of missed households, with some stating they were not informed of the campaign. In the Littoral region, reasons for non-vaccination varied greatly across health districts. According to the WHO independent monitoring, the main reasons of non-vaccination in the Littoral region were other reasons (37%) followed by travel (31%), being in the market (18%), school (18%) farm and playing outside.
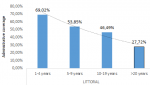
South region: the overall vaccination coverage was low (Figure 2). In the South region, the campaign started in the Londji health area. The vaccination coverage there was good in Londji because population accepted the vaccination because they had fresh dying cases of cholera in their locality. In other health areas, vaccination coverage was also poor because of the torrential rainfalls. This vaccination overall coverage is 64.43% which is far lower than the acceptable vaccination coverage of 85%. The Littoral and the South region follow the same distribution with children between 1-4 years having the highest coverage and adults >20 years having the lowest coverage rate (Figure 3). The South-West region faces a different distribution pattern with adolescents aged 10-19 years having the greatest vaccination.
Challenges, best practices, and lessons learned: WHO has recommended the use of hydro-alcoholic solutions and face masks for mass vaccination campaigns. The term personal protective equipment (PPE) in the following therefore refer to them.
Challenges
Difficulty to practice physical distanciation by the population: even though the predominant vaccination strategy was the door-to-door strategy, we had to set up fixed and temporarily fixed posts in some chiefdoms and health facilities. This favoured people coming together. It was therefore quite impossible to maintain physical distanciation.
Insufficient supply of hydro-alcoholic solutions and face masks for vaccination staff: in general, hydro-alcoholic solutions and face masks were distributed by regions according to their target population but the quantity given in some health areas was insufficient for the staff present. PPEs were distributed according to the target population of each health area.
Difficulty recruiting volunteers especially social mobilizers in the South-West: it was difficult to recruit volunteers´ especially social mobilizers in the South-West region considering the poor socio-political crisis that is ongoing in the region. People were afraid to wear anything concerning the government as it might put their lives at risk. Also, they were so much concerned about their health in the COVID-19 context.
High refusal rate among some populations: vaccine refusal was a great challenge in the Littoral region therefore threatening achievements made so far concerning immunization services. The high refusal rate might be firstly inherent to the Littoral´s population behaviour. People were also hesitant taking OCV thinking this was COVID-19 vaccine that was been administered to them as some teams wore protection gowns which were not recommended.
Heavy rainfalls: during the implementation of the campaign, there were heavy rainfalls in the Littoral and South regions which prevented the effective implementation of vaccination. Vaccination was scheduled for six days but teams were able to vaccinate only for two days therefore reducing our vaccination coverage.
The lack of adequate gadgets to identify vaccination teams: vaccinators and social mobilizers were not identifiable by their badges and their aprons. This undermined the social mobilization. Worse, wearing the combinations sued for the fight against COVID-19 fueled the rumor that it could be COVID-19 vaccine being administered.
Best practices
To carry out the first-ever oral cholera vaccination campaign in COVID-19 context despite restrictions: while we acknowledge WHO recommendations on differing mass vaccination campaigns until COVID-19 stops, we faced a critical cholera epidemic with several active districts in three regions of Cameroon in 2020. Therefore, organizing a reactive mass vaccination against cholera in those districts was mandatory. This campaign helped in stopping the epidemic in those health areas while preventing the spreading of COVID-19.
Screening of all actors and vaccination staff against COVID-19 using rapid diagnostic tests: right before the beginning of the campaign, all the staff involved in the campaign were screened against COVID-19. Above increasing the country testing rate, it also helped to prevent contamination from a staff to a vaccination participant. Fortunately, all of them were COVID-19 free at the moment of testing. Despite testing all actors at the beginning of the campaign, its implementation was not complete because no testing was done at mid-campaign and the end of the campaign. Considering the incubation period of COVID-19 ranges from 5 to 14 days, vaccination teams might have been exposed to either mild or severe COVID-19 cases during the campaign.
The availability of personal protective equipment´s (PPEs): despite being insufficient for all vaccination staff, all the health areas involved in the campaign were offered PPEs.
Training of vaccination staff by pools: in total, 2244 mobilizers and vaccinators were to be trained in the three regions for the campaign. The Cameroonian government said not more than 50 persons should be present in one room. We looked for schools that had several classes to train vaccination team´s classes after classes to maintain physical distanciation.
Lessons learned
An emphasis should be put on sensitization at least 2 weeks before the implementation of the campaign: during the campaign, we noticed populations were much more reluctant because they reported not being aware of an ongoing cholera vaccination campaign. This will allow a significant reduction in reluctance. The availability of a communication plan with emphasis on the involvement of quarter chiefs allow a deep population sensitization. Providing an incentive budget line for the community leaders tightly related to the population might largely influence the outcome of the vaccination campaign. At the beginning of the campaign, in the Littoral region (New-Bell health district), people were too reluctant to take the oral cholera vaccination, but after holding a meeting with the neighbourhood chiefs, the vaccination coverage steeply increased. So we believe motivating community leaders might have a great impact on the vaccinations coverage. This lesson can also help in educating populations on adopting COVID-19 hygienic measures by greatly involving also financially their community leaders.
Provision of water points: in addition to hydro-alcoholic solutions and face masks, we should either provide water points so that vaccinators can wash their hands frequently during a mass vaccination campaign or increase the quantity of hydro-alcoholic solutions.
Implementation of mass vaccination campaigns should be done in the dry season: carrying out a mass vaccination campaign in seaside towns might be challenging. From now on, we will organize our vaccination campaign during the dry season. This to permit effective implementation of the campaign and adequate management of resources.
This article describes vaccination coverage, challenges, best practices and lessons learned during the OCV vaccination coverage in the Littoral, South and South-West regions in Cameroon from August to September 2020. We observed a very low vaccination coverage of 64% overall. This coverage is far lower than that obtained in Guinea in 2012 and in South Sudan in 2016 [23,24]. This probably due to the poor climatic conditions during which the campaign was conducted. Unfortunately, this administrative vaccination coverage also greatly varied across regions: in the Littoral region (38.61%) as compared to the South (72.74%) and South-West (81.94%) regions. One of the reasons explaining this great disparity among regions might be the superiorly urban aspect of the Littoral region and the high density of its population. We observe that as a community grow in urbanity, its OCV vaccination coverage tends to decrease. These findings are comparable to what was obtained in Haiti in 2012 where they had 71% in rural Brocozel [25] and 69% in urban slums in Port au Prince [26].
We also observe an overall tendency of very low vaccination coverage among people aged 20 years and above. Similar findings were observed among people 15 years and above in Haiti [27], Mozambique [28], Bangladesh, India [29], Guinea [23]. Adults usually perceive vaccination as an issue for children and/or pregnant women, therefore it is usually difficult to see adults who make themselves available for vaccination. It is therefore mandatory to develop adult-specific strategies to improve vaccination coverage among adults. Even though insufficient, the availability of PPE was an essential aspect of our best practices as no action could have been taking without the above. These findings are the same as in a similar study carried out in Ethiopia in 2020 on implementing nation-wide measles supplemental immunization activities in the context of COVID-19 [30].
One major lesson learned in this campaign was that intensifying communication activity several weeks before the implementation could improve the vaccination uptake and therefore help the government to reach its goal of addressing mortality due to cholera. This is in line with data a similar oral vaccination campaign carried out in Zanzibar in 2012. They recommended announcing the upcoming of a campaign a few months before the start and also reminding the target health areas frequently [31]. One major weakness of our study was its highly descriptive aspect and no major statistical analysis was carried out due to its objective which was to describe only what was achieved during the vaccination campaign. This is just an informative article and there is therefore limited reproducibility.
The reactive mass vaccination campaign carried out in three regions of Cameroon in 2020 during COVID-19 pandemic was a success despite the high rate of refusal cases. This refusal could have been handled by a deep population sensitization right before the vaccination implementation starting with the central leaders, following with social mobilizers and then community leaders so that dissemination of information could start from there. COVID-19 on its own did not influence much vaccine hesitancy but it is insufficient sensitization that was the main factor fuelling vaccine refusal. COVID-19 did not negatively impact the outcome of the campaign as many vaccine-hesitant cases were handled with deep sensitization.
What is known about this topic
- The case fatality rate of cholera in 2020 in Cameroon;
- The impact of mass vaccination campaign against cholera on reducing disease incidence.
What this study adds
- Vaccination campaign against cholera coverage in 3 regions of Cameroon in 2020;
- Challenges of carrying out a vaccination campaign during COVID-19 pandemic;
- Best practices done during the vaccination against cholera campaign during COVID-19.
The authors declare no competing interests.
Adidja Amani and André Arsène Bita Fouda conceptualized and designed the study; Collins Asaah Tatang, Marie Angèle Mbang and Solange Ngo Bama participated in data collection; Armanda Jeanne Nangmo contributed in data analysis and writing original draft preparation; Mariette Dia and Malika Bouhenia have supervised the whole writing process. All authors have participated in writing, reviewing and have read and agreed to the final manuscript.
This reactive oral cholera vaccination campaign was the result of a collaborative effort of many individuals and organizations. The authors acknowledge contributions from the Cameroonian Ministry of Public Health for administrative support. We would also like to thank community members and leaders in the South, South-West and Littoral regions of Cameroon for their tireless dedication to the project.
Table 1: study target population for the oral cholera vaccines (OCV) in the three regions in August-September 2020
Table 2: OCV social mobilization results in the three regions
Table 3: OCV vaccination coverage in the three regions
Figure 1: vaccination coverage rate according to age ranges during the OCV campaign in the South-West region of Cameroon
Figure 2: outlines that as age increases the vaccination coverage decreases in the Littoral region
Figure 3: administrative vaccination coverage according to the age ranges in the three regions
- Deen J, Mengel MA, Clemens JD. Epidemiology of cholera. Vaccine. 2020;38 Suppl 1:A31-A40. PubMed | Google Scholar
- Lippi D, Gotuzzo E. The greatest steps towards the discovery of Vibrio cholerae. Clin Microbiol Infect. 2014 Mar;20(3):191-5. PubMed | Google Scholar
- Institut Pasteur. Choléra: informations et traitements. 2018.
- Kenneth KF. The Cambridge world history of human disease. Cambridge University Press. 1993. Google Scholar
- Chaignat CL. La place des vaccins dans la lutte contre le cholera. Med Trop (Mars). 2001;61(3):249-50. PubMed | Google Scholar
- Nelson EJ, Harris JB, Morris JG Jr, Calderwood SB, Camilli A. Cholera transmission: the host, pathogen and bacteriophage dynamic. Nat Rev Microbiol. 2009 Oct;7(10):693-702. PubMed | Google Scholar
- Azman AS, Rudolph KE, Cummings DA, Lessler J. The incubation period of cholera: a systematic review. J Infect. 2013 May;66(5):432-8. PubMed | Google Scholar
- Jutla A, Whitcombe E, Hasan N, Haley B, Akanda A, Huq A et al. Environmental factors influencing epidemic cholera. Am J Trop Med Hyg. 2013;89(3):597-607. PubMed | Google Scholar
- Legros D; Partners of the Global Task Force on Cholera Control. Global cholera epidemiology: opportunities to reduce the burden of cholera by 2030. J Infect Dis. 2018;218(suppl_3):S137-S140. PubMed | Google Scholar
- Ali M, Nelson AR, Lopez AL, Sack DA. Updated global burden of cholera in endemic countries. PLoS Negl Trop Dis. 2015 Jun 4;9(6):e0003832. PubMed | Google Scholar
- Ngwa MC, Liang S, Mbam LM, Mouhaman A, Teboh A, Brekmo K et al. Cholera public health surveillance in the Republic of Cameroon-opportunities and challenges. Pan African Medical Journal. 2016;24:222. PubMed | Google Scholar
- Ndié J, Bayoro I, Takoukam I. Étude des aspects épidémiologiques du choléra dans Le district de santé de Tcholliré (Nord- Cameroun). Eur Sci Journal, ESJ. 2016;12:278.
- World Health Organization. The weekly epidemiological record (WER). 2020;95;441-448.
- World Health Organization (WHO) Global Task Force on Cholera Control. Cholera country profile: Cameroon. 2012.
- Djomassi LD, Gessner BD, Andze GO, Mballa GA. National surveillance data on the epidemiology of cholera in cameroon. J Infect Dis. 2013;208 Suppl 1:S92-7. PubMed | Google Scholar
- Ngwa MC, Liang S, Kracalik IT, Morris L, Blackburn JK, Mbam LM et al. Cholera in Cameroon, 2000-2012: spatial and temporal analysis at the operational (health district) and sub climate levels. PLoS Negl Trop Dis. 2016 Nov 17;10(11):e0005105. PubMed | Google Scholar
- Directorate of Control of Epidemic and Pandemic diseases (DLMEP). Rapport de situation sur la gestion de l'épidémie de choléra à la date du 18 janvier 2019. 2019;1-2.
- Cameroun: SITREP #2 Eruption fébrile dans la région de l'Extrême-Nord, Février 2017. 2017.
- Hosangadi D, Warmbrod KL, Martin EK, Adalja A, Cicero A, Inglesby T et al. Enabling emergency mass vaccination: innovations in manufacturing and administration during a pandemic. Vaccine. 2020;38(26):4167-4169. PubMed | Google Scholar
- Guha-Sapir D, Moitinho de Almeida M, Keita M, Greenough G, Bendavid E. COVID-19 policies: remember measles. Science. 2020 Jul 17;369(6501):261. PubMed | Google Scholar
- Organisation Mondiale de la Santé. Cadre pour la prise de décision: mise en œuvre de campagnes de vaccination de masse dans le contexte de la COVID-19. 2020;1-10. Google Scholar
- Global Task Force of Cholera Control. Ending cholera - a global roadmap to 2030. 2018.
- Luquero FJ, Grout L, Ciglenecki I, Sakoba K, Traore B, Heile M et al. First outbreak response using an oral cholera vaccine in Africa: vaccine coverage, acceptability and surveillance of adverse events, Guinea, 2012. PLoS Negl Trop Dis. 2013 Oct 17;7(10):e2465. PubMed | Google Scholar
- Iyer AS, Bouhenia M, Rumunu J, Abubakar A, Gruninger RJ, Pita J et al. Immune responses to an oral cholera vaccine in internally displaced persons in South Sudan. Sci Rep. 2016 Oct 24;6:35742. PubMed | Google Scholar
- Ivers LC, Teng JE, Lascher J, Raymond M, Weigel J, Victor N et al. Use of oral cholera vaccine in haiti: a rural demonstration project. Am J Trop Med Hyg. 2013 Oct;89(4):617-624. PubMed | Google Scholar
- Rouzier V, Severe K, Juste MAJ, Peck M, Perodin C, Severe P et al. Cholera vaccination in Urban Haiti. Am J Trop Med Hyg. 2013 Oct;89(4):671-681. PubMed | Google Scholar
- Tohme RA, François J, Wannemuehler K, Iyengar P, Dismer A, Adrien P et al. Oral cholera vaccine coverage, barriers to vaccination and adverse events following vaccination, Haiti, 2013. Emerg Infect Dis. 2015 Jun;21(6):984-91. PubMed | Google Scholar
- Cavailler P, Lucas M, Perroud V, McChesney M, Ampuero S, Guérin PJ et al. Feasibility of a mass vaccination campaign using a two-dose oral cholera vaccine in an urban cholera-endemic setting in Mozambique. Vaccine. 2006 May 29;24(22):4890-5. PubMed | Google Scholar
- Kar SK, Sah B, Patnaik B, Kim YH, Kerketta AS, Shin S et al. Mass vaccination with a new, less expensive oral cholera vaccine using public health infrastructure in India: the Odisha model. PLoS Negl Trop Dis. 2014 Feb 6;8(2):e2629. PubMed | Google Scholar
- Nigus M, Zelalem M, Abraham K, Shiferaw A, Admassu M, Masresha B. Implementing nationwide measles supplemental immunization activities in Ethiopia in the context of COVID-19: process and lessons learnt. Pan African Medical Journal. 2020.;37(Suppl 1):36. PubMed | Google Scholar
- Schaetti C, Ali SM, Chaignat CL, Khatib AM, Hutubessy R, Weiss MG. Improving community coverage of oral cholera mass vaccination campaigns: Lessons learned in Zanzibar. PLoS One. 2012;7(7):e41527. PubMed | Google Scholar