Eosinophilia in school-going children with Plasmodium falciparum and helminth infections in the Volta region of Ghana
Verner Ndudiri Orish, Jones Ofori-Amoah, Kokou Hefoume Amegan-Aho, Emmanuel Udochukwu Osisiogu, James Osei-Yeboah, Sylvester Yao Lokpo, Emmanuel Alote Allotey, Joseph Adu-Amankwaah, Daniel Edem Azuma, Percival Delali Agordoh
Corresponding author: Percival Delali Agordoh, Department of Nutrition and Dietetics, School of Allied Health Sciences, University of Health and Allied Sciences, Ho, Ghana 
Received: 18 Oct 2018 - Accepted: 06 Jan 2021 - Published: 17 Mar 2021
Domain: Haematology,Parasitology,School health
Keywords: Eosinophilia, school children, Plasmodium falciparum, parasitic infection
©Verner Ndudiri Orish et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Verner Ndudiri Orish et al. Eosinophilia in school-going children with Plasmodium falciparum and helminth infections in the Volta region of Ghana. Pan African Medical Journal. 2021;38:277. [doi: 10.11604/pamj.2021.38.277.17379]
Available online at: https://www.panafrican-med-journal.com//content/article/38/277/full
Research 
Eosinophilia in school-going children with Plasmodium falciparum and helminth infections in the Volta region of Ghana
Eosinophilia in school-going children with Plasmodium falciparum and helminth infections in the Volta region of Ghana
Verner Ndudiri Orish1, Jones Ofori-Amoah2, Kokou Hefoume Amegan-Aho3, Emmanuel Udochukwu Osisiogu4, James Osei-Yeboah5, Sylvester Yao Lokpo5, Emmanuel Alote Allotey5, Joseph Adu-Amankwaah5, Daniel Edem Azuma6, Percival Delali Agordoh7,&
&Corresponding author
Introduction: eosinophilia is seen in children infected with parasitic organisms. This study aimed at evaluating eosinophilia in children infected with Plasmodium falciparum, Schistosoma haematobium and intestinal helminths in the Volta Region of Ghana.
Methods: five hundred and fifty primary school children were selected for this study from 5 primary schools in 2 districts and a municipal area of the Volta Region of Ghana. Blood, stool and urine samples were obtained and screened for P. falciparum, intestinal helminths and S. haematobium respectively. Socio-demographic information were obtained using a standardized questionnaire administration. Pearson chi square analysis was used to evaluate the association between eosinophilia and parasitic infections, and multivariate logistics regression analysis was used to identify factors independently associated with increased risk of eosinophilia.
Results: a total of 145(26.36%) children had eosinophilia of which 107(73.79%) were infected with P. falciparum infection, (p=0.016); 18(12.41%) with S. haematobium infection, (p=0.016); and 3(2.07%) children were infected with intestinal helminth, (p=0.36). Children infected with P. falciparum had 2 times increased risk of eosinophilia (AOR=2.01, 95% CI, [1.29-3.2], p=0.02); while children from Davanu primary school had 4 times increased risk of eosinophilia (AOR=4.3, 95% [2.41-10.10], p<0.001).
Conclusion: there was significantly high prevalence of eosinophilia among children infected with P. falciparum infection. A longitudinal study is needed to further understand the immune response of these children to parasitic infections.
Eosinophil is a granulocytic leucocyte responsible for immunity against parasites and mediation of allergic reactions in humans [1]. It constitutes about 1- 5% of the total white cell count, and a very effective component of innate immune system [2]. An abnormally high eosinophil count in the blood is termed eosinophilia; defined as eosinophil count greater than 450 cells/μL [3]. Eosinophilia is typically seen in adult and children with helminths and Plasmodium falciparum infections [4]. Plasmodium falciparum infection is a very common infection in malaria endemic areas of the world, affecting many, especially pregnant women and children [5]. The immunity against P. falciparum is orchestrated by a timely relay between T-helper cell type 1 (Th-1) and T-helper cell type 2 (Th-2) immune responses. The Th-2 immune response stimulates the production of eosinophils, especially in patients who are recovering from P. falciparum infection or those with uncomplicated infections [6, 7].
Helminth infections are very common in developing countries of the world, especially in sub-Saharan Africa where poor environmental sanitation and inadequate water is prevalent [8]. Intestinal helminths like Ascaris lumbricoides, Hookworm, Trichuris trichiura, and Trematodes like Schistosoma haematobium and mansoni are very common infections of children in endemic areas [8, 9]. Eosinophils are very crucial in the defense against helminths as they are produced through Th-2 immune response to larvae permeation of the human tissues [4, 10]. In Ghana, helminth and P. falciparum infections are endemic, causing significant morbidity and mortality in children [11-13]. It will be very important to evaluate the leucocyte profile of these children in a bid to further understand the immune modulation of these parasitic infections. The aim of this study therefore, was to evaluate the eosinophil count and identify risk factors in school-going children infected with helminth and P. falciparum in the Volta Region of Ghana.
A total of 550 primary school children, aged 6-14 years, from 5 primary schools were enrolled for this study. The primary schools were purposively selected from 2 districts and a municipal area in the southern part of the Volta Region of Ghana. They included Freetown Primary School from the Ho municipality (the administrative capital of the region), Evangelical Presbyterian (EP) Primary Schools in Afegame and Kpetoe in the Agotime-Ziope district, and Dave and Davanu Primary Schools in the Adaklu district.
The study procedure has been previously described elsewhere [14, 15]. Briefly, blood, stool and urine samples were obtained from children who gave assent and whose parents consented to the study. Questionnaire was used to capture socio-demographic data of the children. Samples collected were transported in an ice chest to the laboratory for same day analysis. Laboratory diagnosis of malaria was done using P. falciparum specific Bioline SD rapid diagnostic test (RDT) (Standard Diagnostics, INC., South Korea), and Giemsa-stained thick and thin film preparations. P. falciparum infection was defined as a sample positive for either RDT or microscopy. Stool samples were analyzed using the wet mount technique for the detection of ova of helminthes. Urine centrifugation method was used for the detection of S. haematobium ova.
A complete blood count analysis was done on the blood samples using automated haematology analyzer (Sysmex, Kakogawa, Japan). Haemoglobin concentration and eosinophil counts were obtained from the complete blood profile. Anaemia was classified into severe (Hb<7g/dl), moderate (7-9.9g/dl), and mild anaemia (<11g/dl) [15]; while eosinophilia was classified as eosinophil count greater than 450 cells/μL [3].
Statistical analysis was done using IBM SPSS Statistics version 21.0 (IBM Corporation, Armonk, NY, USA). Frequency distribution was performed for all variables of interest. Pearson chi square tests analysis was used to investigate the association between eosinophilia and other variables. Multivariate logistic regression was then used to investigate factors independently associated with increased risk of eosinophilia among the children. Analyses were done with 95% confidence interval (CI), and p value of 0.05 and below was considered statistically significant.
A total of 145(26.36%) children had eosinophilia; 383(69.64%) were positive for P. falciparum, 22(4.00%) had intestinal parasitic infections (i.e. Ascaris lumbricoides [7], Hookworm [5], Entamoeba spp (10), whilst 101 (18.36%) children were anaemic (Table 1). A significant proportion of the children infected with P. falciparum had eosinophilia (107, 73.79%, p=0.016) than those who were uninfected (Table 1). Also, children infected with S. haematobium infection were less eosinophilic, however this finding was not significant (15, 12.41%, p=0.16) (Table 1). Davanu Primary School in the Adaklu district significantly had more children with eosinophilia (42, 28.98%, p<0.001) (Table 1). Majority of the children with eosinophilia had normal haemoglobin concentration (106, 73.10%), although this finding was not significant (p=0.2).
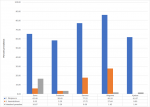
A significant proportion of the children from Afegame E.P Primary School in Agotime-Ziope district (106, 86.14%, p=0.001), and Davanu Primary School in Adaklu district (61, 77.22%, p=0.001), were infected with P. falciparum infection (Figure 1). Though not statistically significant (p=0.16), Davanu (14, 17.72%) and Afegame E.P (34, 27.64%) Primary Schools also recorded a high proportion of the children infected with S. haematobium (Figure 1).
Multivariate logistics analysis (Table 2) showed that children infected with P. falciparum were independently associated with increased risk of eosinophilia (Unadjusted OR, 2.11; 95% CI, 1.35-3.31; p=0.001; Adjusted OR, 2.02, 95% CI, 1.29-3.22; p=0.02). Children from Davanu Primary School was independently associated with higher risk of eosinophilia (Unadjusted OR, 4.83; 95% CI, 2.42-9.67; p<0.001; Adjusted OR, 4.39, 95% CI, 2.41-10.10; p<0.001).
The prevalence of eosinophilia as seen in this study (26.4%) is a fairly common manifestation among children living in P. falciparum and helminths endemic areas [4, 6, 10]. This study recorded only asymptomatic P. falciparum infected children (69.6%) with a significant proportion (73.8%) having elevated eosinophil count. Many studies have reported reduced eosinophil count in acute and complicated malaria, and eosinophilia in patients receiving treatment and recovering from malaria in uncomplicated and asymptomatic infections [6, 7, 16, 17]. This fluctuation of the eosinophil count, in relation to the severity and intensity of P. falciparum infection, vis-a-vis the low levels in acute infections with a progressive increase in recovering patients, indicates a healthy immune response underscoring the anti-plasmodial activity of eosinophils [6, 7]. As infection resolves with elevated eosinophil count, other haematological parameters like haemoglobin concentration also improves [7]. This might explain why majority of the children with eosinophilia (73%) in this study had normal haemoglobin concentration.
There was a less remarkable prevalence of eosinophilia among children infected with S. haematobium (12%) and intestinal helminths (2%). Studies have reported eosinophilia among children with S. haematobium and intestinal helminth infections [10, 18-20]. However, eosinophilia tend to be lowered following treatment with anti-helminthic drugs and periodic deworming exercises [20, 21]. This might explain the reason for the low prevalence of eosinophilia seen in children with helminth infection, because of the regular periodic deworming exercise initiated by the School Health and Education Programme (SHEP) of the Ghana Education Service/Ministry of Education and the NTDCP of the Ghana Health Service/Ministry of Health [22, 23]. Praziquantel and albendazole are administered to target S. haematobium and intestinal helminths through these initiatives, respectively [23].
Children from Davanu Primary School had the highest prevalence of eosinophilia (28.9%). These children had a significantly high prevalence of P. falciparum infection as well. This high prevalence of P. falciparum infection might be responsible for the high prevalence of eosinophilia seen among these children. Another factor that might have been responsible for this high prevalence of eosinophilia is the rural setting of Davanu Primary School. Davanu is located in the Adaklu district, which is a predominantly rural settlement with poor water supply and inadequate sanitation [24]. These conditions are very much linked to higher risk of other various parasitic infections, allergies and skin diseases that can cause eosinophilia [19, 25].
This study showed that there was significant high prevalence of eosinophilia among children infected with P. falciparum infection. Primary school children from deprived rural community had significantly high eosinophilia. Further studies are needed to fully understand the dynamics of parasitic infections, deworming and eosinophilia among these children.
What is known about this topic
- Eosinophil is a granulocytic leucocyte responsible for immunity against parasites and mediation of allergic reactions in humans;
- Abnormally high eosinophil counts in the blood is termed eosinophilia and is defined as eosinophil count greater than 450 cells/μL;
- Eosinophilia is typically seen in adult and children with helminths and Plasmodium falciparum infections.
What this study adds
- This study further supports the finding of eosinophilia in P. falciparum as there was significant high prevalence of eosinophilia among children infected with P. falciparum infection;
- The study demonstrates that the prevalence of eosinophilia is higher among children in the rural areas than among their urban counterparts.
The authors declare no competing interests.
This work was carried out in collaboration among all authors. Author VNO conceived and designed the study, performed the statistical analyses, wrote the protocol and the first draft of the manuscript. Authors JOA, KHAA and PDA assisted in the design of the protocol, conducted the literature searches, and revised the drafted manuscript. Authors EUO, JOY, SYL, EAA, JAA and DEA managed the laboratory work as well as reviewing the protocol and manuscript. All authors read and approved the final manuscript.
We thank the parents and the children who participated in this study, as well as the staff and teachers of the various schools. We also thank our research assistants. Their infallible support made this work a success.
Table 1: characteristics of children stratified by eosinophilia status
Table 2: multivariate logistics regression analysis of the risk of eosinophilia among the primary school children
Figure 1: percentage
prevalence of parasitic infections among children in the 5 primary
schools
- Gleich GJ, Adolphson CR. The Eosinophilic Leukocyte: Structure and Function. Adv Immunol. 1986;39:177-253. PubMed | Google Scholar
- Richards MW, Behrens RH, Doherty JF. Short report: hematologic changes in acute, imported Plasmodium falciparum malaria. Am J Trop Med Hyg. 1998 Dec 1;59(6):859-859. PubMed | Google Scholar
- Jacobsen EA, Helmers RA, Lee JJ, Lee NA. The expanding role(s) of eosinophils in health and disease. Blood. 2012 Nov 8;120(19):3882-90. PubMed | Google Scholar
- Kovalszki A, Weller PF. Eosinophilia. Prim Care. 2016 Dec;43(4):607-617. PubMed | Google Scholar
- van Eijk AM, Hill J, Noor AM, Snow RW, ter Kuile FO. Prevalence of malaria infection in pregnant women compared with children for tracking malaria transmission in sub-Saharan Africa: a systematic review and meta-analysis. Lancet Glob Health. 2015 Oct 1;3(10):e617-28. PubMed | Google Scholar
- Camacho LH, Wilairatana P, Weiss G, Mercader MA, Brittenham GM, Looareesuwan S et al. The eosinophilic response and haematological recovery after treatment for Plasmodium falciparum malaria. Trop Med Int Health. 1999 Jul 1;4(7):471-5. PubMed | Google Scholar
- Kurtzhals JAL, Reimert CM, Tette E, Dunyo SK, Koram KA, Akanmori BD et al. Increased eosinophil activity in acute Plasmodium falciparum infection�association with cerebral malaria. Clin Exp Immunol. 1998 May;112(2):303-. PubMed | Google Scholar
- Karagiannis-Voules D-A, Biedermann P, Ekpo UF, Garba A, Langer E, Mathieu E et al. Spatial and temporal distribution of soil-transmitted helminth infection in sub-Saharan Africa: a systematic review and geostatistical meta-analysis. Lancet Infect Dis. 2015 Jan 1;15(1):74-84. PubMed | Google Scholar
- Adenowo AF, Oyinloye BE, Ogunyinka BI, Kappo AP, Adenowo AF, Oyinloye BE et al. Impact of human schistosomiasis in sub-Saharan Africa. Braz J Infect Dis. 2015 Apr;19(2):196-205. PubMed | Google Scholar
- Huang L, Appleton JA. Eosinophils in Helminth Infection: Defenders and Dupes. Trends Parasitol. 2016 Oct 1;32(10):798-807. PubMed | Google Scholar
- Mirisho R, Neizer ML, Sarfo B. Prevalence of Intestinal Helminths Infestation in Children Attending Princess Marie Louise Children´s Hospital in Accra, Ghana. J Parasitol Res. 2017;2017:8524985. PubMed | Google Scholar
- Anto F, Asoala V, Adjuik M, Anyorigiya T, Oduro A, Akazili J et al. Childhood activities and schistosomiasis infection in the Kassena-Nankana district of Northern Ghana. J Infect Dis Ther. 2014 Jul 5;2(4). Google Scholar
- Sicuri E, Vieta A, Lindner L, Constenla D, Sauboin C. The economic costs of malaria in children in three sub-Saharan countries: Ghana, Tanzania and Kenya. Malar J. 2013 Sep 3;12(1):307. PubMed | Google Scholar
- Orish VN, Ofori-Amoah J, Amegan-Aho KH, Mac-Ankrah L, Jamfaru I, Afeke I et al. Low Prevalence of Helminth Infections among Primary School Children in the Volta Region of Ghana. Asian J Med Health. 2017;5(3):1-9. Google Scholar
- Orish VN, Amegan-Aho KH, Mac-Ankrah L, Jamfaru I, Afeke I, Adzaku FK. High Prevalence of Asymptomatic Plasmodium falciparum Infections in Primary School Children in the Volta Region of Ghana. Texila Int J Public Health. 2017;5(3):4. Google Scholar
- Maina RN, Walsh D, Gaddy C, Hongo G, Waitumbi J, Otieno L et al. Impact of Plasmodium falciparum infection on haematological parameters in children living in Western Kenya. Malar J. 2010 Dec 13;9 Suppl 3(Suppl 3):S4. PubMed | Google Scholar
- Elbadr AM, Saif NA, Mahmoud E, Osman R, Elmatary AMA. Severe anemia in children infected with malaria in Taiz-Yemen and its relation to age, parasitaemia and eosinophilia. Life Sci J. 2011;8(1):40-43. Google Scholar
- Bejon P, Mwangi TW, Lowe B, Peshu N, Hill AVS, Marsh K. Helminth Infection and Eosinophilia and the Risk of Plasmodium falciparum Malaria in 1- to 6-Year-Old Children in a Malaria Endemic Area. PLoS Negl Trop Dis. 2008 Feb 6;2(2):e164. PubMed | Google Scholar
- Panggabean YC, Nasution SW, Siahaan L, Pasaribu AP. Eosinophilia in Children with Single and Mixed Soil Transmitted Helminthiasis. Adv Health Sci Res. 2017;1:33-5. Google Scholar
- Mohammed EH, Eltayeb M, Ibrahim H. Haematological and Biochemical Morbidity of Schistosoma Haematobium in School Children in Sudan. Sultan Qaboos Univ Med J. 2006 Dec;6(2):59-64. PubMed | Google Scholar
- Fincham JE, Markus MB, Adams VJ, Lombard CJ, Bentwich Z, Mansvelt EPG et al. Association of deworming with reduced eosinophilia?: implications for HIV / AIDS and co-endemic diseases: research letters. South Afr J Sci. 2003 Mar 1;99(3-4):182-4. Google Scholar
- Abdul-Rahman L, Agble R. Review of School Health and Nutrition Interventions and Mapping of Existing Programmes in Ghana. Lond UK Partnersh Child Dev. 2012.
- Ayeh-Kumi PF, Addo-Osafo K, Attah SK, Tetteh-Quarcoo PB, Obeng-Nkrumah N, Awuah-Mensah G et al. Malaria, helminths and malnutrition: a cross-sectional survey of school children in the South-Tongu district of Ghana. BMC Res Notes. 2016 Apr 27;9(1):242. PubMed | Google Scholar
- Ghana Statistical Service (GSS), International ICF. Ghana Demographic and Health Survey 2014. Rockville, Maryland, USA: GSS, GHS, and ICF International. 2015.
- Roldán WH, Espinoza YA, At�ncar A, Ortega E, Martinez A, Saravia M. Frequency of eosinophilia and risk factors and their association with Toxocara infection in schoolchildren during a health survey in the north of Lima, Peru. Rev Inst Med Trop S�o Paulo. 2008 Oct;50(5):273-8. PubMed | Google Scholar