Management and outcome of hypertrophic obstructive cardiomyopathy in pregnant women: a case report
Maryem Assamti, Ramia Bougrine, Nabila Ismaili, Noha El Ouafi
Corresponding author: Maryem Assamti, Department of Cardiology, Mohamed VI University Hospital, Mohamed First University of Oujda, Oujda, Morocco 
Received: 22 Aug 2020 - Accepted: 19 Oct 2020 - Published: 08 Feb 2021
Domain: Cardiology
Keywords: Hypertrophic cardiomyopathy, pregnancy, sudden cardiac death, case report
©Maryem Assamti et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Maryem Assamti et al. Management and outcome of hypertrophic obstructive cardiomyopathy in pregnant women: a case report. Pan African Medical Journal. 2021;38:140. [doi: 10.11604/pamj.2021.38.140.25699]
Available online at: https://www.panafrican-med-journal.com//content/article/38/140/full
Case report 
Management and outcome of hypertrophic obstructive cardiomyopathy in pregnant women: a case report
Management and outcome of hypertrophic obstructive cardiomyopathy in pregnant women: a case report
![]() Maryem Assamti1,&, Ramia Bougrine1, Nabila Ismaili2, Noha El Ouafi1,2
Maryem Assamti1,&, Ramia Bougrine1, Nabila Ismaili2, Noha El Ouafi1,2
&Corresponding author
Hypertrophic cardiomyopathy (HCM) is a common inherited cardiomyopathy, with an estimated prevalence of 1 in 500 people. Despite overall favorable outcomes with modern treatment and early diagnosis of disease, adverse complications could occur during times of physiological stress like pregnancy. Complications of HCM include sudden cardiac death, heart failure, and arrhythmia. We report the case of a 32-year-old pregnant woman with obstructive HCM, presenting with recurrent episodes of ventricular arrhythmia despite medical therapy. This case exhibits how close monitoring and proper management during pregnancy according to the latest recommendations, resulted in a successful and uneventful delivery.
Hypertrophic cardiomyopathy (HCM) is a genetic condition characterized by primary left ventricular hypertrophy that is not related to abnormal loading conditions (such as hypertension or valvular disease) [1]. This disease is often caused by mutations within genes encoding cardiac sarcomeric proteins [2]. Although pregnancy is well tolerated by HCM patients [3]; related hemodynamic burdens may lead to unfavorable events requiring close monitoring and adequate treatment.
A 32-year-old pregnant woman presented at 36 weeks gestation for further management of known HCM. She was recently diagnosed with HCM at 22 weeks of gestation, after she presented with paroxysmal palpitations. She had a history of a sudden cardiac death of her sister at the age of 15, and one of her brothers at the age of 11. Her first pregnancy was well tolerated. Her first echocardiogram showed a mild left ventricular outflow tract obstruction with a gradient at 30 mmHg, and a septum thickness of 15 mm. An ambulatory electrocardiogram monitoring showed isolated monomorphic ventricular extrasystoles, without ventricular tachycardia. Her SD score was then at 4%, an implantable cardioverter defibrillator (ICD) was proposed but the patient refused it; she was treated with bisoprolol only.
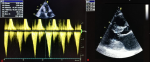
At 36 weeks of gestation, she consulted in our hospital. She reported palpitations; without dyspnea, nor chest pain, or syncope. Her physical examination revealed a blood pressure (BP) of 110/65 mmHg, a regular pulse rate of 108 beats per minute, anda mid-systolic murmur over the lower left sternal border.The electrocardiogram showed sinus rhythm. An echocardiogram demonstrated a posterior wall thickness at 26 mm, a septum thickness at 17 mm; and a mild increase in LVOT obstruction with agradient of 42 mmHg at rest; without an increase at standing up or Valsalva maneuvers; nor systolic anterior motion of the mitral valve, and a restrictive mitral profile (Figure 1). A 48 hours electrocardiography (ECG) monitoring revealed multiple short runs of non-sustainedventricular tachycardia. Pro BNP and BNP dosage were unavailable. The reassessed sudden cardiac death risk score was higher at 14%.
After a comprehensive discussion among cardiologists, gynecologists, and anesthesiologists, we decided to perform a vaginal delivery. At 37 weeks of gestation, the patient was admitted to the obstetric department, and an induction of labor was initiated under close maternal and fetal monitoring with maintained medical therapy. The patient gave birth to a healthy female newborn after an uneventful delivery. The patient remained on beta-blockers, and she is currently asymptomatic.
Hypertrophic cardiomyopathy (HCM) is the most common genetic cardiac disease, with an estimated prevalence of 0,2% in the general population [4], and less than 0,1% in childbearing women [3]. Hypertrophic cardiomyopathy is associated with an increased risk of sudden cardiac death, arrhythmia, and heart failure. The incidence of complications varies between 5 and 48%, depending on the size of the samples and the study design. While the maternal mortality is estimated at 0.5% [3]. According to the ROPAC registry, which prospectively included 60 pregnant women with HCM, 23% of the patients developed important MACE (including HF and arrhythmias). They observed that NYHA functional class > 2 and signs of heart failure before pregnancy were significantly associated with cardiovascular events. Surprisingly, no significant differences in pregnancy outcome were found between women with obstructive and non-obstructive HCM [5]. Autore et al. also noticed that clinical deterioration occurred more frequently in women with LV outflow tract obstruction, without statistically significant difference [6]. In contrast, in a retrospective review of 27 pregnancies in 23 women with HCM, Tanaka et al. reported a cardiovascular event rate of 48%; the vast majority involved arrhythmia and occurred during the 3rd trimester. They also identified the use of cardiac medication prior to pregnancy as a predictor of maternal cardiac events [7]. This condition may influence the fetal outcome. Schinkel reported in his literature review a 15% rate of spontaneous abortion and a 2% stillbirth rate, which is comparable to the general population. However, premature birth was more frequent, and was observed 26% of cases [8].
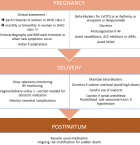
Therefore, an appropriate assessment antepartum and proper monitoring and medical management are crucial to avoid related morbidity. In general, the diagnosis of HCM is confirmed when left ventricular (LV) myocardial thickness exceeds 15 mm on echocardiography, cardiac magnetic resonance (CMR) imaging, or computed tomography (CT) [9]. Once the diagnosis is confirmed, clinical status, left ventricular outflow tract (LVOT) obstruction, ventricular function, and arrhythmias should be assessed. Besides, establishing baseline levels of serum BNP or NT-proBNP, before or at the beginning of pregnancy, and throughout pregnancy in symptomatic patients, or at the onset of symptoms, can reflect hemodynamic deterioration [10]. Women in WHO class II(mild to moderate LVOT obstruction, asymptomatic with or without medication, well-controlled arrhythmia, normal systolic LV function, or mild LV dysfunction) should be assessed each trimester, and those in WHO class III (severe LVOT obstruction, symptoms or arrhythmias despite optimal medication, moderate systolic LV dysfunction) should be assessed monthly or bimonthly [1, 3].
In WHO class IV with severe symptomatic LVOT obstruction or severe systolic LV dysfunction, pregnancy is in principle contraindicated [1]. In order to prevent undesired events, and culminate in a smooth delivery, appropriate peripartum management and close monitoring are essential. According to the latest ESC guidelines, beta-blockers should be continued if they are already being taken, and should be started in case of new-onset of arrhythmia (preferably metoprolol) [3]. Complications such as neonatal hypoglycemia, bradycardia, and apnea should be anticipated [9]. Verapamil and disopyramide are a second choice treatment if beta-blockers are not tolerated [11]. Antiarrhythmic drugs should be administered with caution because of the potential fetotoxic effects [9]. Anticoagulation is recommended for paroxysmal or persistent AF, regardless of the CHADS VASC score. Low molecular weight heparin with anti-factor-Xa monitoring (peak anti-Xa level 0.8-1.2 U/mL 4-6 h post-dose) in the first trimester and from the 36th week onwards, and VKAs are recommended in the second and third trimester. NOACs are not recommended because of proven toxicity in animals and insufficient data in humans [3]. Patients with a history or family history of sudden death (such as in our case) need close surveillance and prompt investigation if they present symptoms of palpitations or presyncope. When indicated, an ICD should be implanted preferably with echocardiographic guidance [3].
In order to reduce the outflow gradient, alcohol septal ablation has been described. There are only two recent case reports of alcohol septal ablation during pregnancy, but only one of them was successful [12]. This suggests that ASA could be performed, when conservative therapy is ineffective, but should ideally be considered before conception in high-risk patients. Regarding the mode of delivery, cesarean section should be considered mostly for obstetric indications, or in patients with severe left ventricular (LV) outflow tract obstruction, pre-term labor while on oral anticoagulation, or severe heart failure (HF) [3]. Tocolytic agents with �-adrenergic stimulating activity should be avoided in women with obstructive HCM [9]. Oxytocin should be given as a slow infusion, because of vasodilatory properties. Epidural and spinal anesthesia must be used cautiously, especially with severe LV outflow tract obstruction, since it may induce hypotension. A close monitoring of heart rhythm should be considered in patients with a high risk of developing arrhythmias [3]. Despite the high risk of endocarditis associated with HCM, prophylactic antibiotic treatment before delivery is not recommended [13] (Figure 2).
In conclusion, and despite the paucity of data, the complication rate in pregnant women with HCM appears to increase in case of a history of previous cardiac events, poor functional class, and LVOT obstruction. Therefore, early screening of this disease, identifying women at high risk of adverse outcomes and counseling on contraception, is essential in HCM women. Furthermore, it is critical to inform women with HCM regarding the risk of genetic transmission to their offspring.
The authors declare no competing interests.
All authors have read and agreed to the final version of this manuscript.
Figure 1: transthoracic echocardiography showing: A) a max instantaneous late peak gradient of 42 mmHg measured with continuous wave (CW) Doppler; B) a posterior wall thickness at 26 mm and a septum thickness at 17 mm measured on a parasternal long axis view
Figure 2: proposed algorithm for assessment and management of HCM during pregnancy
- Authors/Task Force members, Elliot PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F, Charron P et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J. 2014 Oct 14;35(39):2733-79. PubMed | Google Scholar
- Van Driest SL, Ommen SR, Tajik AJ, Gersh BJ, Ackerman MJ. Yield of genetic testing in hypertrophic cardiomyopathy », Mayo Clin Proc. 2005 Jun;80(6):739-44. PubMed | Google Scholar
- Regitz-Zagrosek V, Roos-Hessenlink JW, Bauersachs J, Blomstrom-Lundqvist C, Cifkova R, Michele De Bonis et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. European Heart Journal. 2018 Sep 7;39(34):3165-3241. PubMed | Google Scholar
- Maron MS, Hellawell JL, Lucove JC, Farzaneh-Far R, Olivotto I. Occurrence of Clinically Diagnosed Hypertrophic Cardiomyopathy in the United States. Am J Cardiol. 2016 May 15;117(10):1651-1654. PubMed | Google Scholar
- Goland S, van Hagen IM, Elbaz-Greener G, Elkayam U, Shotan A, Merz WM et al. Pregnancy in women with hypertrophic cardiomyopathy: data from the European Society of Cardiology initiated Registry of Pregnancy and Cardiac disease (ROPAC). European Heart Journal. sept. 2017;38(35):2683-2690. Google Scholar
- Autore C, Conte MR, Piccininno M, Bernabo P, Bonfiglio G, Bruzzi P et al. Risk associated with pregnancy in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2002 Nov 20;40(10):1864-9. PubMed | Google Scholar
- Tanaka H, Kamiya C, Katsuragi S, Tanaka K, Miyoshi T, Tsuritani M et al. Cardiovascular events in pregnancy with hypertrophic cardiomyopathy. Circ J. 2014;78(10):2501-6. PubMed | Google Scholar
- Schinkel AFL. Pregnancy in women with hypertrophic cardiomyopathy. Sep-Oct 2014;22(5):217-22. PubMed | Google Scholar
- Hagen IM, Elkayam U, Goland S, Roos-Hesselink JW. Hypertrophic Cardiomyopathy and Pregnancy », in Cardiac Problems in Pregnancy, 4th Edition, 1re éd., U. Elkayam, Éd. Wiley, 2019, p167-180.
- Tanous D, Siu SC, Mason J, Greutmann M, Wald RM, Parker JD et al. B-type natriuretic peptide in pregnant women with heart disease. J Am Coll Cardiol. oct. 2010 Oct 5;56(15):1247-53. PubMed | Google Scholar
- Gersh BJ, Maron BJ, Bonow RO, Dearani JA, Fifer MA, Link MS et al. 2011 ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. 2011 Dec 13;124(24):2761-96. PubMed | Google Scholar
- Shaikh A, Bajwa T, Bush M, Tajik AJ. « Successful alcohol septal ablation in a pregnant patient with symptomatic hypertrophic obstructive cardiomyopathy. Journal of Cardiology Cases. 2018 May 3;17(5):151-154. PubMed | Google Scholar
- Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta J-P, Francesco Del Zotti et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015 Nov 21;36(44):3075-3128. PubMed | Google Scholar