Pulmonary infarction mimicking a lung mass: a case report
Balakrishnan Jayakrishnan, Saif Musabah Al-Mubaihsi, Nisrien Elfatih Elhassan, Rashid Fuad Al-Sukaiti, Jojy George, Younis Said Al-Alawi, Adil Hashim Al-Kindi
Corresponding author: Balakrishnan Jayakrishnan, Department of Medicine, Sultan Qaboos University Hospital, Muscat, Oman 
Received: 26 Dec 2020 - Accepted: 07 Jan 2021 - Published: 04 Feb 2021
Domain: Nuclear medicine,Radiology,Pulmonology
Keywords: Pulmonary embolism, pulmonary infarction, lung cancer, case report
©Balakrishnan Jayakrishnan et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Balakrishnan Jayakrishnan et al. Pulmonary infarction mimicking a lung mass: a case report. Pan African Medical Journal. 2021;38:127. [doi: 10.11604/pamj.2021.38.127.27595]
Available online at: https://www.panafrican-med-journal.com//content/article/38/127/full
Pulmonary infarction mimicking a lung mass: a case report
![]() Balakrishnan Jayakrishnan1,&, Saif Musabah Al-Mubaihsi1, Nisrien Elfatih Elhassan1, Rashid Fuad Al-Sukaiti2, Jojy George1, Younis Said Al-Alawi3, Adil Hashim Al-Kindi4
Balakrishnan Jayakrishnan1,&, Saif Musabah Al-Mubaihsi1, Nisrien Elfatih Elhassan1, Rashid Fuad Al-Sukaiti2, Jojy George1, Younis Said Al-Alawi3, Adil Hashim Al-Kindi4
&Corresponding author
Pulmonary infarction usually appears as a wedge-shaped opacity with its base placed laterally. Rarely, pulmonary infarctions may appear as a well-defined rounded opacity mimicking lung cancer and surgical lung biopsy may often be required for definitive diagnosis. We report a patient who was admitted with submassive pulmonary embolism who had an incidental finding of a well-defined opacity in computed tomography (CT) scan. The lesion was avid on positron emission tomography (PET) scan and the patient was a smoker. So, we investigated him further with a percutaneous and later a thoracoscopic lung biopsy. Tumour-like pulmonary infarction is often a challenge for the clinicians.
Pulmonary infarction usually appears as a wedge-shaped pleural based opacity with no evidence of air bronchograms [1]. Rarely, pulmonary infarctions may appear as a well-defined rounded opacity mimicking lung cancer [2, 3]. Pulmonary infarctions can also result from a variety of non thromboembolic causes and the radiologic appearance may vary with the underlying cause and temporal evolution of the lesion. Solitary pulmonary nodules can be due to infectious or non-infectious causes and often benign or malignant tumours are considered as the first differentials. We present a case of submassive pulmonary embolism with a focal pulmonary infarct who needed a complete work up as the radiological characteristics mimicked a lung mass.
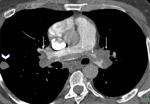
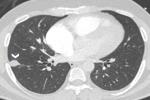
A 39-year-old male, smoker with generalized anxiety disorder, presented with a two-week history of chest pain and dyspnoea. Hemodynamically he was stable with a pulse rate of 78beats per minute, blood pressure of 130/105mmHg and an oxygen saturation of 87%. Electrocardiogram showed sinus tachycardia and symmetrical T-wave inversions in leads V1 to V4. Cardiac troponin T was elevated, 37ng/L. Chest radiograph was unremarkable. Computed tomography angiogram of pulmonary arteries (CTPA) revealed a large saddle shaped thrombus in main pulmonary artery, extending into the right and left pulmonary arteries as well as to the segmental branches on both sides (Figure 1). In addition, a solitary nodule was seen in the right lower lobe measuring 16 x 10 mm, laterally and just posterior to oblique fissure (Figure 2). Echocardiogram, showed features of right ventricular strain. Pulmonary Embolism Severity Index score was 89 points suggesting Class III, intermediate risk.
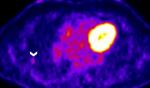
Since the patient was not hemodynamically unstable, systemic thrombolysis was not considered. Owing to persistence of symptoms and desaturation, the high thrombus load and the young age, a multidisciplinary team decision was made to proceed with catheter directed thrombolysis. He showed a remarkable response with total disappearance of his symptoms and improvement in oxygen saturation. Anticoagulation was continued with Rivaroxaban. Patient did not have any known predisposing factors for pulmonary embolism (PE). Malignancy being a risk factor, the rounded opacity in the lung of a smoker needed further evaluation. Patient was readmitted after 4 weeks and a CT guided percutaneous biopsy was done. Unfortunately, it was reported as nondiagnostic as it showed only normal tissue without any evidence for granuloma or malignancy. Whole body [18F]-fluorodeoxyglucose (FDG) positron emission tomography-computed tomography (PET/CT) three months later showed the nodule to be FDG avid with a maximum standardized uptake value of 2.8. (Figure 3).
Knowing about a possible mass in his lungs, the patient was very anxious all along. Again, a multidisciplinary decision was made for excision biopsy weighing the surgical risks in this case with the possibility of malignancy. Excision of the nodule was done through video assisted thoracoscopic surgery. Histopathology showed only a localized area of old infarction, 8 mm in maximum dimension without any evidence for malignancy or granuloma. Rivaroxaban was withheld for 48 hours for the percutaneous biopsy and was bridged with unfractionated heparin for the surgical biopsy. Patient was reassured and later had an uneventful follow up in the out-patient clinic.
Here we report a patient who developed a well-defined mass like pulmonary infarct following a submassive PE. The CT characteristics, mildly avid PET finding, smoking status and the chances of PE in malignancy necessitated the need for a lung biopsy. Typical radiographic feature of pulmonary infarction is a wedge-shaped, pleural based opacity without air bronchograms [1]. However, it may also present as a nodule resembling a primary or metastatic lung tumour.
Solitary pulmonary nodules can be due to neoplasms, infectious or non-infectious granulomas, developmental lesions, arterio-venous malformations, hematoma, intrapulmonary lymph node, inflammatory pseudotumour, amyloidoma, rounded atelectasis, mucoid impaction, progressive massive fibrosis, pulmonary artery aneurysm and pulmonary infarct. The diagnosis of tumour like infarction remains a challenge for many reasons [2]. Primarily, pulmonary infarction can closely mimic the radiological features of lung cancer [3]. Pulmonary thrombus seen as a solitary pulmonary nodule on a chest radiograph has also been reported [4]. Even the PET scan can show an increased uptake [5]. Moreover, cytological changes in pulmonary infarctions may also produce malignant-appearing cells in respiratory secretions and aspiration biopsy specimens [6]. Our patient had a well-defined rounded, fairly large opacity which was also FDG avid.
Occlusion of a pulmonary artery usually does not produce tissue necrosis as the lungs receive oxygen from bronchial arteries and the airways as well. The incidence of pulmonary infarction in patients with pulmonary embolism is usually 10%, but may go up to 30% [7]. Predisposing factors include left heart failure, accompanying pneumonia, sepsis, malignancy, advanced age and a higher clot burden. Infarction is more common with emboli that are distal rather than proximal [8]. Non-thromboembolic causes of pulmonary infarctions include pulmonary infections, diffuse alveolar damage, pulmonary torsion, lung cancer, amyloidosis, bronchial artery embolization therapy, vasculitis, Swan-Ganz catheter use, or sickle-cell disease [9].
Finally, round pneumonia should also be entertained in the differential diagnosis of pulmonary infarct and lung tumour [10]. Nevertheless, persistence of the lesion without any change in size for 5 months, from presentation to the time of lung biopsy, was not in favour of an acute infective process. On the other hand, some pulmonary infarctions are known to change only slowly over time.
Our patient with a submassive pulmonary embolism developed a pulmonary infarct, the characteristics of which resembled a lung mass. Since he was a smoker and the lesion was PET avid, a lung biopsy was done. In spite of advancements in diagnostic methods tumour like pulmonary infarction will continue to pose a challenge for the clinicians. Often, a definitive surgical intervention is needed for a proper diagnosis.
The authors declare no competing interests.
All authors were involved in the clinical management of the patient and in the review of literature and writing up of the manuscript. All authors read and approved the final version of the manuscript.
Figure 1: CT pulmonary angiogram mediastinal window axial coronal oblique view demonstrating saddle embolus involving distal main pulmonary artery and extending into both left and right pulmonary arteries ( white arrows) and a well-defined right lung lateral subpleural opacity (white arrow head)
Figure 2: CT pulmonary angiogram axial lung window demonstrating a well-defined right lung lateral opacity (White arrow head)
Figure 3: PET CT image that demonstrates a mildly FDG avid right lung lateral subpleural nodule (white arrow head) with SUV of 2.8
- Suut S, Al-Ani Z, Allen C, Rajiah P, Durr-E-Sabih, Al-Harbi A et al. Pictorial essay of radiological features of benign intrathoracic masses. Ann Thorac Med. Oct-Dec 2015;10(4):231-42. PubMed | Google Scholar
- Lacout A, Marcy PY, El Hajjam M. To avoid operating on pseudo tumoral pulmonary infarctions mimicking lung cancer. Pan Afr Med J. 2012;12:11. PubMed | Google Scholar
- Teramoto K, Kawaguchi Y, Hori T, Ohshio Y, Hashimoto M, Kitamura S et al. A Pulmonary Infarction Mimicking Metastatic Lung Tumor. Surg Sci. 2011;2:360-62. Google Scholar
- Gupta M, Gupta R, Gogia N, Thulkar S. Pulmonary artery thrombus seen as a solitary pulmonary nodule on chest radiograph. Singapore Med J. 2010 Jan;51(1):e22-3. PubMed | Google Scholar
- George CJ, Tazelaar HD, Swensen SJ, Ryu JH. Clinicoradiological features of pulmonary infarctions mimicking lung cancer. Mayo Clin Proc. 2004 Jul;79(7):895-8. PubMed | Google Scholar
- Lawther RE, Graham AN, McCluggage WG, McGuigan JA. Pulmonary infarct cytologically mimicking adenocarcinoma of the lung. Ann Thorac Surg. 2002 Jun;73(6):1964-5. PubMed | Google Scholar
- Sonmez M, Aboussouan LS, Farver C, Murthy SC, Kaw R. Pulmonary infarction due to pulmonary embolism. Cleve Clin J Med. 2018 Nov;85(11):848-852. PubMed | Google Scholar
- Kirchner J, Obermann A, Stuckradt S, Tüshaus C, Goltz J, Liermann D et al. Lung Infarction Following Pulmonary Embolism: A Comparative Study on Clinical Conditions and CT Findings to Identify Predisposing Factors. Rofo. 2015 Jun;187(6):440-4. PubMed | Google Scholar
- Parambil JG, Savci CD, Tazelaar HD, Ryu JH. Causes and presenting features of pulmonary infarctions in 43 cases identified by surgical lung biopsy. Chest. 2005 Apr;127(4):1178-83. PubMed | Google Scholar
- Cimen D, Bulcun E, Ekici A, G�ng�r Ö, Ekici M. Case of round pneumonia: pulmonary infarct and a rare situation that is similar with the lung cancer. Clin Respir J. 2015 Oct;9(4):512-5. PubMed | Google Scholar