Prehabilitation program for African sub-Saharan surgical patients is an unmet need
Antero do Vale Fernandes, Daniel Moreira-Goncalves, Jotamo Come, Nilton Caetano Rosa, Victor Costa, Lygia Vieira Lopes, Paulo Matos da Costa, Lúcio Lara Santos
Corresponding author: Lucio Lara Santos, Experimental Pathology and Therapeutics Group of Portuguese Institute of Oncology of Porto Francisco Gentil, E.P.E (IPO-Porto), Portugal 
Received: 06 Dec 2019 - Accepted: 27 May 2020 - Published: 03 Jun 2020
Domain: Surgical oncology
Keywords: Africa, patients, postoperative, surgery, risk
©Antero do Vale Fernandes et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Antero do Vale Fernandes et al. Prehabilitation program for African sub-Saharan surgical patients is an unmet need. Pan African Medical Journal. 2020;36:62. [doi: 10.11604/pamj.2020.36.62.21203]
Available online at: https://www.panafrican-med-journal.com//content/article/36/62/full
Prehabilitation program for African sub-Saharan surgical patients is an unmet need
Antero do Vale Fernandes1,2, Daniel Moreira-Gonçalves1,3, Jotamo Come4, Nilton Caetano Rosa5, Victor Costa6, Lygia Vieira Lopes7, Paulo Matos da Costa8,9, Lúcio Lara Santos1,10,11,&
1Experimental Pathology and Therapeutics Group of Portuguese Institute of Oncology of Porto Francisco Gentil, E.P.E (IPO-Porto), Portugal, 2Intensive Care Service of Hospital Garcia de Orta, E.P.E, Almada, Portugal, 3Research Centre in Physical Activity, Health and Leisure (CIAFEL), Faculty of Sport, University of Porto, Porto, Portugal, 4Surgical Department of Maputo Central Hospital, Maputo, Mozambique, 5Surgical Oncology Department of Angolan Institute Against Cancer, Luanda, Angola, 6Surgical Department of Agostinho Neto Hospital, Praia, Cape Verde, 7Oncological Unit of Sagrada Esperança Clinic, Luanda, Angola, 8General Surgery Service, Hospital Garcia de Orta, E.P.E, Almada, Portugal, 9Faculty of Medicine of the University of Lisbon, Lisbon, Portugal, 10Surgical Oncology Department of Portuguese Institute of Oncology of Porto Francisco Gentil, E.P.E (IPO-Porto), Portugal, 11ONCOCIR, Education and Care in Oncology, Lusophone Africa, Angola
&Corresponding author
Lúcio Lara Santos, Experimental Pathology and Therapeutics Group of Portuguese Institute of Oncology of Porto Francisco Gentil, E.P.E (IPO-Porto), Portugal
Approximately 4.2 million people worldwide die within 30 days of surgery each year. Half of these deaths occur in low- and middle-income countries. Postoperative deaths account for 7.7% of all deaths globally, making it the third-highest contributor to deaths, after heart disease and stroke. In sub-Saharan Africa, there is a higher rate of mortality following postoperative complications compared to high-income countries. The WHO has tools to help countries provide safer surgery. However, implementation remains poor in most African countries. Interventions focused on intraoperative or postoperative measures to improve perioperative prognosis may be too late for high-risk patients. Poor preoperative cardiorespiratory functional capacity, poor management of pre-existing comorbidities and risk factors and no assessment of the patient´s surgical risk is associated with adverse postoperative outcomes, including mortality, complications, slower recovery, longer intensive care stay, extended hospital length of stay and reduced postoperative quality of life. To significantly decrease morbidity and mortality following surgery in Africa, we propose the implementation of a comprehensive preoperative intervention, that must include: i) risk assessment of surgical patients to identify those at greater risk of postoperative complications for elective surgery; ii) increase the preoperative functional reserve of these high-risk patients, to enhance their tolerance to surgical stress and improve postoperative recovery; iii) anticipate postoperative care needs and organize tools, resources and establish simple workflows to manage postoperative complications. We believe this approach is simple, feasible and will significantly reduce postoperative burden for both patients, hospitals and society.
An African continental study of 25 low- and middle-income countries was recently published by Biccard BM et al. characterizing perioperative outcomes of 11193 surgical patients. The patients were 66.4% women, 87.3% classified with an American Society of Anesthesiologists (ASA) score of I and II, 29.7% undergoing major surgery and 57.1% urgent/emergency surgery [1]. Arterial hypertension (16.3%), diabetes mellitus (6.8%) and HIV positive/AIDS (11%) were the main comorbidities of the operated patients. Non-communicable diseases (NCD) were the most frequent indication for surgical treatment (42.2%), followed by caesarean section (27.3%), trauma (17.8%) and acute infection (12.7%). Despite being younger, presenting a lower risk profile and low complication rates, surgical patients in Africa were twice as likely to die after surgery in comparison to the global average. Indeed, in hospital mortality was 2.1%, with 18% developing postoperative complications (POC) and 9.5% of the patients died following POC [1]. When considering elective surgery (ES) only, mortality occurred in 1% of 4658 patients, with an incidence of postoperative complications of 13.4% and death after POC of 4.8%.
A greater incidence of POC and death were reported following surgery for NCD (37.3% and 40.3%, respectively), infection (20.2% and 26.5%, respectively) and trauma (20.5% and 25.5%, respectively). Infectious, cardiovascular and respiratory complications were the most prevalent [1]. The factors contributing to POC and death are multifactorial and may include insufficient medical staff, poor infrastructure, low procedural volumes and failure to identify and/or treat POC by health professionals [2,3]. Intensive care admission should also be scheduled in advance. In their study, Biccard BM et al. [1] identified that only 16.3% of patients who developed POC, the vast majority after ES, were admitted to intensive care units (ICU) to prevent and treat early complications. The lack of immediate postoperative surveillance and intervention is responsible for many deaths in many African countries. Therefore, acute care surgery (ACS) services should be implemented even in a low-resource setting [4].
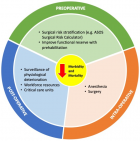
In Rwanda, the implementation of an ACS service resulted in decreased length of hospital stay [5]. Thus, while surgical care is a major need for African countries, surgical outcomes will remain poor unless effective perioperative care based on affordable resources is made universally available. Perioperative care is a multicomponent intervention implemented by a multidisciplinary team with the purpose to provide safe surgery, accelerate recovery and reduce morbidity and mortality (Figure 1). While the intra- and postoperative care have already received some attention, the potential of the perioperative period remains poorly explored in African countries [6-11]. This time frame represents a major opportunity for decreasing postoperative morbidity and mortality through appropriate surgical risk stratification and patient optimization [12].
Risk assessment of surgical complications in sub-Saharan Africa: in an environment with limited resources for postoperative care, the early identification of high-risk patients for POC is likely to be a key factor to consider. Several tools are available to estimate perioperative risk for both planned and emergency surgeries in high-income countries [13,14]. However, their use in low-income countries is often limited because the pattern of risk for poor outcomes differs from high-income countries and due to the lack of resources, the access to biochemical and imagological tests required by more sophisticated tools is reduced [15]. Recently, Kluyts H-L et al. proposed the use of the ASOS surgical risk calculator as a simple tool to identify African surgical patients at risk for in-hospital postoperative mortality and severe complications and thus, to identify those patients in greater need for enhanced postoperative surveillance [16]. However, its external validation needs to be assessed before. To predict complications and risk of death before surgery, other African authors have conducted relevant studies, including the use of online tools, provided that this tool has wide distribution [17,18]. This is a field that deserves further research effort as it may greatly contribute to save lives.
Estimating the risk of complications and mortality of surgical patients before surgery can be helpful: risk stratification of patients is supposed to support better decisions by informing about the risks and benefits of proceeding with surgery, about discussing treatment alternatives and guide the use of available resources, with the ultimate purpose of improving postoperative outcomes. Ntobeko Ntusi, a South African cardiologist, in a recent editorial in the South African Medical Journal, asked: “does the preoperative evaluation of patients improves surgical outcomes?”. He found that the data on the effect of preoperative medical consultation on cost measures is conflicting [19]. While some studies reveal a decrease in-hospital stay after preoperative evaluation and care of patients [20,21], other studies have shown an increase in costs and a similar length of stay for consulted patients [22-24]. He also points that while medical teams can successfully identify conditions that may affect surgical outcomes, it is not clear if they make evidence-based recommendations to target those conditions and assuming they make it, it is also not clear if the consultative recommendations are implemented [19]. With this data and his experience, Ntusi argued that an experienced perioperative medicine physician should be able to identify the pertinent medical problems, anticipate potential perioperative problems, recommend evidence-based interventions to optimize the patient and communicate and work effectively with all the preoperative team members (e.g. nursing, physiotherapist, medical, surgical and anesthetic) [19]. Thus, to deal with the problem of postoperative morbidity and mortality, the perioperative care, particularly the potential of the pre-surgical period to optimize the patient for surgery, needs to be taken more seriously. These patients need and deserve better care and prehabilitation programs can make the difference once incorporated in the routine practice of surgical teams.
Prehabilitation to prepare for surgery: the impact of surgery leads to significant homeostatic disturbance which, together with reduced functional capacity (physical, nutritional and psychological status) and poor medical optimization (e.g. unappropriated management of chronic diseases, anemia, hypertension, hyperglycemia and smoking), act as risk factors for negative surgical outcomes [25]. Prehabilitation is a multimodal strategy implemented in the preoperative period, aiming to increase preoperative functional reserve and leading to better postoperative functional recovery and reduced incidence of complications. In practice, prehabilitation programs may include cardiovascular and resistance training exercises, nutritional advice designed to support an increase in lean body mass, the introduction of coping strategies to deal with surgical anxiety, smoke cessation support, treating preoperative anemia and other modifiable risk factors [26]. An increasing number of studies support the safety, feasibility and efficacy of multimodal prehabilitation to improve surgical outcomes in cancer patients undergoing major abdominal and cardiothoracic surgery [27]. The benefits range from lower rate of postoperative complications, to less deterioration of physical function and better quality of life [28]. However, this evidence comes mainly from high-income countries and thus, there is an urgent need to test the potential of prehabilitation programs in African countries.
Decreasing morbidity and mortality following surgery in Africa will require adequate perioperative optimization and better postoperative care planning. Preoperative diagnosis of comorbidities and social habits that are considered risk conditions for surgery should be identified throughout appropriate risk assessment tools. Patients considered to be at high-risk for complications following surgery should be proposed for prehabilitation to increase their preoperative functional reserve and enhance recovery following surgical treatment. The knowledge about most common surgical complications should be used to anticipate postoperative burden, care needs and organize available resources in advance. We believe that this approach to perioperative care will play a decisive role in sub-Saharan Africa in changing surgical morbidity and mortality for better.
The authors declare no competing interests.
AVF and LLS were responsible for the primary conception and design of the article with input from co-authors; AVF, DMG and LLS prepared initial drafts of the article. Additions, modifications and critical revisions for the relevant intellectual content of the report were performed by AVF, DMG, JC, NCR, VC, LVL, PMC and LLS, including final approval of the version to be published. All the authors have read and agreed to the final manuscript.
We thank Professor Bruce M Biccard of Nelson R Mandela School of Medicine, University of Kwazulu-Natal, South Africa, for his critical reading of the manuscript.
Figure 1: stratification of measures to decrease perioperative morbidity and mortality in surgical patients
- Biccard BM, Madiba TE, Kluyts HL, Munlemvo DM, Madzimbamuto FD, Basenero A et al. Perioperative patient outcomes in the African Surgical outcomes study: a 7-day prospective observational cohort study. Lancet. 2018;391(10130):1589-1598. PubMed | Google Scholar
- Fehlberg T, Rose J, Guest GD, Watters D. The surgical burden of disease and perioperative mortality in patients admitted to hospitals in Victoria, Australia: a population-level observational study. BMJ Open. 2019;9(5):e028671. PubMed | Google Scholar
- The International Surgical Outcomes Study group. Global patient outcomes after elective surgery: a prospective cohort study in 27 low-, middle- and high-income countries. Br J Anaesth. 2016;117(5):601-609. PubMed | Google Scholar
- Clack L, Willi U, Berenholtz S, Aiken AM, Allegranzi B, Sax H. Implementation of a surgical unit-based safety program in African hospitals: a multicentre qualitative study. Antimicrob Resist Infect Control. 2019;8:91. PubMed | Google Scholar
- Abahuje E, Sibomana I, Rwagahirima E, Urimubabo C, Munyaneza R, Rickard J. Development of an acute care surgery service in Rwanda. Trauma Surg Acute Care Open. 2019;4(1):e000332. PubMed | Google Scholar
- Bishop DG, Gibbs MW, Dyer RA. Post-caesarean delivery analgesia in resource-limited settings: a narrative review. Int J Obstet Anesth. 2019;40:119-127. PubMed | Google Scholar
- White MC, Randall K, Capo-Chichi NFE, Sodogas F, Quenum S, Wright K et al. Implementation and evaluation of nationwide scale-up of the surgical safety checklist. Br J Surg. 2019;106(2):e91-e102. PubMed | Google Scholar
- Clack L, Willi U, Berenholtz S, Aiken AM, Allegranzi B, Sax H. Implementation of a surgical unit-based safety programme in African hospitals: a multicentre qualitative study. Antimicrob Resist Infect Control. 2019;8:91. PubMed | Google Scholar
- Rayne S, Burger S, Straten SV, Biccard B, Phaahla MJ, Smith M. Setting the research and implementation agenda for equitable access to surgical care in South Africa. BMJ Glob Health. 2017;2(2):e000170. PubMed | Google Scholar
- Foy KE, Pearson J, Kettley L, Lal N, Blackwood H, Bould MD. Four early warning scores predict mortality in emergency surgical patients at University Teaching Hospital, Lusaka: a prospective observational study. Can J Anaesth. 2020;67(2):203-212. PubMed | Google Scholar
- du Toit L, Bougard H, Biccard BM. The developing world of pre-operative optimisation: a systematic review of Cochrane reviews. Anaesthesia. 2019;74(1):89-99. PubMed | Google Scholar
- Hewitt-Smith A, Bulamba F, Olupot C, Musana F, Ochieng JP, Lipnick MS et al. Surgical outcomes in eastern Uganda: a one-year cohort study. South Afr J Anaesth Analg. 2018;24(5):122-127. Google Scholar
- Moonesinghe SR, Mythen MG, Das P, Rowan KM, Grocott MP. Risk stratification tools for predicting morbidity and mortality in adult patients undergoing major surgery: qualitative systematic review. Anesthesiology. 2013;119(4):959-981. PubMed | Google Scholar
- Oliver CM, Walker E, Giannaris S, Grocott MP, Moonesinghe SR. Risk assessment tools validated for patients undergoing emergency laparotomy: a systematic review. Br J Anaesth. 2015;115(6):849-860. PubMed | Google Scholar
- Osinaike B, Ayandipo O, Onyeka T, Alagbe-Briggs O, Mohammed A, Oyedepo O et al. Nigerian surgical outcomes - report of a 7-day prospective cohort study and external validation of the African surgical outcomes study surgical risk calculator. Int J Surg. 2019;68:148-56. PubMed | Google Scholar
- Kluyts HL, Manach YL, Munlemvo DM, Madzimbamuto F, Basenero A, Coulibaly Y et al. The ASOS surgical risk calculator: development and validation of a tool for identifying African surgical patients at risk of severe postoperative complications. Br J Anaesth. 2018;121(6):1357-1363. PubMed | Google Scholar
- Spence RT, Chang DC, Chu K, Panieri E, Mueller JL, Hutter MM. An online tool for global benchmarking of risk-adjusted surgical outcomes. World J Surg. 2017;41(1):24-30. PubMed | Google Scholar
- Allan N, Godfrey IM, Edwin GM. Validation of POSSUM, P-POSSUM and the surgical risk scale in major general surgical operations in Harare: a prospective observational study. Ann Med Surg (Lond). 2019;41:33-39. PubMed | Google Scholar
- Ntobeko N. Guest editorial perioperative evaluation of patients who are due to undergo surgery. S Afr Med J. 2018;108(5):367-368. Google Scholar
- Macpherson DS, Parenti C, Nee J, Petzel RA, Ward H. An internist joins the surgery service: does comanagement make a difference. J Gen Intern Med. 1994;9(8):440-444. PubMed | Google Scholar
- Phy MP, Vanness DJ, Melton LJ, Long KH, Schleck CD, Larson DR et al. Effects of a hospitalist model on elderly patients with hip fracture. Arch Intern Med. 2005;165(7):796-801. PubMed | Google Scholar
- Vazirani S, Lankarani-Fard A, Liang LJ, Stelzner M, Asch SM. Perioperative processes and outcomes after implementation of a hospitalist-run preoperative clinic. J Hosp Med. 2012;7(9):697-701. PubMed | Google Scholar
- Auerbach AD, Wachter RM, Cheng HQ, Maselli J, McDermott M, Vittinghoff E et al. Comanagement of surgical patients between neurosurgeons and hospitalists. Arch Intern Med. 2010;170(22):2004-2010. PubMed | Google Scholar
- Auerbach AD, Rasic MA, Sehgal N, Ide B, Stone B, Maselli J. Opportunity missed: medical consultation, resource use and quality of care of patients undergoing major surgery. Arch Intern Med. 2007;167(21):2338-2344. PubMed | Google Scholar
- Minnella EM, Carli F. Prehabilitation and functional recovery for colorectal cancer patients. Eur J Surg Oncol. 2018;44(7):919-926. PubMed | Google Scholar
- Blyth VW, Moorthy K. Prehabilitation: preparing patients for surgery. BMJ. 2017;358:j3702. PubMed | Google Scholar
- Kamarajah SK, Bundred J, Weblin J, Tan BHL. Critical appraisal on the impact of preoperative rehabilitation and outcomes after major abdominal and cardiothoracic surgery: a systematic review and meta-analysis. Surgery. 2020;167(3):540-549. PubMed | Google Scholar
- van Rooijen S, Carli F, Dalton S, Thomas G, Bojesen R, Guen ML et al. Multimodal prehabilitation in colorectal cancer patients to improve functional capacity and reduce postoperative complications: the first international randomized controlled trial for multimodal prehabilitation. BMC. 2019 Jan 22;19(1):98. PubMed | Google Scholar