Challenges and barriers to HIV service uptake and delivery along the HIV care cascade in Cameroon
Albert Frank Zeh Meka, Serge Clotaire Billong, Ismael Diallo, Ousseni Wendlassida Tiemtore, Brian Bongwong, Georges Nguefack-Tsague
Corresponding author: Albert Frank Zeh Meka, National Aids Control Committee, Ministry of Public Health, Yaounde, Cameroon 
Received: 06 May 2019 - Accepted: 19 Jan 2020 - Published: 27 May 2020
Domain: HIV epidemiology
Keywords: Cascade, stock-outs, barriers
©Albert Frank Zeh Meka et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Albert Frank Zeh Meka et al. Challenges and barriers to HIV service uptake and delivery along the HIV care cascade in Cameroon. Pan African Medical Journal. 2020;36:37. [doi: 10.11604/pamj.2020.36.37.19046]
Available online at: https://www.panafrican-med-journal.com//content/article/36/37/full
Research 
Challenges and barriers to HIV service uptake and delivery along the HIV care cascade in Cameroon
Challenges and barriers to HIV service uptake and delivery along the HIV care cascade in Cameroon
Albert Frank Zeh Meka1,&, Serge Clotaire Billong1,2, Ismael Diallo3,4, Ousseni Wendlassida Tiemtore4, Brian Bongwong2, Georges Nguefack-Tsague2
1National Aids Control Committee, Ministry of Public Health, Yaounde, Cameroon, 2Faculty of Medicine and Biomedical Sciences, University of Yaounde, Yaounde, Cameroon, 3Centre Hospitalier Universitaire Yalgado Ouedraogo, Ouagadougou, Burkina-Faso, 4Initiatives Conseil International-Santé (ICI-Santé), Ouagadougou, Burkina-Faso
&Corresponding author
Albert Frank Zeh Meka, National Aids Control Committee, Ministry of Public Health, Yaounde, Cameroon
Introduction: the year 2017 marked a transition period with the end of the implementation of Cameroon´s 2014-2017 HIV/AIDS National Strategic Plan (NSP) and the development of the 2018-2022 NSP. We assessed barriers and challenges to service delivery and uptake along the HIV care cascade in Cameroon to inform decision making within the framework of the new NSP, to achieve the UNAIDS 90-90-90 target.
Methods: we conducted a cross sectional descriptive study nationwide, enrolling HIV infected patients and staff. Data were collected on sociodemographic characteristics, HIV testing, antiretroviral therapy and viral load testing delivery and uptake and factors that limit their access.
Results: a total of 137 staff and 642 people living with HIV (PLHIV) were interviewed. Of 642 PLHIV with known status, 339 (53%) repeated their HIV test at least once, with range: 1-10 and median: 2 (IQR: 1-3). Having attained secondary level of education (OR: 2.07, 95% CI: 1.04-4.14; P=0.04) or more (OR: 2.91, 95% CI: 1.16-7.28; P=0.02) were significantly associated with repeat testing. Psychological (refusal of service uptake and existence of HIV), community-level (stigmatization and fear of confidentiality breach) and commodity stock-outs "HIV test kits (21%), antiretrovirals (ARVs) (71.4%), viral load testing reagents (100%)" are the major barriers to service delivery and uptake along the cascade.
Conclusion: we identified individual, community-level, socio-economic and health care system related barriers which constitute persistent bottlenecks in HIV service delivery and uptake and a high rate of repeat testing by PLHIV with known status. Addressing all these accordingly can help the country achieve the UNAIDS 90-90-90 target.
The continuum of care (also referred to as the cascade of care) for successful HIV treatment includes: HIV testing, linkage, engagement in care, and retention on antiretroviral therapy (ART) with viral suppression as the ultimate clinical goal to improve individual health outcomes of people living with HIV/AIDS (PLHIV) and reduce HIV acquisition and transmission, thus conferring community and public health benefits [1]. Over the last decade, millions of individuals in sub-Saharan Africa (SSA) have started ART, however, low HIV testing rates and losses between the point of testing and the initiation of ART have mitigated this success, and the majority of people in need of treatment are not receiving it [2]. Several studies have described the performance of service delivery and uptake or dwelled on barriers to uptake of services and attrition along the HIV care continuum cascade for the general population and in pregnant women across the world including SSA [3-11]. The factors identified are individual, socio-economic or community-level or health system related factors. Efforts are needed to optimize the HIV care continuum in order to achieve the 90-90-90 target, that is by 2020, 90% of all people living with HIV will know their HIV status, 90% of all people with diagnosed HIV infection will receive sustained antiretroviral therapy and 90% of all people receiving antiretroviral therapy will have viral suppression [12]. Key to these efforts will be to overcome and/or eliminate legal, social, environmental, and structural barriers that prevent PLHIV from accessing and utilizing HIV services [13]. In 2015, Cameroon subscribed to the UNAIDS 90-90-90 ambitious target which aims to end the AIDS epidemic in 2030. Several efforts have therefore been undertaken by the Cameroonian government to provide HIV services to all Cameroonians by breaking financial, social and geographical barriers in order to achieve this target in 2020. As of 31st December 2016, 2 418 986 HIV tests were done but the number of people tested is not known. Also, the ART coverage amongst PLHIV was 32% [14] far below the 60% objective set by the 2014-2017 NSP. Furthermore, only 46 993 viral load tests were done within the same year amongst the 168 349 PLHIV on ART at the end of the year 2015. As we get close to the year 2020, to achieve the 90-90-90 ambitious target, ART has experienced a rapid scale-up with the adoption of the test and treat strategy, systematic offer of HIV-testing in health facilities, decentralisation through the creation of more ART and option B+ sites across the country, community dispensation of ART to stable PLHIV on ART and the enrollment of 8 reference laboratories to conduct viral load (VL) testing covering the 10 regions of the country and reduction of the cost of the test. It is in this context that we sought to explore factors associated with attrition along the HIV care cascade in an attempt to contribute in the reorientation and reinforcement of strategies within the framework of the new 2018-2022 HIV/AIDS NSP, so as to achieve the UNAIDS ambitious 90-90-90 target. More specifically, we sought to determine the magnitude of repeat testing among PLHIV with a previous HIV diagnosis.
Study setting: the study was conducted on the national scale in health facilities (ART and PMTCT services, laboratories, and outpatient department), regional drug procurement/distribution institutions (regional funds for the promotion of health (RFPH)), regional technical groups (RTG) for the fight against AIDS and reference laboratories for viral load testing (a total of 8 laboratories were operating as of 31 December 2016). ART and PMTCT sites provide HIV screening and treatment services to the general population and pregnant women respectively. There are two categories of ART sites: approved treatment centers (ATC) which are found in health facilities of 1st, 2nd and 3rd, categories of our health system and HIV management units (HMU) found in 4th, 5th and 6th category facilities.
Study design and population: we conducted a cross-sectional and descriptive study from 8th May to the 10th of July 2017 to assess service delivery and uptake at the various stages of the HIV care cascade. Participants for this study were PLHIV enrolled on ART and staff in: i) health facilities (ART services, PMTCT services, laboratories and outpatient department): medical doctors and nurses, ii) national and regional drug procurement and distribution institutions (pharmacist or ware house in-charge) and iii) HIV viral load testing reference laboratories (laboratory in-charge), iv) mobile HIV testing units in RTGs (psychosocial support agents).
Sampling and data collection: we sampled sites distributed around the 8 national viral load reference laboratories, a total of 193 facilities were involved. The list of facilities distributed around the country with the numbers of regular PLHIV ART users per site as of 31st December 2016 were obtained from the National Aids Control Committee (NACC). The selection of health facilities was then done by stratified sampling with probability proportional to the size of regular ART users per site. As variables for stratification we used the regions (all 10 regions of the country were involved), the location (urban/rural), type of ART site in the facility (ACT/HMU) and ownership (private/public). A total of 25 sites were sampled nationwide of which 12 rural and 13 urban, 9 ATC and 16 HMU, 19 public and 6 private. The sample size of PLHIV was calculated using the formula:
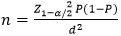 [15]
[15]
Statistical analysis: at the end of data collection, questionnaires were sorted out to check for errors and any missing information. Data was entered and analysed with Epi info 7 software. Data double checking was done before analysis to ensure consistency and accuracy. Some variables were recoded to facilitate analysis. We used descriptive statistics in the form of proportions (%) for categorical data, mean if data followed normal distribution with standard deviation otherwise median with interquartile range for continuous data. Chi-square and Fisher´s exact test (if expected counts less than 5) were used to compare proportions and associations with outcome variables (repeat testing and ART discontinuation). For bivariate analyses, associations between outcome variable and covariates were quantified using odd ratios (OR) with 95% confidence interval (CI). Variables found to be associated with p-values less than 0.25 were reexamined in a logistic regression model. P-values less than 0.05 were considered statistically significant.
Ethical considerations: administrative authorizations (hospital, regional fund and viral load testing laboratory directors, regional coordinators for the fight against HIV) were obtained. Ethical clearance was obtained from the national ethics committee for human research. Informed consent was also obtained from study participants and each was assigned a unique identification code which was written on the questionnaire.
A total of 779 participants were interviewed, of which 137 staff and 642 PLHIV. Amongst PLHIV (N=642), the 30-39 (30%) and 40-49 years (35%) age groups and women (68%) were most represented. The age ranged from 12 to 78 years and the mean age was 41±11.03 years. Most PLHIV interviewed were married (43%), and more than half attended secondary school while more than 1/4 had never been to school (Table 1). As shown in Table 2 we assessed repeat of HIV testing among PLHIV with a previous HIV diagnosis within the first 3 months of HIV diagnosis and the lifetime HIV positive testing. More than half of PLHIV repeated their HIV test since they were known HIV positive (53%, 339/642) while 45% (287/642) repeated their test within the first 3 months of HIV diagnosis. Overall, most retesters repeated HIV tests 1-3 times (Range 1-10), with a median of 2 (IQR:1-2) tests within the first 3 months of HIV diagnosis. Patients with secondary school (OR: 2.07, 95% CI: 1.04-4.14; P=0.04) and higher level of education (OR: 2.91, 95% CI 1.16-7.28; P=0.02) were significantly more likely to be repeat testers compared to those with no education.
Barriers to HIV testing were identified at all levels: health system, community-level and patient-level. PLHIV (N=995) reported stigmatisation (53%), fear of confidentiality breach (21%) and insufficient counselling (11%) as major barriers to HIV screening uptake while staff (N=146) reported refusal by patients to get tested (25%), HIV test kits stock-outs (21%), and fear to know HIV status (10%) as the major barriers to HIV screening delivery. Also, 5% (52/995) of PLHIV and 8% (11/146) of staff in health facilities reported the cost of the HIV test was a barrier to HIV testing (Table 3). On assessment of barriers to ART uptake by PLHIV (n=986), stigmatization (51%), fear of confidentiality breach (20%) and insufficient counseling (12%) were the most reported while staff reported refusal by patients to initiate ART (2/5) and their clinical condition (2/5) as the most common barriers to ART delivery (Table 4). In health facilities and regional drug procurement and distribution institutions, most staff interviewed (20/28, 71.4%) reported pediatric ARV stock-outs within the last 3 months before the study and the length of stock-outs ranged from 7 to 60 days (Table 5).
Concerning viral load testing delivery and uptake, of the 7 VL reference laboratories surveyed, 6 reported interruption of viral load testing delivery within the last 12 months before the study and all these laboratories encountered reagent stock-out as main reason for interruption. Other reasons included VL testing machine breakdown (1/6) and staff shortages (1/6) (Table 6). Despite the fact that VL testing was recently instituted as a work-up for follow-up, 71% (456/642) of patients interviewed were aware of VL testing and 78% of these (358/456) had performed a VL test at least once since they initiated ART. Viral load testing reagent stock-out (49%), viral load testing machine breakdown (12%), lack of information/ignorance (9%) and distance to laboratory (6%) were the most reported barriers to VL testing delivery (Table 7). We assessed staff availability for the delivery of services along the HIV care cascade, of 106 staff interviewed in facilities, 53% (56/106) reported staff shortage.
In our study we found a high proportion of repeat HIV testing amongst PLHIV who know their status, especially within the first 3 months following initial diagnosis. Our reporting system double counts all the HIV positive individuals and sums them, thus inflating the number of PLHIV making it appear that there is a greater loss to follow-up between testing positive and enrolling in care than actually exists. This has a serious impact on planning and response to the HIV epidemic as resources might be wasted. Contrary to our findings, repeat/multiple testing has been mostly reported in people with prior HIV negative or unknown statuses [16-20]. Secondary and higher levels of education were associated with increasing odds of ever being tested, similar to findings in studies in SSA and Europe [16,17,20]. Sex was not associated with repeat HIV testing in our study as reported by Matković Puljić et al. in Croatia [16] and Hensen et al. in Zambia [17]. Forty per cent (40%) of people living with HIV are not aware of their HIV-positive status [21]. Refusal to get tested, HIV tests kits stock-outs, stigmatization and fear of confidentiality breach were the most reported barriers to HIV testing, consistent with reports and findings in systematic reviews and studies in countries in SSA [4,22-28].
This study reveals a wide spectrum of barriers to ART access reported by both patients and staff which undermine the ART programme. These factors are individual, community-level, socio-economic and health-care system related. Our findings are similar to those reported in other systematic reviews and studies in sub-Saharan Africa [5,8,10,28]. The benefits of virological monitoring for patients on ART are well established and include the ability to diagnose adherence problems and treatment failure, and optimize therapy to support reduced transmission [29]. Routine virological monitoring is rarely available in most high-HIV prevalence settings. Of the 7 reference viral load testing laboratories surveyed, 6 reported a break-off of viral load testing performance within 12 months before the study and all reported reagent stock-outs as the main factor. A wide range of factors, individual, socio-economic and health care system-related were reported in our study. In accordance with our results, a systematic review in low and middle income countries (LMIC) by Roberts et al. [30] revealed that a recent survey by WHO targeting 122 LMICs found that only 20% of ART patients receive VL testing. In the LMICs surveyed, there were only 2 VL instruments, on average, per 8 706 people on ART, with 10% of these machines not in operation because they had not yet been installed or required repair, or due to lack of reagents and the absence of staff training. Findings from an in-depth qualitative survey of experts based in India, Kenya, Malawi, South Africa, and Zimbabwe found that in Malawi, with >400 000 patients on ART, only 37 000 received a VL test in 1 year. Respondents cited financial constraints as a key reason for incomplete or slow implementation. In addition, insufficient and overburdened healthcare professionals, poor training and lack of knowledge, and weak transport and laboratory systems were all considered barriers to scale-up of VL testing. Most of the health facilities surveyed reported staff shortage in HIV health services delivery. The various steps in the cascade are heavy enough and need adequate staffing. Staffing problems in HIV service delivery have been reported in systematic reviews involving SSA [8,31].
As Cameroon is scaling-up the fight against HIV by adhering to the UNAIDS 90-90-90 target with the objective to end the AIDS epidemic in 2030, there are still a lot of challenges and barriers to overcome to meet this target. Individual, community-level, socio-economic and health system and facility-related barriers constitute persistent bottlenecks in service delivery along the HIV care cascade which can undermine the achievement of the 90-90-90 target in 2020. Psychological factors (refusal of HIV existence, to get tested and to take ART), community-level factors (stigmatization and fear of confidentiality breach) and commodity stock-outs (HIV tests, ARV, viral load testing reagents) appear to be the major barriers to service delivery and uptake along the cascade. Repeat testing and staff shortage constitute major challenges to the reporting system and service delivery respectively. Findings from this study inform decision making within the framework of Cameroon´s 2018-2022 HIV/AIDS national strategic plan, the challenges and barriers identified should be taken into consideration in its implementation for better decision-making, and common efforts should be made by patients, staff and the decision-makers for successful achievement of the 90-90-90 target.
What is known about this topic
- There exist barriers to HIV testing, access to antiretroviral therapy and viral load testing at the patient-level, community-level and health-system;
- Staffing shortage in the health system is a challenge for HIV service delivery.
What this study adds
- There is a high rate of repeat testing among PLHIV with a previous HIV diagnosis which is not properly captured by the reporting system. Our data collecting system is still mainly paper-based, the use of unique identifier code, case based surveillance and electronic medical records therefore appear as necessities. There is also a need to understand and better address reasons for repeat testing among PLHIV with known status.
The authors declare no competing interests.
Albert Frank Zeh Meka, Serge Clotaire Billong, Ismael Diallo, and Georges Nguefack-Tsague designed the study, coordinated data collection and interpretation of data and revised the manuscript. Albert Frank Zeh Meka did the data analysis and drafted the manuscript. Ousseni Wendlassida Tiemtore contributed to the design of the study and interpretation of data and revised the manuscript, Brian Bongwong contributed to the design of the study, data collection and interpretation of data and revised the manuscript. All authors approved the final version of manuscript.
We are grateful to all staff in the various institutions and facilities that were involved in the study and all the study participants that consented to participate in the study.
Table 1: socio-demographic characteristics of patients
Table 2: assessment of repeat testing among PLHIV with known status (n=642)
Table 3: barriers to HIV testing uptake and delivery
Table 4: barriers to ART uptake and delivery
Table 5: history of ARV stock-outs in ART sites and supplying institutions
Table 6: history of and reasons for interruption of viral load testing activities in reference laboratories within the last 12 months before the study
Table 7: barriers to viral load testing
- Gardner E, McLees M, Steiner J, Del Rio C, Burman W. The spectrum of engagement in HIV care and its relevance to test-and-treat strategies for prevention of HIV infection. Clin Infect Dis. 2011;52(6): 793-800. PubMed | Google Scholar
- Rosen S, Fox M. Retention in HIV care between testing and treatment in sub-Saharan Africa: a systematic review. PloS Med. 2011;8(7):e1001056. PubMed | Google Scholar
- Kranzer K, Govindasamy D, Ford N, Johnston V, Lawn SD. Quantifying and addressing losses along the continuum of care for people living with HIV infection in sub-Saharan Africa: a systematic review. J Int AIDS Soc. 2012;15(2):17383. PubMed | Google Scholar
- Maurice Musheke, Harriet Ntalasha, Sara Gari, Oran McKenzie, Virginia Bond, Adriane Martin-Hilber et al. A systematic review of qualitative findings on factors enabling and deterring uptake of HIV testing in Sub-Saharan Africa. BMC Public Health. 2013;13:220. PubMed | Google Scholar
- Lankowski A, Siedner M, Bangsberg D, Tsai A. Impact of geographic and transportation-Related Barriers on HIV Outcomes in Sub-Saharan Africa: a systematic review. AIDS Behav. 2014;18(7): 1199-1223. PubMed | Google Scholar
- Hodgson I, Plummer ML, Konopka SN, Colvin CJ, Jonas E, Albertini J et al. A systematic review of individual and contextual factors affecting ART initiation, adherence, and retention for HIV-infected pregnant and postpartum women. PLoS One. 2014;9(11):e111421. PubMed | Google Scholar
- Kirakoya-Samadoulougou F, Jean K, Maheu-Giroux M. Uptake of HIV testing in Burkina Faso: an assessment of individual and community-level determinants. BMC Public Health. 2017;17(1):486. PubMed | Google Scholar
- Gourlay A, Birdthistle I, Mburu G, Iorpenda K, Wringe A. Barriers and facilitating factors to the uptake of antiretroviral drugs for prevention of mother-to-child transmission of HIV in sub-Saharan Africa: a systematic review. J Int AIDS Soc. 2013;16(1): 18588. PubMed | Google Scholar
- Wachira J, Naanyu V, Genberg B et al. Health facility barriers to HIV linkage and retention in Western Kenya. BMC Health Serv Res. 2014;14:646. PubMed | Google Scholar
- Kulkarni S, Hoffman S, Elul B. Identifying perceived barriers along the HIV care continuum: findings from providers, peer educators, and observations of provider-patient interactions in Ethiopia. J Int Assoc Provid AIDS Care. 2016;15(4): 291-300. PubMed | Google Scholar
- Dombrowski JC, Simoni JM, Katz DA, Golden MR. Barriers to HIV care and treatment among participants in a public health HIV care relinkage program. AIDS Patient Care STDS. 2015;29(5):279-287. PubMed | Google Scholar
- UNAIDS. 90-90-90: an ambitious treatment target to help end the AIDS epidemic. 2014. Geneva: UNAIDS. Accessed 18th July 2016.
- Granich R, Gupta S, Montaner J, Williams B, Zuniga J. Pattern, determinants and impact of HIV spending on care and treatment in 38 High-burden Low-and Middle-Income countries. J Int Assoc Provid AIDS Care. 2016; 15(2):91-100. PubMed | Google Scholar
- Comit� National de lutte contre le VIH/SIDA. Rapport 2016 des activit�s de lutte contre le VIH, le Sida et les IST au Cameroun. Mars 2016. Comit� National de lutte contre le VIH/SIDA. Accessed 18th July 2016.
- Charan J, Biswas T. How to calculate sample size for different study designs in medical research?. Indian J Psychol Med. 2013;35(2):121-126. PubMed | Google Scholar
- Matković Puljić V, Kosanović Ličina ML, Kavić M, Nemeth Blažić T. Repeat HIV testing at voluntary testing and counseling centers in Croatia: successful HIV prevention or failure to modify risk behaviors?. PloS one. 2014;9(4):e93734. PubMed | Google Scholar
- Hensen B, Lewis JJ, Schaap A, Tembo M, Vera-Hern�ndez M, Mutale W et al. Frequency of HIV-testing and factors associated with multiple lifetime HIV-testing among a rural population of Zambian men. BMC Public Health. 2015;15:960. PubMed | Google Scholar
- Raphael Isingo, Alison Wringe, Jim Todd, Mark Urassa, Doris Mbata, Griter Maiseli et al. Trends in the uptake of voluntary counselling and testing for HIV in rural Tanzania in the context of the scale up of antiretroviral therapy. Trop Med Int Health. 2012;17(8): e15-e25. PubMed | Google Scholar
- Susan Regan, Elena Losina, Senica Chetty, Janet Giddy, Rochelle P Walensky, Douglas Ross et al. Factors associated with self-reported repeat HIV testing after a negative result in Durban, South Africa. PloS one. 2013; 8(4): e62362. PubMed | Google Scholar
- Kudakwashe Collin Takarinda, Lydia Kudakwashe Madyira, Mutsa Mhangara, Victor Makaza, Memory Maphosa-Mutsaka, Simbarashe Rusakaniko et al. Factors Associated with Ever Being HIV-Tested in Zimbabwe: an extended analysis of the Zimbabwe demographic and health survey (2010-2011). PloS one. 2016;11(1):e0147828. PubMed | Google Scholar
- UNAIDS. Prevention gap report. 2016.Accessed 1st August 2017
- Matovu J, Makumbi F. Expanding access to voluntary HIV counselling and testing in sub-Saharan Africa: alternative approaches for improving uptake, 2001-2007. Trop Med Int Health. 2007;12(11): 1315-1322. PubMed | Google Scholar
- Meiberg A, Bos A, Onya H, Schaalma H. Fear of stigmatization as barrier to voluntary HIV counselling and testing in South Africa. East Afr J Public Health.2008;5(2): 49-54. PubMed | Google Scholar
- Meremo A, Mboy B, Ngilangwa D, Dulle R, Tarimo E, Urassa D et al. Barriers to accessibility and utilization of HIV testing and counseling services in Tanzania: experience from Angaza Zaidi programme. The Pan Afr Med J. 2016;23:189. Google Scholar
- Ngangue P, Gagnon M-P, Bedard E. Challenges in the delivery of public HIV testing and counselling (HTC) in Douala, Cameroon: providers perspectives and implications on quality of HTC services. BMC Int Health Hum Rights. 2017;17(1):9. PubMed | Google Scholar
- Kiptoo M, Ireri M, Mutugi M, Muthami N, Songok E. A retrospective evaluation of proficiency testing, and rapid HIV test kits stock-outs among HTC facilities within Nairobi County. East Afr Med J. 2015;92(12). Google Scholar
- Wagenaar BH, Gimbel S, Hoek R, Pfeiffer J, Michel C, Manuel J et al. Stock-outs of essential health products in Mozambique - longitudinal analyses from 2011 to 2013. Trop Med Int Health. 2014;19(7):791-801. PubMed | Google Scholar
- An S, George A, LeFevre A, Mpembeni R, Mosha I, Mohan D et al. Supply-side dimensions and dynamics of integrating HIV testing and counselling into routine antenatal care: a facility assessment from Morogoro Region, Tanzania. BMC Health Serv Res. 2015;15: 451. PubMed | Google Scholar
- Roberts T, Bygrave H, Fajardo E, Ford N. Challenges and opportunities for the implementation of virological testing in resource-limited settings. J Int AIDS Soc. 2012;15(2):17324. PubMed | Google Scholar
- Roberts T, Cohn J, Bonner K, Hargreaves S. scale-up of routine viral load testing in resource-poor settings: current and future implementation challenges. Clin Infect Dis. 2016; 62(8): 1043-1048. PubMed | Google Scholar
- Govindasamya D, Ford N, Kranzer K. Risk factors, barriers and facilitators for linkage to antiretroviral therapy care: a systematic review. AIDS. 2012; 26(16):2059-67. PubMed | Google Scholar











