Trends and factors associated with acute respiratory infection among under five children in Zambia: evidence from Zambia´s demographic and health surveys (1996-2014)
Nelia Langa Mulambya, Francis Hamaimbo Nanzaluka, Ntazana Nana Sinyangwe, Mpundu Makasa
Corresponding author: Nelia Langa Mulambya, Zambia Field Epidemiology Training Program, Lusaka, Zambia 
Received: 10 Apr 2019 - Accepted: 09 Apr 2020 - Published: 20 Jul 2020
Domain: Epidemiology,Nursing education,Public Health Nursing
Keywords: Acute respiratory infection, children, under-five underweight trends, Zambia
©Nelia Langa Mulambya et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Nelia Langa Mulambya et al. Trends and factors associated with acute respiratory infection among under five children in Zambia: evidence from Zambia´s demographic and health surveys (1996-2014). Pan African Medical Journal. 2020;36:197. [doi: 10.11604/pamj.2020.36.197.18799]
Available online at: https://www.panafrican-med-journal.com//content/article/36/197/full
Research 
Trends and factors associated with acute respiratory infection among under five children in Zambia: evidence from Zambia´s demographic and health surveys (1996-2014)
Trends and factors associated with acute respiratory infection among under five children in Zambia: evidence from Zambia´s demographic and health surveys (1996-2014)
Nelia Langa Mulambya1,2,3,&, Francis Hamaimbo Nanzaluka1,2,4, Ntazana Nana Sinyangwe2, Mpundu Makasa2
1Zambia Field Epidemiology Training Program, Lusaka, Zambia, 2School of Public Health, University of Zambia, Lusaka, Zambia, 3Zambia National Public Health Institute, Lusaka, Zambia, 4Ministry of Health, National Tuberculosis and Leprosy Program, Lusaka, Zambia
&Corresponding author
Nelia Langa Mulambya, Zambia Field Epidemiology Training Program, Lusaka, Zambia
Introduction: acute respiratory infection (ARI) is one of the leading causes of morbidity and mortality among children under the age of five years globally accounting for 16% of deaths. In Zambia, ARI accounts for 30-40% of children's outpatient attendance and 20-30% of hospital admissions. We assessed trends and factors associated with ARI among under-five children in Zambia from 1996 to 2014.
Methods: we analysed the Zambia demographic and health survey data for 1996, 2002, 2007 and 2014 of under five children and their mothers. We extracted data using a data extraction tool from the women's file. We analysed trends using chi square for trends. We conducted a complex survey multivariable logistic regression analysis, reported adjusted odds ratios (AOR) 95% confidence intervals (CI) and p-values.
Results: we included a total of 6,854 and 2,389 (8%) had symptoms consistent with ARI. A 2% upward trend was noted between the 1996 and 2002 surveys but a sharp decline of 10% occurred in 2007. The chi2 trend test was significant p < 0.001. Children whose mothers had secondary or higher education were less likely to have ARI (AOR 0.30 95% CI 0.15-0.58) compared to those with no education. Underweight children had 1.50 times increased odds of having ARI (AOR 1.50 95% CI 1.25 - 1.68) compared with children who were not. Use of biomass fuels such as charcoal (AOR 2.67 95% CI 2.09 - 3.42) and wood (2.79 95% CI 2.45 -3.19) were associated with high odds for ARI compared to electricity.
Conclusion: the prevalence of ARI has declined in Zambia from 1996 to 2014. Factors associated with occurrence of ARI included being a child under one year, underweight, use of biomass fuel such as charcoal and wood. Interventions to reduce the burden of ARI should be targeted at scaling up nutrition programs, as well as promoting use of cleaner fuels.
Acute respiratory infection is an acute disease affecting the lungs causing fluid and inflammatory waste to accumulate covering alveolar, inhibiting gaseous exchange and leading to difficult and painful breathing [1]. ARI affects all ages but the effects are particularly life threating among children under the age of five [2, 3]. Globally it is a leading cause of morbidity and mortality among under five children, in 2015, 922,000 deaths occurred accounting for 16% of all deaths in this age group [1, 4]. Low and middle income countries (LMIC) in sub-Saharan Africa and southern Asia account for half a million deaths [4, 5]. It is further estimated that six out of ten deaths among children are due to ARI. More than 160 million children develop ARI each year, 20 million get hospitalized and 1 million die [4]. WHO estimates that in developing countries, 40-60% of out-patients attendance and 30-40% of hospitalizations among children are due to ARI [1]. In Zambia, acute lower respiratory infections have remained among the major causes of morbidity and mortality.
The health management information system (HMIS) report of 2016-2017 shows ARI among the top ten causes of morbidity among under five children accounting [6] for 14% of deaths after malaria and diarrhoea [6, 7]. Thirty to forty percent (30 - 40%) of children´s out-patient attendance as well as 20-30% of hospitalizations in Zambia are due to acute respiratory infection [2]. Several factors are associated with ARI. These include age, residence and environmental factors such as, exposure to infectious diseases, malnutrition, poor hygiene and sanitation and biomass fuels and unhealthy environments [5, 8-10]. Children living in developing countries in remote regions where poverty, hunger and poor access to healthcare exist have increased risk of developing acute respiratory infections [8]. Little is known about the trends and the influence of sociodemographic, socioeconomic and environmental factors associated on ARI among under five children at country level. We sought to assess the trends of ARI across the four Zambia demographic and health survey (ZDHS) surveys and investigate sociodemographic and environmental factors associated with ARI among under five children in Zambia.
We conducted a cross-sectional study using nationally representative Zambia demographic and health surveys of 1996, 2002, 2007 and 2014. Sociodemographic, socioeconomic and environmental data from the women´s file was extracted for analysis. Details of the ZDHS design are described elsewhere [11, 12]. Sociodemographic variables included mother´s age, education level, residence, child´s age, sex, vaccination, breast feeding, vitamin A in the last 6 months and anthropometric measurements using WHO definition. Others included employment status and wealth index were also extracted, environmental variables such as household overcrowding (based on Zambia public health act) and type of cooking fuel. We defined the dependent variable ARI as occurrence of cough accompanied by short rapid breaths that was chest related occurring two weeks preceding the survey. This was a binary variable coded as 1 if child had ARI and 0 otherwise. We analysed data using statistical software, STATA version 14.0 SE (STATA corporation college station, Texas).
A weighted survey analysis was done and a design effect of 1.490 [11] was used. Cross-tabulations and Pearson´s Chi-square tests were done to examine associations of study variables and p-values of < 0.05 were considered significant. We analysed trends using Chi-square tests for trend based on the prevalence of ARI across the datasets. We conducted bivariate logistic regression analysis and multivariable logistic regression analysis to control for confounding. P-value < 0.05 were considered significant at 95% confidence level (CI). We reported unadjusted and adjusted odds ratios (AOR) to measure the strength of association. We used the F-test to determine the best fit model. Ethical approval was obtained from excellence in research ethics and science ethics committee and the national health research authority and permission to use the ZDHS data sets was obtained from the Zambia central statistical office and measure DHS website. No harm was paused to the participants since the study utilized secondary data which was already de-anonymized.
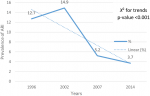
A total of 30,391 children under 5 years were analysed from 1996, 2002, 2007 and 2014 ZDHSs. A total of 6,854 (23%) children were reported to have had cough two weeks preceding the survey. Out of these, only 2,389 (8%) had symptoms consistent with ARI. Of these 50.2% were females and majority 21,274 (70%) lived in rural areas. The mean age of the children was 28 months. Majority 22,185 (73%) of the mothers were aged between 20-35 years and more than half 16,715 (55%) were working. The most common type of cooking fuel was wood 13,979 (46%) and only 19% of the households had electricity (Table 1, Table 2). Based on the trend analysis from 1996 to 2014, a 3% increase in prevalence was noted from 1996 to 2002 when the highest prevalence of ARI was recorded at 15%. The prevalence of ARI decreased drastically from 15% in 2002 to 5% in 2007. Chi square for trend was significant at p-value < 0.001 (Figure 1). The prevalence of ARI was higher in rural areas across the surveys except 2007 which showed high prevalence in the urban areas (Figure 2).
At bivariate analysis, an increase in mother´s age and higher level of education were associated with a decreased chance for ARI (odds ratio [OR] 0.63 95% confidence interval [CI] 0.50 - 0.80 P-value [p-] < 0.001) (OR 0.27 95% CI 0.17 - 0.40 p -< 0.001) compared with young mothers and those with no education (Table 3). Breast feeding children were more likely to suffer from ARI (OR1.94 95% CI 1.73-2.17 p -< 0.001) compared to those that were not. Lack of vitamin A supplementation in the last six months was 2.48 times associated with likelihood for ARI (OR 2.48 95% CI 2.22-2.77) (Table 3). Being in a crowded home was associated with increased odds for ARI (OR 1.06 95% CI 1.04-1.08 p < 0.001). The number of children five years and under in the household showed an increased chance of ARI (OR 1.10 95% CI 1.03-1.16 p-0.002). Using charcoal (OR 3.24 95% CI 1.73-2.17 p -< 0.001) for cooking had an increased odds for ARI compared to electricity (Table 3).
Multivariable analysis of factors associated with ARI among under five children: at multivariable logistic regression, children whose mothers´ were older than 20 years had a reduced odds of having ARI (AOR 0.68 95% CI 0.52-0.89 p- 0.005) as compared to mothers younger than 20 years after controlling for education level, underweight, child´s age, vitamin A and overcrowding. Children whose mothers had higher education were 74% less likely (AOR 0.30 95% CI 0.15-0.58 p -< 0.001) odds of ARI. Children who were underweight had 1.50 times the odds of ARI compared to those who were not (AOR 1.50 95% CI 1.28 -1.68 p -< 0.001). Using charcoal for cooking was associated with 2.67 times increased odds of ARI (AOR 2.67 95% CI 2.09-3.4 p-<0.001) as compared to using electricity (Table 4).
The main aim of the study was to assess the trends of ARI and determine factors associated with ARI in Zambia. In the current analysis, age of child, underweight, mothers´ education level, vitamin A and type of cooking fuel were among factors associated with ARI. Findings from our study showed a reduction in the prevalence of ARI from 13% in 1996 to 4% in 2014. These findings are in agreement with global efforts to reduce under five mortality and efforts to end child deaths due to ARI and diarrhoea [1, 4]. The reduction that was noted in the prevalence of ARI can be attributed to the efforts by the Zambian government through the introduction of the haemophilus influenza vaccine (HibHepB) in 2005 and the pneumococcal conjugate vaccine (PCV10) in 2013 [11, 13]. The findings of our study are consistent with the study conducted in Nigeria which saw a reduction in the prevalence of ARI related to the introduction of new vaccines to the routine EPI schedule [14]. Analysis of trends by residence showed a higher prevalence in the rural area as compared to urban areas. Similar findings were seen in a study conducted in Ghana which showed a higher prevalence of ARI in the rural area [15]. This finding can be related to the poor socioeconomic status associated with rural areas including overcrowded homes, mostly poor ventilation and use of unclean fuels. The opposite was noted in 2007 ZDHS were the prevalence was higher in urban areas. However, a contrary finding was observed in the 2007 ZDHS showed a higher prevalence in the urban areas [16].
Similar results were seen from a study conducted in India which showed a higher prevalence in the urban areas [17]. The reason for this finding could be related to overcrowding that occurs in some urban settings as a result of people moving from rural to urban areas. Although vaccination plays a major role in disease prevention, it is worth noting that our study findings did not show statistically significant association between ARI morbidity and vaccination at multivariable analysis. However this was not a finding unique to this study as studies conducted in India and a meta-analysis conducted in sub-Saharan Africa have shown a reduction in the prevalence of ARI with the introduction of the PCV vaccine [5, 17, 18]. Among sociodemographic variables associated with ARI, mother´s age, education level and child´s age were associated with ARI. Maternal age was associated with reduced odds for developing ARI. This result is in line with findings from studies conducted in Brazil, Ghana, Nigeria and Tanzania which showed that children born to mothers younger than 20 years had higher odds for developing ARI compared to mothers older than 20 years [8, 15, 19, 20]. This finding can be related to the inexperience, immaturity and unpreparedness among these adolescent mothers. On the contrary, studies conducted in different parts of Ethiopia found no significant associations between mother´s age and occurrence of ARI among under five children [5, 21].
Children who were under one year old were more likely to have ARI compared to older children. The finding is consistent with studies conducted in Rwanda, Ethiopia, Ndola (Zambia) [2, 22, 23] which showed increased likelihood of ARI. The increase in the risk for ARI in this age group is most likely related to the low immunity among the children which tends to improve secondary to exposures to immunizations and generally developing resistance to infections. Children younger than 11 months may be at increased risk due to incomplete vaccinations such as PCV, DPT HibHepB and measles [19]. Contrary to our study findings as well as findings from most studies which show the most affected age group to be among the 0-11 months, a study conducted in Nigeria in 2014 found higher prevalence among children aged 10-19 months, an age which was consistent with high complementary feeding which also increases the risk of exposure to other risk factors associated with ARI [19]. ARI prevalence of 16% was noted among underweight children. This finding is consistent with studies done in Nigeria and Tanzania which equally showed a higher prevalence of ARI among children who were malnourished and of low weight for age [19, 20, 24, 25]. A study conducted at the University Teaching Hospital Zambia also showed high prevalence of ARI and diarrhoea among malnourished children [26]. This finding further underscores the effect of underweight and undernutrition on the occurrence of ARI. Underweight indicates long term malnutrition which weakens the body and increases risk for infections.
This finding is related to the low immunity of underweight children which increases their vulnerability to developing infections including acute respiratory infections [19]. Children who did not receive vitamin A in the last 6 months before the survey were more likely to have ARI. A study conducted by Ramezani et al. 2015, showed that vitamin A deficiency was associated with increased susceptibility to infection due to the changes that occur in the epithelial tissue of the lung tissue [27]. Our study showed that children with mothers having secondary or higher education were less likely to develop ARI. These findings are consistent with findings from a study conducted in Kenya, Ethiopia and Rwanda [21, 28]. This finding may be related to the fact that these mothers may have access to literature which helps them to protect their children. A study conducted in Uganda found no association between maternal education and occurrence of ARI [29]. Among variables related to housing and environmental, cooking fuel and associated with ARI. Cooking fuel was associated with increased odds for ARI, children from homes were charcoal and wood were used for cooking had increased odds for ARI as compared to those were electricity was used. These findings are similar to the findings from a multi-country study which was conducted in Africa showed that the use of charcoal coal and wood was associated with increased risk for acute respiratory infections and ARI [25, 30].
A number of studies conducted in various parts of Ethiopia have also shown increased risk for ARI among children from homes which use unclean cooking fuel such as charcoal and firewood [5, 21]. Studies conducted in India, Nepal, Rwanda and Nigeria also showed that cooking fuel such as wood and charcoal was associated with increased risk for ARI as compared to electricity [22, 24, 25, 31, 32]. These findings can be attributed to the environmental pollutants such as wood smoke and charcoal smoke which results in the production of respirable particulate matter which increases the risk for respiratory infections such as ARI in children below the age of five years [8, 19, 22, 24, 33]. Wood smoke and charcoal smoke also reduces respiratory clearance functions and increases risk for infections [32]. Our study utilized secondary data from four different surveys as such the effect of some variables noted in literature were not assessed for example, the effect of wealth index, type of roofing material. Secondly, the outcome variable ARI was based on self-report from the mothers, possible recall bias. The newest survey was at least four years at the time of the analysis. However, the study was able to chat the trends and identify the factors associated with ARI among under five children and it is hoped that these findings will help the ministry of health child health unit as well as health promotion and social determinants to plan interventions which will contribute to the reduction of ARI. The findings of the study are generalizable since we utilised nationally representative data.
A downward trend has been observed in the prevalence of ARI among under five children in Zambia. Young age, being underweight, use of cooking fuels such as wood and charcoal have been associated with increased risk for ARI. Having vitamin A supplementation, maternal education and have been found to be protective against ARI among children. There is need to introduce/scale up nutrition programs focusing on reducing malnutrition among under five children and introduction of health promotion programs to address overcrowding and use of unclean cooking fuels such as charcoal and firewood.
What is known about this topic
- The prevalence of ARI among under five children in Zambia;
- The predisposing factors to ARI at global level and strategies to end child death due to pneumonia and diarrhoea.
What this study adds
- The trends of ARI among children in Zambia;
- Using nationally representative data factors associated with ARI among under five children are identified and include biomass fuel and poor nutrition which are more common in the rural areas;
- Micronutrient supplementation such as vitamin A has been found to be protective hence it should be emphasized at all levels of healthcare.
The authors declare no competing interests.
NLM, FHN designed the study, performed the statistical analysis and data interpretation and wrote the manuscript; MM, NS contributed to the study design, analysis interpretation of data and supervision of the study. All the authors have read and agreed to the final manuscript.
U.S. president´s emergency plan for AIDS relief (PEPFAR) for the financial support. We thank Dr. Ellen Yard, Dr. Danielle Barradas, Dr. Nyambe Sinyange, Ms. Mazyanga Liwewe and Dr. Raymond Hamoonga for their mentorship. Central statistical office and measure DHS for providing the data. We would also like to appreciate the support and mentorship from the University of Zambia School of Public Health and the Zambia National Public Health Institute.
Table 1: frequency distribution of background characteristics of under five children with ARI in Zambia
Table 2: frequency distribution of housing characteristics of under-five children with ARI in Zambia
Table 3: predictor´s variables of ari among under-five children in Zambia
Table 4: multivariable analysis of factors associated with ARI among under-five children in Zambia
Figure 1: ARI trends among under five children in Zambia 1996-2014
Figure 2: ARI trends among under five children by urban rural residence in Zambia
- UNICEF. Pneumonia. 2019. Accessed on April 16 2019.
- Banda W, Mazaba-Liwewe ML, Mulenga D, Siziya S. Risk factors associated with acute respiratory infections among under-five children admitted to Arthur Davisons Children's Hospital, Ndola Zambia. Asian Pacific Journal of Health Science. 2016;3: p. 153-159. Google Scholar
- Grimwood K, Chang AB. Long-term effects of pneumonia in young children. Pneumonia. 2015;6:101-114. PubMed | Google Scholar
- WHO. Pneumonia fact sheet. WHO. 2016.
- Abuka T. Prevalence of pneumonia and factors associated among children 2-59 months in Wondo genet district sidama zone SNNPR Ethiopia. Current Paediatric. 2017;21(1):19-25. Google Scholar
- WHO. WHO country cooperation strategy at a glance: Zambia. 2017: p. 2.
- Mara D. Disease priorities in Zambia. American journal of tropical medicine hygiene. 2016;95(1):248-9. PubMed | Google Scholar
- Fonseca LEJ, Mello MJG, de Albuquerque MFPM, Lopes MIL, Serra GHC, Lima DEP et al. Risk factors for community-acquired pneumonia in children under five years of age in the post-pneumococcal conjugate vaccine era in Brazil: a case control study. BMC Pediatr. 2016;16(1):157. PubMed | Google Scholar
- Gothankar J, Doke P, Dhumale G, Pore P, Lalwani S, Quraishi S et al. Reported incidence and risk factors of childhood pneumonia in India: a community-based cross-sectional study. BMC Public Health. 2018;18(1):1111. PubMed | Google Scholar
- Jroundi I, Mahraoui C, Benmessaoud R, Moraleda C, Tligui H, Seffar M et al. Risk factors for a poor outcome among children admitted with clinically severe pneumonia to a university hospital in Rabat, Morocco. Int J Infect Dis. 2014;28:164-70. PubMed | Google Scholar
- CSO. Zambia demographic and health survey 2013-14. Central Statistical Office/Zambia, Ministry of Health/Zambia and ICF International: Rockville, Maryland, USA. 2014.
- MoH. Roadmap for accelerating reduction of maternal, newborn and child mortality, 2013-2016. 2013.
- Beres LK, Tate JE, Njobvu L, Chibwe B, Rudd C, Guffey MB et al. A preliminary assessment of rotavirus vaccine effectiveness in Zambia. Clin Infect Dis. 2016;62 Suppl 2:S175-82. PubMed | Google Scholar
- Akinyemi JO, Morakinyo OM. Household environment and symptoms of childhood acute respiratory tract infections in Nigeria, 2003-2013: a decade of progress and stagnation. BMC Infect Dis. 2018;18(1):296. PubMed | Google Scholar
- Amugsi DA, Aborigo RA, Oduro AR, Asoala V, Awine T, Amenga-Etego L. Socio-demographic and environmental determinants of infectious disease morbidity in children under 5 years in Ghana. Glob Health Action. 2015;8:29349. PubMed | Google Scholar
- CSO. Zambia demographic and health survey 2007. Central Statistical Office/Zambia and Macro International: Calverton, Maryland, USA. 2009.
- Kumar SG, Majumdar A, Kumar V, Naik BN, Selvaraj K, Balajee K. Prevalence of acute respiratory infection among under-five children in urban and rural areas of puducherry, India. J Nat Sci Biol Med. 2015;6(1):3-6. PubMed | Google Scholar
- Magitta N. Impact of Hib and pneumococcal conjugate vaccines on the epidemiology of childhood pneumonia in sub-Saharan Africa: analysis of WHO/UNICEF Data. Ecronicon. 2018: p. 7.
- Ujunwa F, Ezeonu C. Risk factors for acute respiratory tract infections in under-five children in Enugu Southeast Nigeria. Ann Med Health Sci Res. 2014;4(1):95-9. PubMed | Google Scholar
- PrayGod G, Mukerebe C, Magawa R, Jeremiah K, Török ME. Indoor air pollution and delayed measles vaccination increase the risk of severe pneumonia in children: results from a case-control study in Mwanza, Tanzania. PLoS One. 2016;11(8):e0160804. PubMed | Google Scholar
- Fekadu GA, Terefe WM, Alemie GA. Prevalence of pneumonia among under five children in Este town and the surrrounding rural kebeles, Northwest Ethiopia; a community based cross sectional study. Science Journal of Public Health. 2014;2(3):150-155. Google Scholar
- Harerimana JM, Nyirazinyoye L, Thomson DR, Ntaganira J. Social, economic and environmental risk factors for acute lower respiratory infections among children under five years of age in Rwanda. Arch Public Health. 2016;74:19. PubMed | Google Scholar
- Geberetsadik A, Worku A, Berhane Y. Factors associated with acute respiratory infection in children under the age of 5 years: evidence from the 2011 Ethiopia demographic and health survey. Pediatric Health Medical Journal. 2015;6:9-13. PubMed | Google Scholar
- Selvaraj K, Chinnakali P, Majumdar A, Krishnan IS. Acute respiratory infections among under-5 children in India: a situational analysis. J Nat Sci Biol Med. 2014;5(1):15-20. PubMed | Google Scholar
- Ramani VK, Pattankar J, Puttahonnappa SK. Acute respiratory infections among under-five age group children at urban slums of Gulbarga city: a longitudinal study. J Clin Diagn Res. 2016;10(5):LC08-13. PubMed | Google Scholar
- Munthali T, Jacobs C, Sitali L, Dambe R, Michelo C. Mortality and morbidity patterns in under-five children with severe acute malnutrition (SAM) in Zambia: a five-year retrospective review of hospital-based records (2009-2013). Arch Public Health. 2015;73(1):23. PubMed | Google Scholar
- Ramezani M, Aemmi S, Moghadam ZE. Factors affecting the rate of pediatric pneumonia in developing countries: a review and literature study. International Journal of Pediatrics. 2015;3(6):1173-1181. Google Scholar
- Onyango D, Kikuvi G, Amukoye E, Omolo J. Risk factors of severe pneumonia among children aged 2-59 months in western Kenya: a case control study. Pan African Medical Journal. 2012;13:45. PubMed | Google Scholar
- Bbaale E. Determinants of diarrhoea and acute respiratory infection among under-fives in Uganda. Australas Med J. 2011.4(7):400-9. PubMed | Google Scholar
- Buchner H, Rehfuess EA. Cooking and season as risk factors for acute lower respiratory infections in African children: a cross _sectional multi-country analysis. PLOS ONE. 2015;10(6):e0128933. PubMed | Google Scholar
- Acharya P, Mishra SR, Berg-Beckhoff G. Solid fuel in kitchen and acute respiratory tract infection among under five children: evidence from Nepal demographic and health survey 2011. J Community Health. 2015;40(3):515-21. PubMed | Google Scholar
- Admasie A, Kumie A, Worku A. Children under five from houses of unclean fuel sources and poorly ventilated houses have higher odds of suffering from acute respiratory infection in Wolaita-Sodo, Southern Ethiopia: a case-control study. J Environ Public Health. 2018;2018:9320603. PubMed | Google Scholar
- Nandasena A, Wickremasnghe RA, Nalini SA. Indoor air pollution and respiratory health of chilren in the developing world. World Journal of Clinical Paediatrics. 2013;2(2):6-15. PubMed | Google Scholar