Parasitic contamination and public health risk of commonly consumed vegetables in Ibadan-Nigeria
Oluwasola Olaiya Obebe, Olufemi Oludare Aluko, Olufarati Oludunsin Falohun, Kayode Blessing Akinlabi, ThankGod Emmanuel Onyiche
Corresponding author: Oluwasola Olaiya Obebe, Department of Veterinary Parasitology and Entomology, University of Ibadan, Ibadan, Nigeria 
Received: 03 Jun 2019 - Accepted: 03 Jun 2020 - Published: 25 Jun 2020
Domain: Epidemiology,Microbiology,Molecular Biology
Keywords: Parasites, vegetables, contamination, Ibadan-Nigeria
©Oluwasola Olaiya Obebe et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Oluwasola Olaiya Obebe et al. Parasitic contamination and public health risk of commonly consumed vegetables in Ibadan-Nigeria. Pan African Medical Journal. 2020;36:126. [doi: 10.11604/pamj.2020.36.126.19364]
Available online at: https://www.panafrican-med-journal.com//content/article/36/126/full
Research 
Parasitic contamination and public health risk of commonly consumed vegetables in Ibadan-Nigeria
Parasitic contamination and public health risk of commonly consumed vegetables in Ibadan-Nigeria
Oluwasola Olaiya Obebe1,&, Olufemi Oludare Aluko2, Olufarati Oludunsin Falohun1, Kayode Blessing Akinlabi3, ThankGod Emmanuel Onyiche4
1Department of Veterinary Parasitology and Entomology, University of Ibadan, Ibadan, Nigeria, 2Department of Community Health, College of Health Sciences, Obafemi Awolowo University, Ile Ife, Nigeria, 3Department of Veterinary Physiology, Biochemistry and Pharmacology University of Ibadan, Ibadan, Nigeria, 4Department of Veterinary Parasitology and Entomology, University of Maiduguri, Maiduguri, Nigeria
&Corresponding author
Oluwasola Olaiya Obebe, Department of Veterinary Parasitology and Entomology, University of Ibadan, Ibadan, Nigeria
Introduction: vegetables form a major component of the human diet. However, poor agronomic practices may put consumers at risk of parasitic infections. This study evaluated the parasitic contamination of vegetables grown in selected farms in Ibadan, Nigeria.
Methods: Two hundred and eigthy vegetable species: African eggplant (Solanum macrocarpon), lettuce (Lactuca sativa), cucumber (Brassica oleracea), spinach (Amaranthus cruentus), white jute (Corchorus olitorius), pumpkin (Telfaria occidentalis), green pepper (Capsicum sp.), okro (Abelmoschus esculentus), quill grass (Celosia argenta L), tomato (Lycopersicum sativus) were collected from farms within Ibadan. Samples were washed in water, and the resulting washing solution was filtered and centrifuged to concentrate the parasitic stages. Sediments were examined by iodine and modified Ziehl-Neelsen stained smears technique.
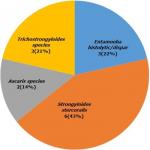
Results: parasites were detected in 14 (5.0%, 95% CI 32.6%-67.3%) of samples. The highest contaminated vegetable was white jute 32.1 (95% CI 17.9%-50.6%), followed by pumpkin 7.1(95% CI 1.9-22.6), quill grass 7.1 (95% CI 1.9-22.6) and lettuce 3.5 (95% CI 0.6-17.7). The commonest parasites were Strongyloides stercoralis larvae 42.9 (95% CI 21.3-67.4), Entamoeba histolytica/E.dipaar 21.4 (95% CI 7.5-47.5), Trichostrongylus spp 21.4 (95% CI 21.3-67.4), and Ascaris sp. 14.3 (95% CI 4.0-39.9).
Conclusion: these findings provide evidence of contamination of vegetables from farms in Ibadan with parasites of public health importance. Information on best practices should be designed, packaged and disseminated through appropriate channels to enhance positive behavior change among farmers.
The consumption of vegetables has increased in recent years because of their nutritional importance and health benefits [1]. Vegetables form a key component of a healthy diet, highly beneficial for the maintenance of health and prevention of diseases [2, 3]. However, consumption of contaminated vegetables plays a significant role in the transmission of parasitic foodborne illnesses [4, 5]. In recent times, vegetables had been shown to be contaminated with different types of enteric parasites, among which Entamoeba histolytica, Giardia duodenalis, Cryptosporidium sp. Hymenolepis sp. Taenia sp. Ascaris lumbricoides, hookworms, Enterobius vermicularis, Trichuris trichiura, Toxocara sp. and the genus Trichostrongylus have been regarded as most common [5-7]. Human Infection by these parasites can cause various clinical symptoms [8], thereby making the control of those parasites in vegetables a public health intervention priority.
Oocysts/cysts, eggs or larvae of enteric parasites can contaminate vegetables from polluted naturally composted manure, manure from grazing animals, raw, or partially treated sewage sludge, irrigation water, and wastewater from livestock operations [9, 10] and humans get infected through consumption of improperly washed or uncooked vegetables containing infective stage of these parasites [8, 11]. There may currently be an increase in parasitic diseases in human population in developing countries due to an increase in the consumption of meals in canteens, restaurants and fast food service premises, increase in the at-risk population such as the elderly, immunocompromised and children as well as changes in agronomic and vegetable processing practices [12, 13]. Several studies have reported a high prevalence of intestinal parasites on vegetable worldwide, such as found in the Philippines [14], Iran [15], Ghana [12], Kenya [16] and in Nigeria [17, 18]. However, Information on the level of contamination by parasites on vegetables from farms are lacking, especially in developing countries including Nigeria where parasitic diseases are endemic in the population. This study is therefore aimed at assessing the parasitic contamination of commonly consumed vegetables from selected farms in Ibadan metropolis.
Study area: this study was carried out in Ibadan city, the largest indigenous city in sub-Saharan Africa. Ibadan, the capital of Oyo State is located between longitude 70 20´ and 70 40´ East of the Greenwich meridian and between latitude 30 55´ and 40 10´ North of the equator. Climatically Ibadan falls under the tropical wet and dry climates (Koppen climate classification, Aw), with a lengthy wet season, which runs from March to October, and relatively constant temperatures throughout the year, between 23 °C and 33 °C during the dry season. Ibadan has 11 local government areas (LGAs). Five LGAs were chosen for the study (Figure 1) while fourteen commercial farms were selected from the Five LGAs. These farms were considered important as the majority of fresh vegetables sold in different markets of Ibadan metropolis were brought from them.
Sample collection: a total of 280 vegetables samples comprising of ten leafy vegetable species: African eggplant (Solanum macrocarpon), lettuce (Lactuca sativa), cucumber (Brassica oleracea), spinach (Amaranthus cruentus), white jute (Corchorus olitorius), pumpkin (Telfaria occidentalis), green pepper (Capsicum sp.), okro (Abelmoschus esculentus), quill grass (Celosia argenta L) and tomato (Lycopersicum sativus), were included in the study. Socio-demographic characteristic and agronomic practice variables of 30 farmers were also obtained using semi-structured questionnaire.
Parasitological analysis of vegetables: 200-250 g samples of each vegetable were washed in distilled water in a plastic container for the removal of parasitic ova, larva or cysts. The suspension was strained through a sterile sieve to remove undesirable materials [19] and then centrifuged at 5000 rpm for 5 minutes [18]. The supernatant was discarded while the sediment obtained was transferred unto labeled clean slides for examination under the microscope using X10 and X40 objectives [19]. Modified Ziehl-Neelsen stained smears were also prepared for detection of coccidian oocysts [20]. A pictorial species identification guide from “District Laboratory Practice in Tropical Medicine” was used. Parasites were recorded as either present or not.
Data analysis: data entry and analysis were carried out using SPSS version 21.0 (SPSS Inc. Chicago, IL, USA). Frequency tables and percentages were used to display categorical data. The 95% confidence intervals were constructed around the identified levels of contamination.
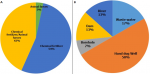
Socio-demographic characteristics of respondents: the result indicates that age composition of the farmers ranged from a minimum of 23 to a maximum of 75 years with a mean of 41.6 years and standard deviation of 12.027. There are more educated farmers than non-educated and more males (80%) than females (20%). About 90% of the respondents are married while only 10% are single. More farmers 19(63.3%) were full-time vegetable growing farmers rather than part-time 11(36.7%). (Table 1). Agronomic Practices of vegetable farmers in Ibadan showed that majority of them (54.0%) use chemical fertilizers while 3% of the farmers use animal feces as fertilizer. Only 5(17.0%) of farmers irrigate their farms with wastewater while half 15(50.0%) of them uses water from hand dug well. (Figure 2, Figure 3).
This study showed that fourteen (5.0%, 95% CI 32.6%-67.3%) samples were contaminated with intestinal parasites with white jute (32.1%, 17.9-50.6) being the highest contaminated vegetable, followed by pumpkin (7.1%, 1.9-22.6), quill grass (7.1%, 1.9-22.6) and lettuce as the least contaminated (3.5%, 0.6-17.7). No parasites were detected in African eggplant, cucumber, pepper, okra and tomato (Table 2). The distributions of the parasites were Strongyloides stercoralis larvae (42.9%, 21.3-67.4), Entamoeba histolytica (21.4%, 7.5-47.5), Trichostrongylus spp 3(21.4%, 21.3-67.4), and Ascaris spp. (14.3%, 4.0-39.9) (Figure 3). Multiple contaminations of cysts of Entamoeba histolytica, ova of Ascaris spp., Trichostrongylus spp., and Strongyloides stercoralis larvae were detected in white jute and quill grass.
The study assessed the parasitic contamination of commonly consumed vegetables from selected commercial vegetable farms in Ibadan metropolis. In addition, Socio-demographic characteristic and agronomic practices of farmers were also obtained. The age of the farmers engaged in vegetable farming in Ibadan metropolis falls within the economically active age group when they can productively carry out the rigor of farming. The distribution of the respondents by sex shows that male involvement in farming is high compared to females. This is an indication of the patriarchal head of the family where more men than women are involved in farming especially in Yoruba land in order to provide food and other essential needs of their households. While more of the farmers engaged in vegetable farming as a full-time occupation rather than part-time, their farming experience also revealed that most of the farmers had many years of farming experience. The implication is the present economic recession in the country has pushed more people to farm.
The examination of vegetables in the current study revealed a low, overall prevalence of 5% when compared with previous studies within Nigeria and outside Nigeria by Daryani et al. [21] in Ardabil city, Iran; Damen et al. [17] in Jos, Nigeria; Uga et al. [22] from Hanoi and Gharavi et al. [23] from Tehran. However, the prevalence is comparable with 3.5% in Northeastern Nigeria [24] and 6.3% in Turkey [25]. The low and the disparity in prevalence may be attributed to the diagnostic test used, environmental factors, geographical location [26, 27], as well as the differences in shape and surface of vegetables with uneven surfaces facilitating sticking of ova, cysts, and oocysts of parasite than smooth surfaces [17, 28]. The most frequent occurring parasite in sampled vegetables was Strongyloides stercoralis larvae. This is similar to the reports from Koforidua, Ghana [12] and Ibadan, Nigeria [29]. The common occurrence of Strongyloides stercoralis may be associated with poor sanitation where soil and water can be contaminated by human feces [12]. Strongyloides stercoralis have been reported as agents of diarrhoea in HIV/AIDS infected persons [30].
The detection of Ascaris spp. and Trichostrongylus sp in this study agrees with the findings of Ebrahimzadeh et al. [31] in Iran, Uneke [32] in Nigeria and Wafa [33] in Saudi Arabia who reported similar nematodes in their different studies. Contamination of vegetable by Ascaris spp. and Trichostrongylus is possibly due to animal and human fecal matter polluting the water supply used for irrigation [34]. Ascaris eggs are more resistant than other intestinal parasites due to diverse adverse environmental conditions [35], while Ascaris suum from pigs are known to be zoonotic [36]. Human trichostrongylosis cases have been reported sporadically from different countries [37, 38]. The detection of the only protozoan, Entamoeba histolytica/E.dispar in this study agrees with the findings of Benti and Gemechu [39] and Ali et al. [40] in Ethiopia and Saudi Arabia respectively. The occurrence of this protozoan cyst could be attributed to the contamination of fresh vegetables before harvest, either by irrigation with wastewater contaminated with human feces or directly from human feces [41, 42].
In our study, cucumber, pepper, and tomatoes were found to be free of parasites. This is consistent with the findings of other investigators who also reported the absence of parasites in leafy vegetables like cucumber, pepper and tomatoes [18, 43, 44]. Smooth surface which reduces the rate of parasitic attachment may suggest the non-contamination of these vegetables [17, 28]. Other parasites of public health importance like Giardia intestinalis and Cryptosporidium parvum, which were detected by previous studies [12, 28, 45] were not found in the present study. The reasons could be associated with varying ecological and climatic conditions, differences in methods of detection, and difference in endemicity of parasites from one area to the other. Despite the valuable information provided by this study, it is not devoid of limitations. Level of contamination of irrigation water, manure and soil in which green vegetables are cultivated on these farms were not assessed. Further studies are also needed to determine the risk factors associated with vegetable contamination in the study.
Our results clearly show that raw leafy vegetables grown in farms in Ibadan metropolis are quite often contaminated with parasites. These types of vegetables should be considered as a potential risk to the farming communities, handlers, transporters, and consumers of these vegetables. It is therefore recommended that information on contamination preventive practices, from farm-to fork continuum should be packaged and disseminated through appropriate channels to enhance vegetables contamination preventive behavior change among farmers.
What is known about this topic
- Vegetables had been revealed to be contaminated with enteric parasites;
- Oocysts/cysts, eggs or larvae of parasites can contaminate vegetables through poor agronomic practices.
What this study adds
- Parasites were detected in 5.0% vegetable sample;
- Detection of parasites of public health importance;
- Highest contaminated vegetable was White jute 32.1%.
The authors declare no competing interests.
OOO, and OOA, participated in the design of the work. OOO, KBA OET. OOF did data collection and laboratory work. OOO and OET performed statistical analysis and interpretation of the data. OOO and OOA wrote the first draft of the manuscript. OOO and OOF provided critical revision of the manuscript. All authors read and approved the final version of the manuscript.
The authors appreciate the cooperation of the Oyo State Agricultural and rural development (OSADEP) and the farmers who gave their consent during the course of the research work.
Table 1: socio demographic characteristics of vegetable farmers in Ibadan, Nigeria
Table 2: distribution of parasites in different vegetables from selected farms in Ibadan, Nigeria
Figure 1: map of Nigeria showing the study area
Figure 2: agronomic practices of vegetable farmers (A,B)
Figure 3: distribution of parasites identified from leafy vegetable samples in Ibadan
- Idahosa OT. Parasitic contamination of fresh vegetables sold in Jos markets. Global J Med Res. 2011;11(1):21-5.
- Hanif R, Iqbal Z, Iqbal M, Hanif S, Rasheed M. Use of vegetables as nutritional food: role in human health. J Agric Bio Sci. 2006;1(1):18-2. Google Scholar
- Onwordi CT, Ogungbade AM, Wusu AD. The proximate and mineral composition of three leafy vegetables commonly consumed in Lagos, Nigeria. African Journal of Pure and Applied Chemistry. 2009;3(6):102-107. Google Scholar
- Al-Megrm WAI. Prevalence intestinal parasites in leafy vegetables in Riyadh, Saudi Arabia. International Journal of Tropical Medicine. 2010;5(2):20-23. Google Scholar
- Kozan E, Sevimi FK, Kose M, Eserm M, Cicek H. Examination of helminth contaminated wastewaters used for agricultural purposes in Afyonkarahisar. Turkiye Parazitol Derg. 2007;31(3):197-200. PubMed | Google Scholar
- Beuchat LR. Surface decontamination of fruits and vegetables eaten raw. A review. Food Safety unit, World Health Organisation: Geneva. 1998; 98-102. Google Scholar
- Beuchat LR. Ecological factors influencing survival and growth of human pathogens on raw fruits and vegetables. Microbes Infect. 2002 Apr;4(4):413-23. PubMed | Google Scholar
- Duedu KO, Yarnie EA, Tetteh-Quarcoo PB, Attah SK, Donkor ES, Ayeh-Kumi PF. A comparative survey of the prevalence of human parasites found in fresh vegetables sold in supermarkets and open-aired markets in Accra, Ghana. BMC Res Notes. 2014;7:836. PubMed | Google Scholar
- Doyle MP, Erickson MC. Summary meeting 2007 the problems with fresh produce: an overview. J Appl Microbiol. 2008 Aug;105(2):317-30. PubMed | Google Scholar
- Johannessen, GS, Heier, BT, Rorvik, LM, Bengtsson, GB, Bredholt, S, Johannessen, S and Wasteson, Y. Potential uptake of Escherichia coli 0157:H7 from organic manure into crisphead lettuce. Appl Environ Microbiol. 2005 May;71(5):2221-5. PubMed | Google Scholar
- Bekele F, Tefera T, Biresaw G, Yohannes T. Parasitic contamination of raw vegetables and fruits collected from selected local markets in Arba Minch town, Southern Ethiopia. Infect Dis Poverty. 2017 Mar 7;6(1):19. PubMed | Google Scholar
- Kudah C, Sovoe S and Baiden F. Parasitic contamination of commonly consumed vegetables in two markets in Ghana. Ghana Med J. 2018 Jun;52(2):88-93. PubMed | Google Scholar
- Tauxe RV. Emerging foodborne diseases: an evolving public health challenge. Emerg Infect Dis. 1997;3(4): 425-434. PubMed | Google Scholar
- Sia Su G L, Mariano CMR, Matti NSA, Ramos GB. Assessing parasitic infestation of vegetables in selected markets in Metro Manila, Philippines. Asian Pac J Trop Dis. 2012;2(1):51-54. Google Scholar
- Ezatpour B, Chegeni AS, Abdollahpour F, Aazami M, Alirezaei M. Prevalence of parasitic contamination of raw vegetables in Khorramabad, Iran. Food contr. 2013; 34:92-5. Google Scholar
- Nyarango RM, Aloo PA, Kabiru EW, Nyanchongi BO. The risk of pathogenic intestinal parasite infections in Kisii Municipality, Kenya. BMC public health. 2008; 8:237. PubMed | Google Scholar
- Damen JG, Banwat EB, Egah DZ, Allanana JA. Parasitic contamination of vegetables in Jos, Nigeria. Ann Afr Med. 2007 Sep;6(3):115-8. PubMed | Google Scholar
- Adejayan A, Morenikeji O. Prevalence of Intestinal parasites in vegetables sold in major markets in Ibadan city, south-west Nigeria. Global journal of pure and applied sciences. 2015;21:7-12. Google Scholar
- Nyarango RM, Aloo PA, Kabiru EW, Nyanchongi BO. The risk of pathogenic parasitic infections in Kismi municipality, Kenya. BMC Public Health. 2003; 8.237. PubMed | Google Scholar
- Henriksen SA, Pohlenz JF. Staining of Cryptosporidia by a modified Ziehl-Neelsen technique. Acta veterinaria Scandinavica. 1981;22(3-4):594-596. PubMed | Google Scholar
- Daryani A, Ettehad GH, Sharif M, Ghorbani L, Ziaei H. Prevalence of intestinal parasites in vegetables consumed in Ardabil, Iran. Food Contr. 2008; 19:790-794. Google Scholar
- Uga S, Hoa NT, Noda S, Moji K, Cong L, Aoki Y et al. Parasite egg contamination of vegetables from a suburban market in Hanoi, Vietnam. Nepal Med Coll J. 2009 Jun;11(2):75-8. PubMed | Google Scholar
- Gharavi MJ, Jahani MR, Rokni MB. Parasitic contamination of vegetables from farms and markets in Tehran. Iranian Journal of Public Health. 2002; 13:83-86. Google Scholar
- Adamu, NB, Adamu, JB, Mohammed D. Prevalence of helminth parasites found on vegetables sold in Maiduguri, Northeastern Nigeria. Food Contr. 2012; 25 (1): 23-26. Google Scholar
- Adnnir R, Tasci F. Prevalence of helminth eggs in raw vegetables consumed in Burdur, Turkey. Food Contr. 2013; 31(2):482-484. Google Scholar
- Orlandi PA, Chudmt Bier JW, Jackson GJ. Parasites and the food supply. Fd Technol. 2002; 56:72-81. Google Scholar
- Jemaneh L. The epidemiology of schistosomiasis and soil-transmitted helminths in elementary school children from South Gonder zone of the Amhara National Regional State, Ethiopia. Ethiop Med J. 2000 Apr;38(2):105-18. PubMed | Google Scholar
- Said D. Detection of parasites in commonly consumed raw vegetables. Alexandria Journal of Medicine. 2012; 48:345-52. Google Scholar
- Ogbolu DO, Alli OA, Ogunleye VF, Olusoga OF, Olaosun I. The presence of intestinal parasites in selected vegetables from open markets in south western Nigeria. Afr J Med Med Sci. 2009 Dec;38(4):319-24. PubMed | Google Scholar
- Nissapatorn V, Sawangjaroen N. Parasitic infections in HIV infected individuals: Diagnostic and therapeutic challenges. Indian J Med Res. 2011 Dec;134(6):878-97. PubMed | Google Scholar
- Ebrahimzadeh A, Jamshidi A, Mohammadi S. The parasitic contamination of raw vegetables consumed in Zahedan, Iran. Health Scope International Journal. 2013;1(4):205-209. Google Scholar
- Uneke CJ. Potential for Geohelminth Prasaite Transmission by Raw fruits and vegetables in Nigeria: Implication for a Risk profile. Journal of Nutritional and Environmental Medicine. 2004;16(1):59-68. Google Scholar
- Wafa AI Al-Megrin. Prevalence of Intestinal Parasites in Leafy Vegetables in Riyadh, Saudi Arabia. International Journal of Zoological Research. 2010;6:190-195. Google Scholar
- Nejsum P, Betson M, Bendall RP, Thamsborg SM, Stothard JR. Assessing the zoonotic potential of Ascaris suum and Trichuris suis: looking to the future from an analysis of the past. J Helminthol 2012; 86 (2): 148-55. PubMed | Google Scholar
- Pescon BM, Barrios JA, Jiménez BE, Nelson KL. The Effects of Temperature, pH and Ammonia Concentration on the inactivation of Ascaris eggs in sewage sludge. Water Res. 2007; 41(13):2893-90. PubMed | Google Scholar
- Olsun ME, Guselle N. Are pigs´ parasites a human health risk? Adv pork production. 2000; 11: 153-162. Google Scholar
- Beaver PC, Jung RC, Cupp EW. Clinical Parasitology. Philadelphia, PA: Lea and Febiger. 1984; 289-291.
- Boreham RE, McCowan MJ, Ryan AE, Allworth AM, Robson JM. Human trichostrongyliasis in Queensland. Pathology. 1995 Apr;27(2):182-5. PubMed | Google Scholar
- Benti G, Gemechu F. Parasitic contamination on vegetables irrigated with Awash river in selected farms, eastern Showa, Ethiopia. J Parasitol Vector Biol. 2014;6(7):103-9. PubMed | Google Scholar
- Ali M Al-Binali, Cornelius SB, Khalid El-Shewy, Salah EA. The Prevalence of Parasites in Commonly Used Leafty Vegetables in South Western Saudi Arabia. Saudi Med J. 2006 May;27(5):613-6. PubMed | Google Scholar
- Olayemi AB. Microbiological Hazards Associated with Agricultural Utilization of Urban Polluted Water. International J Environ Health Res. 2007;149-154. Google Scholar
- Ward EF, Irvring LG. Virus Survival on Vegetables Spray Irrigated with waste water. Wat Res. 1987; 21:57-63. Google Scholar
- Shafa-ul-Haq Maqbool, Azhar Khan, Umbreen Javed Yasmin, Ghazalah Sultana, Razia. Parasitic contamination of vegetables eaten raw in Lahore, Pakistan. J Zool. 2014;46(5):1303-9. Google Scholar
- Mohamed MA, Siddig EE, Elaagip AH, Edris AM, Nasr AA. Parasitic contamination of fresh vegetables sold at central markets in Khartoum state, Sudan. Ann Clin Microbiol Antimicrob. 2016; 15:17. PubMed | Google Scholar
- Hassan A, Farouk H, Abdul-Ghani R. Parasitological contamination of freshly eaten vegetables collected from local markets in Alexandria, Egypt: a preliminary study. Food Contr. 2012; 26 (2):500-503. Google Scholar