Antibiotic resistance patterns in human, animal, food and environmental isolates in Ghana: a review
Pilar García-Vello, Bruno González-Zorn, Courage Kosi Setsoafia Saba
Corresponding author: Courage Kosi Setsoafia Saba, Biotechnology Department, Faculty of Agriculture, University for Development Studies, Tamale, Ghana 
Received: 31 Jan 2019 - Accepted: 10 Oct 2019 - Published: 12 Feb 2020
Domain: Microbiology,Public health
Keywords: Infections, antibiotic resistance, public health, Ghana
©Pilar García-Vello et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Pilar García-Vello et al. Antibiotic resistance patterns in human, animal, food and environmental isolates in Ghana: a review. Pan African Medical Journal. 2020;35:37. [doi: 10.11604/pamj.2020.35.37.18323]
Available online at: https://www.panafrican-med-journal.com//content/article/35/37/full
Review 
Antibiotic resistance patterns in human, animal, food and environmental isolates in Ghana: a review
Antibiotic resistance patterns in human, animal, food and environmental isolates in Ghana: a review
Pilar Garc�a-Vello1, Bruno Gonz�lez-Zorn2, Courage Kosi Setsoafia Saba3,&
1Personal Support for Projects of Molecular Biology and Biotechnology, Faculty of Pharmacy, Complutense University of Madrid, Madrid,Spain, 2Animal Health Department, Faculty of Veterinary, Complutense University of Madrid, Madrid, Spain, 3Biotechnology Department, Faculty of Agriculture, University for Development Studies, Tamale, Ghana
&Corresponding author
Courage Kosi Setsoafia Saba, Biotechnology Department, Faculty of Agriculture, University for Development Studies, Tamale, Ghana
Many articles have been published on resistant microorganisms isolated from humans, animals, foods and the environment in Ghana. However, there are no reviews that summarize the information on the isolates and antibiotics tested so far in the country. This literature review was completed through "PubMed" and "Google Scholar" searches. We included publications from the period 1975-2015 with a laboratory-based methodology to determine antibiotic resistance of strains isolated in Ghana. In total, 60 articles were included in the analysis with 10% of the articles carrying out nationwide research on antibiotic resistance. The regions of Ghana with the highest published articles were Greater Accra (40%), Ashanti (21.7%) and Northern Region (10%). Most of the studies (86.7%) were related to isolates collected from human samples followed by environmental (5%), animal (3%) and food samples (2%). Ten different bacteria genera were observed in the studies. The most common was Escherichia coli, followed by Staphylococcus spp., Mycobacterium spp. and Streptococcus spp. The highest mean resistance rate was encountered in Escherichia coli (62.2%) followed by Klebsiella spp. (60.4%) and Pseudomonas spp. (52.1%). High resistance rates have been found in Ghana, however, the data are skewed and some regions of the country have been neglected. There is a need for higher quality research to establish and monitor resistance patterns in Upper West, Brong-Ahafo, Volta and Eastern Regions of Ghana.
The increasing problem of antibiotic resistance remains a major global health problem and causes a huge medical burden [1]. There are significant international efforts, notably the World Health Organization (WHO) and Food and Agriculture Organizations (FAO), working towards tackling this crisis through planned programs such as antibiotic stewardship programs coordinated by the Global Action Plan on Antimicrobial Resistance of the WHO [2]. These programs promote optimal antimicrobial use [3-6], resistance surveillance [7], research into the molecular bases for resistance [8], strict regulations on antibiotic usage [9-11] and education of medical, public health officials and the general populace [12-14]. Some private organizations and individuals have made efforts to tackle the antibiotic crisis in some African countries. An example can be seen in Ghana through the sponsorship of research projects and educational programs [15-22]. Unfortunately, some of these projects may be duplicated and have narrow coverage area due to limited funding. Others may be executed successfully but their impacts are minimal. Furthermore, complex socio-economic and political conditions in developing and low-income countries [23] make antibiotic resistance a difficult problem to tackle [24]. This is further hindered by the inadequate and unpublishable data produced [25]. A systematic review of publications of these research works is important to evaluate the overall impacts of antibiotics on human, animal, food and environmental isolates. The objective of this study was to compile all the available information concerning antibiotic resistance in Ghana to assist clinicians and researchers to know the most commonly isolated microbes and their resistance patterns. This research aims to provide further information on the current antibiotic resistance problems, which may also help institutions to create programs and policies to enhance antimicrobial stewardship, reduce misuse of antibiotics by health professionals and unskilled practitioners, control drug quality, limit the spread of resistant bacteria, develop adequate surveillance systems and update the Standard Treatment Guidelines of hospitals in Ghana [26]. These will help to prevent or slow the emergence of resistance among microorganisms. Furthermore, this review aims to inspire researchers to conduct more investigations, particularly in neglected areas of the country.
A thorough search of literature was done in Medline (PubMed) and Google Scholar electronic databases. The search was carried out in June 2015. The following keywords were used to search in both databases with no language restriction or methodological filters: Antimicrobial OR Antibiotic AND resistance AND Ghana. Abstracts of potentially relevant titles were then reviewed manually for eligibility and were selected for closer examination to be included in the study.
�
Search on PubMed: the articles from PubMed included all publications concerning antibiotics resistance in Ghana from 1975 to July 2015.�
Search on Google Scholar: the same search words were used on Google Scholar, producing over ten thousand results. Consequently, a selection was done of every 10th page (pages 1, 10, 20, 30, 40, 50) and articles related to antibiotics in Ghana were selected based on their titles and abstracts.�
Inclusion and exclusion criteria: the inclusion criteria were: (i) abstract availability, (ii) information about antibiotic resistance of at least one bacterium, (iii) laboratory-based methodology, (iv) bacterial strains collected in Ghana. The exclusion criteria were: (i) articles already included from PubMed but found on Google Scholar (ii) articles without abstract (iii) abstracts without factual data on antibiotic resistance.�
Data analysis: data were gathered from each article concerning the bacteria isolated, the origin of isolation (human, animal, food and environment), the methods of antibiotic susceptibility test and the antibiotics used for them, the most frequent antibiotics used, the most resistant bacteria to the antibiotics used. The particular regions where the researches were conducted were also recorded. Data compilation and analysis were performed using the software MS Excel� and results were presented in percentages using tables and figures.
Current status of knowledge: the search on PubMed resulted in 288 publications of which 188 came from the search word "Antimicrobial AND resistance AND Ghana" and 100 for "Antibiotic AND resistance AND Ghana". Over ten thousand results were obtained using Google Scholar. After the screening against the inclusion criteria, only 60 articles were included in the analysis (Figure 1). Of the 60 articles analysed, 20 articles were not available in full hence only the abstracts were reviewed [27-46].
�
Sample collection�
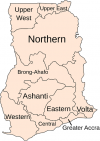
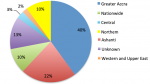
Geographical/regional distribution of articles: Ghana has ten different administrative regions (Figure 2). Research on antibiotics resistance was not conducted in the Upper West Region, Brong Ahafo Region, Volta Region and Eastern Region per this review as of the time of data collection. The majority, 23(38.3%) of the articles came from the Greater Accra Region [29,34,42,46,47-65] of which the capital city of Ghana, Accra is located (Figure 3). Six (10.0%) of the 60 articles carried out nationwide research on antibiotic resistance [43,66-70], thus these studies were designed to take samples from the whole of Ghana. Eight (13.3%) of the articles could not be traced due to inadequate information on the locations of the research [27,30,31,33,38,41,71,72].�
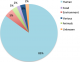
Types of samples collected: the 60 articles analysed obtained samples from different sources (Figure 4). The human isolates were taken from both healthy individuals and patients with previous or no treatment. Samples were mostly blood (8 articles) [34,41,46,54,68,73-75], and nasopharyngeal and nasal samples (6) [32,37,49,52,61,66]. There were also faecal (5) [53,59,63,71,76], sputum (3) [39,70,77], cerebrospinal fluid (3) [29,78,79], urine (3) [33,47,54], skin (1 diabetic foot ulcers and 1 Buruli ulcer) [80,65], bile (1) [30] and vaginal, cervix and urethra samples (1) [28]. Nine articles obtained samples from different sources and analysed the results together [40,51,56,57,60,69,81,82]. Ten of the articles described the kind of patient as adult or child but did not describe the origin of the samples [31,35,38,43-45,58,62,67,83]. From the three environmental articles, samples were collected from beds, floors and drainage systems of hospitals (1) [84], water samples (1) [72] and mobile phones (1) [85]. Additionally, one research group exclusively sampled tiger nuts [48]. The articles that studied antibiotic resistance in animals sampled from domesticated animals (rectal and cloacae swabs) [27] and hospital cockroaches [64]. There were two articles with combined sample sources; one collected human and animal samples; faecal samples from farmers and animals from farms [50]. Another investigated animal and food sources; samples were taken from poultry in three farms and carcasses from two open markets and a cold store [42].�
Genus and species of bacteria isolated: the bacterial species isolated and identified in the sixty articles were from ten different genera: Escherichia coli, Staphylococcus spp., Streptococcus spp., Klebsiella spp., Salmonella spp., Pseudomonas spp., Mycobacterium spp., Neisseria spp., Proteus spp. and Shigella spp. More information about the isolation of each genus of bacteria can be found in Table 1.�
Antibiotic resistance patterns: the information on the most tested antibiotics and the antibiotics recording the highest resistance rates was compiled, producing a table with forty antibiotics and 3 antibiotic combinations (Table 2). The resistance level of all the organisms isolated and their resistance patterns to forty-three antibiotic formulations and the number of bacteria tested were also included in the table. Weighted averages were calculated to obtain data that allows a comparison between bacteria. The highest resistance was found in E. coli (62.2%) followed by Klebsiella spp. (60.4%) and Pseudomonas spp. (52.1%) (Table 2).�
Antibiotic resistance pattern of E. coli: the general resistance rates from human isolates were: 100% to Amoxicillin [42,47], 86.8% to Ampicillin [40,42,50,58,59,64,71,74,80,81,84,85], 81.6% to Cotrimoxazole [40,50,59,64,73,74,80,81,85], 73.6% to Tetracycline [42,58,59,64,74,80,84], 48.7% to Chloramphenicol [42,50,58,64,71,73,80,81,84], 34.42% to Cefuroxime [50,59,74,80,81,84], 22.6% to Gentamicin [40,47,50,59,64,71,73,74,80,81,84,85], 12.74% to Amikacin [50,59,64,73,74], 6.97% to Cefotaxime [50,59,64,80] and 5.7% to Ciprofloxacin [58,59,64,71,73,74,80,84,85]. The total resistance rates in animals were: 95% to Cefuroxime, 83.4% to Tetracycline, 66% to Cefotaxime, 62% to Chloramphenicol, 60.5% to Gentamicin, 56.2% to Amikacin, 50% to Cotrimoxazole, 25.4% to Ampicillin and 20% to Ciprofloxacin [42,50,64]. In the environmental samples, antibiotic resistance rates were: 90.7%, to Ampicillin, 78.4% to Cotrimoxazole, 46.4% to Gentamicin, and 32 % to Ciprofloxacin [85]. All the E. coli isolates tested were resistant (100%) to Tetracycline in Ashanti Region [80], Cefadroxil and Cefotiam in Accra [42], Cloxacillin and Erythromycin in Northern Region [74] and Penicillin in Accra and Northern Region [42,74].�
Antibiotic resistance pattern of Staphylococcus spp.: the resistance rates in human isolates were: 94.1% to Ampicillin [49,65,74], 90.2% to Penicillin [40,49,55,65,73], 60.9% to Cefuroxime [49,65,74], 63.8% to Methicillin [55,65,73], 59.9% to Cotrimoxazole [49,65,73,74], 40.4% to Tetracycline [55,65,74], 22.8% to Ciprofloxacin [73,74] and 7.3% to Erythromycin [49,55,65,73,74]. Antibiotic resistance rates in food were: 0% to Gentamicin, 12.5% to Ampicillin, 0% to Cotrimoxazole, 82.5% to Tetracycline and 0% to Ciprofloxacin [48]. All isolates from the Northern Region were resistant to Cotrimoxazole and Ampicillin [74]. In addition, the resistance was very high against Penicillin (92.4%), Flucoxacillin (83.3%), Ampicillin (82.8%), Amoxicillin (75.0%), Methicillin (72.0%), Amoxicillin (62.5%) and Cefuroxime (62.2%) in Accra [34,47,48,49,51,55,65]; Penicillin (90.0%) and Cotrimoxazole (66.0%) in Ashanti Region [40,73]; Ciprofloxacin (66.7%) and Tetracycline (66.7%) in Northern Region [74]. There were no animal isolates.�
Antibiotic resistance pattern of Salmonella spp.: the resistance rates in human isolates were: 92.3% to Ampicillin [54,68,76], 83.2% to Chloramphenicol [54,68,75,76], 72.1% to Amoxicillin-Clavulanic acid [54,68,75,76], 76.5% to Cotrimoxazole [54,75], 32% to Cefuroxime [54,68,75], 16.9% to Gentamicin [54,68,75,76], 10.7% to Tetracycline [54,68,75,76], 0% to Ciprofloxacin [54,68,75,76]. There were no quantified resistance to Amikacin [54,76], Cefotaxime [54,76] and Nalixilic acid [76]. The resistance rates in animals and foods were also not clearly specified [42]. The highest resistance was found in Accra, where all tested bacteria were resistant to Ampicillin, Cefadroxil, Cefotiam, Erythromycin and Penicillin [42,46,54,56,57], followed by Ashanti Region where the resistance was also high against Aminopenicillin (84.5%), Cefuroxime (53.5%), Chloramphenicol (81.9%), Cotrimoxazole (76.5%) and the combination Amoxicillin-Clavulanic acid (74.3%) [75].�
Antibiotic resistance pattern of Streptococcus spp.: the resistance rates in human isolates were: 100% to Cotrimoxazole [35,49,61,75], 66.84% to Ampicillin [49,61], 12.67% to Erythromycin [32,49,61], 4.19% to Ceftriaxone [32,61,75] and 2.38% to Cefotaxime [32]. There were resistances to Penicillin [32,35,40,49,61], Tetracycline [61,75], Chloramphenicol [32,40,65] and Cefuroxime [32,49,65] but the exact percentages were not specified. There were no animal isolates. The highest resistance registered was Cotrimoxazole (100%) in Accra and Ashanti Regions [35,49,61,75]. In addition, high resistance rates found for Ampicillin (67.2%), Penicillin (48.1%), Erythromycin (20.9%) and Tetracycline (78.0%) in Accra [29,49,61]; and Gentamicin (77.8%), Ciprofloxacin (47.4%), Chloramphenicol (20.5%) and Tetracycline (75.0%) in Ashanti Region [32,35,40,75].�
Antibiotic resistance pattern of Mycobacterium spp.: the antibiotics tested in the different publications were: Isoniazid (6 articles) [36,37,39,44,70,77], Ethambutol (4) [37,44,70,77], Pyrazinamide (3) [37,44,70], Rifampicin (3) [37,44,77], Streptomycin (4) [37,44,77], Thioacetazone (1) [44], and Ciprofloxacin (1) [37]. The resistance rates in human isolates were: 20.6% to Isoniazid [36,37,39,44,70,77], 18.17% to Streptomycin [37,44,77], 4.08% to Thioacetazone [44], 3.22% to Rifampicin [37,44,77], 1.54% to Pyrazinamide [37,44,70], 0.24% to Ethambutol [37,44,70,77], and 0% to Ciprofloxacin [37]. The highest resistance was found against Streptomycin in Central Region (33%) [77]; followed by the resistance in Ashanti Region against Thioacetazone (29%), Isoniazid (26.2%) and Streptomycin (18.4%) [37,44] and in Central Region against Isoniazid (13.4%), Rifampicin (6.7%) and Ethambutol (4.1%) [77]. No animal isolates were obtained.�
Antibiotic resistance pattern of Neisseria spp.: resistance rates in human isolates were: 100% to Colistin [45], 41.86% to Sulphadiazine [45,78], 38.8% to Streptomycin [45], 13% to Septrim [45], 8.1% to Sulphafurazole [45,78], and 4.9% to Oleandomycin [45]. There was also resistance to Penicillin [28,33,43,45,78], Tetracycline [28,33,43,45,78], Ciprofloxacin [33,43,78], Chloramphenicol [45,78], and Erythromycin but were not quantified [28,45]. No animal isolates were obtained. The highest resistance rates were found in Upper East Region [78] and Northern Region [45]. In Upper East Region, all the Neisseria species tested were resistant to Sulphadiazine [78]. In the Northern Region, all the Neisseria species tested were resistant to Colistin, followed by Streptomycin (38.8%), Septrim (13.0%), Sulphafurazole (8.1%), Sulphadiazine (8.1%) and Oleandomycin (4.9%) [45]. The bacteria from other regions were susceptible to all antibiotics tested, except Ashanti Region. The data from Ashanti Region were not quantitative but instead labelled as "R" signifying resistance against Ceftriaxone, Cotrimoxazole, Erythromycin, Norfloxacin, Penicillin, Spectinomycin and Tetracycline [28].�
Antibiotic resistance pattern of Pseudomonas spp.: the most tested antibiotics were Cefotaxime (4 articles) [51,65,80,83], Cefuroxime (4) [51,65,80,83], Ceftriaxone (3) [65,80,83], Gentamicin (3) [65,80,83], Ampicillin (3) [51,65,80], Chloramphenicol (3) [51,65,80], Cotrimoxazole (3) [51,65,80], Tetracycline (3) [51,65,80], Ceftazidime (2) [80,83] and Ciprofloxacin (2) [80,83], and were also the antibiotics that recorded the highest resistance rates. Resistance rates in human isolates were: 45.5% to Cefotaxime [51,65,80,83], 52% to Ceftriaxone [65,80,83], 78.8% to Cefuroxime, [51,65,80,83], 20.57% to Gentamicin [65,80,83], 100% to Ampicillin [51,65,80], 7.8% to Ceftazidime [80,83], 84.6% to Chloramphenicol [65,80], 11.4% to Ciprofloxacin [80,83], 81.6% to Cotrimoxazole [51,65,80] and 85.38% to Tetracycline [51,65,80]. No animal isolates were obtained. The highest resistance rates were found in Accra and Ashanti Regions. In Accra, the resistance was 100% to Ampicillin and Cefuroxime [51]. In the Ashanti Region, the resistance was 100% to Ampicillin, Cotrimoxazole, Tetracycline and Chloramphenicol [80,83]. Other high resistance rates were recorded for Cefotaxime (90%), Tetracycline (81.9%), Chloramphenicol (81%) and Cotrimoxazole (77.3%) in Accra [51]; and Cefuroxime (76.5%) and Ceftriaxone (55.2%) in Ashanti Region [80,83].�
Antibiotic resistance pattern of Proteus spp.: all of the 6 articles used the disk diffusion methods. Resistance rates in human isolates were: 5.48% to Amikacin [82,83], 80.97% to Ampicillin [80,82,83], 37.9% to Cefotaxime [80,82,83], 41.4% to Ceftriaxone [80,82,83], 51.4% to Cefuroxime [80,82,83], 90.3% to Chloramphenicol [80,82,83], 35.9% to Ciprofloxacin [80,83], 84.2% to Cotrimoxazole [80,82,83], 25.7% to Gentamicin [80,82,83] and 82.4% to Tetracycline [80,82]. The bacteria isolated from food were resistant to Ampicillin, Cefotaxime, Chloramphenicol, Cotrimoxazole and Tetracycline, but susceptible to Amikacin, Ciprofloxacin and Gentamicin [48]. The highest resistance rates were found in Accra with 100% resistance to Ampicillin, Cefotaxime, Chloramphenicol, Cotrimoxazole and Tetracycline [48,51]. In the Ashanti Region, the isolates were resistant to Chloramphenicol (90.3%), Cotrimoxazole (84.2%), Tetracycline (82.4%), Ampicillin (81.6%) and Cefotaxime (37.1%) [80,82,83].�
Antibiotic resistance pattern of Klebsiella spp.: the resistance rates in human isolates were: 17.9% to Amikacin [60,73,74], 100% to Ampicillin [74,80], 20% to Cefotaxime [80], 85% to Chloramphenicol [73,80], 17.9% to Ciprofloxacin [60,73,80], 82.4% to Cotrimoxazole [73,74,80], 34.7% to Gentamicin [73,74,80] and 100% to Tetracycline [74,80]. The resistance rates in animal isolates were: 25% to Amikacin, 50% to Ampicillin, 75% to Cefotaxime, 75% to Chloramphenicol, 52% to Ciprofloxacin, 88% to Cotrimoxazole, 31% to Gentamicin and 81% to Tetracycline [64]. In food samples, isolates recorded the following resistance rates: 0% to Amikacin, 33.3% to Ampicillin, 33.3% to Cefotaxime, 33.3% to Chloramphenicol, 66.7% to Cotrimoxazole and 66.7% to Tetracycline [48]. The bacteria isolated from the environment did not have any data specifying antibiotic resistance [86]. The highest resistance rates were found in the Ashanti Region [73,80] and the Northern Region [74]. In the Ashanti Region, 100% resistance was found against the combination of Amoxicillin-Ampicillin and Ampicillin and Tetracycline [73,80]. In Northern Region, the highest resistance rates were 100% to Ampicillin, Cotrimoxazole, Erythromycin, Penicillin and Tetracycline [74]. Also, high resistance rates were found against Cotrimoxazole (86.3%), Tetracycline (79.8%), Cefotaxime (71.2%) and Chloramphenicol (71.6%) in the Greater Accra Region [48,51,60,64].�
Antibiotic resistance pattern of Shigella spp.: resistance rates in human isolates were: 37.5% to Amikacin, 95.8% to Ampicillin, 12.5% to Cefotaxime, 16.7% to Cefuroxime, 83.3% to Chloramphenicol, 0% to Ciprofloxacin, 91.7% to Cotrimoxazole, 0% to Fluoroquinolone, 37.5% to Gentamicin, 91.7% to Tetracycline, 0% to Nalixilic acid and 0% for Oxafloxacin. There was resistance to Amoxicillin-Ampicillin and Trimethoprim-Sulfamethoxazole but was not quantified [63]. The bacteria from food were susceptible to Chloramphenicol and Fosfomycin, but the resistance percentages were not specified [42]. The highest resistance rates were found in the Greater Accra Region: Ampicillin (95.8%), Cotrimoxazole (91.7%), Tetracycline (91.7%), Chloramphenicol (66.6%), Amikacin (37.5%), Gentamicin (37.5%), Cefuroxime (16.7%) and Cefotaxime (12.5%) [42].�
Antibiotic resistance is one of the biggest threats to international health nowadays; its influence goes beyond health, impacting food security and the holistic development of Ghana. Substantial evidence worldwide indicates that there has been significant increase in antibiotic resistance. Furthermore, high antibiotic resistance rates were found in different bacterial isolates in Ghana [1]. As this study observed, there were numerous gaps in the knowledge about resistance in Ghana. There is a need for investment in research and monitoring systems especially in the neglected regions of Ghana: Upper West, Brong-Ahafo, Volta and Eastern Regions, where little or no work is conducted to know the extent of antibiotic resistance menace. Additionally, there should be support for higher quality research and incentives for publication in higher impact journals, which will ensure a wider readership. Consequently, this would make researchers visible and complement global efforts to monitor resistance patterns in the country.�
This systematic review shared light on an important medical Ghanaian and global issue, however, it has some limitations. The literature search was restricted to only two databases: PubMed and Google Scholar; some journals are not included in those databases and consequently, relevant articles could have been missed through the systematic search methodology used. There is also the possibility that some investigations conducted in Ghana have not been published or yet to be published. There is evidence that a substantial number of researches remain unreported because of the limitations in time and resources as well as researches with findings that do not support the researcher's hypothesis [87]. The sample size is also a limiting factor with only sixty articles included in this analysis. Twenty of them were not fully available and only the abstracts were screened. Besides, most articles studying human isolates were in the Greater Accra Region. Despite limitations, the findings of this review provide relevant evidence for further actions to address the menace of antibiotic resistance worldwide.�
Antibiotic resistance occurs naturally as a survival adaptation of the bacteria, nevertheless, antibiotics misuse in humans and animals accelerates the process. Due to the misuse of antibiotics, governments and health authorities in Ghana and several other African countries must enforce legislation to prevent commercial and indiscriminate use of antibiotics [2]. For example, limiting the sale of antibiotics to pharmacies to ensure that the dispensation of antibiotics is performed by a pharmacist and only if prescribed by a medical doctor. There must be educational programs to sensitize professionals and patients to ensure that antibiotics are only prescribed when needed and that the patients complete the prescribed treatments [88-90]. The enforcement of these measures may look difficult and complicated because of the limiting socio-economic factors. However, in the long term, those measures are cheaper than the higher medical costs and increased mortality as a result of antibiotic resistance [91].
High resistance rates of microbial isolates from human, animal, food and environment samples have been found in Ghana, especially in E. coli (62.2%), Klebsiella spp. (60.4%) and Pseudomonas spp. (52.1%). Ampicillin, Cefadroxil, Cefotiam, Cloxacillin, Cotrimoxazole, Erythromycin, Penicillin and Trimethoprim showed very high resistance rates (Table 2). However, the data were skewed because some regions of the country have been totally neglected in terms of antibiotic resistance research. There is a need for higher quality research to establish and monitor resistance patterns in Ghana to contribute to the global efforts to curb antibiotic resistance.
What is known about this topic
- Antibiotic resistance is one of the biggest threats to international health, food security and development nowadays;
- Antibiotic resistance occurs naturally, as a survival adaptation of the bacteria, nevertheless, antibiotics misuse in humans and animals accelerates the process;
- It is crucial to improve surveillance and raise public awareness about this global crisis; enabling public knowledge will increase understanding of proper use of antibiotics and reduce misuse.
What this study adds
- Compiled available information concerning antibiotic resistance research in Ghana;
- Evidenced the significantly high antibiotic resistance rates in Ghana, especially in E. coli, Klebsiella spp. and Pseudomonas spp.;
- Identified some gaps about resistance in Ghana: neglected regions and low-quality research.
The authors declare no competing interests.
Courage Kosi Setsoafia Saba created the idea and designed the study. Pilar Garc�a-Vello performed data collection and analysis. Pilar Garc�a-Vello and Courage Kosi Setsoafia Saba wrote the manuscript. Pilar Garc�a-Vello created the accompanying tables and figures. Bruno Gonz�lez-Zorn assisted in editing and revising the manuscript.
The authors wish to thank Molly D. Pither from Keele University for proofreading this paper.
Table 1: isolation, antibiotic susceptibility test and focus of the study
Table 2: summary of resistance rates findings
Figure 1: selection of
records for the analysis
Figure 2: Ghana map
Figure 3: geographical distribution of the studies
Figure 4: type of samples included in the studies
- World Health Organization. Antimicrobial resistance. 2018. Access November 2019
- World Health Organization (WHO). Global action plan on antimicrobial resistance. 2019. Geneva: World Health Organization (WHO). Access November 2019
- Center for disease control. Drug Resistance. 2017. Access November 2019
- Cosgrove SE, Avdic E, Dzintars K, Smith J. Treatment recommendations for adult inpatients: antibiotic Guidelines 2015-2016. Baltimore: Johns Hopkins Medicine Press. 2015; 163.
- World Health Organization. Growing antibiotic resistance forces updates to recommended treatment for sexually transmitted infections. 2016. Access November 2019
- Leaper DJ, Edmiston CE. World Health Organization: global guidelines for the prevention of surgical site infection. J Hosp Infect. Feb 2017;95(2):135-136. PubMed | Google Scholar
- Aspevall O, Chowdhary T, Eremin S, Grape M, Lofmark S, Oxenford C et al. Implementation of global antimicrobial resistance surveillance system (GLASS) in Patients With Bacteriuria. World Health Organization Press. 2019. PubMed | Google Scholar
- Blair JM, Webber MA, Baylay AJ, Ogbolu DO, Piddock LJ. Molecular mechanisms of antibiotic resistance. Nat Rev Microbiol. 2015; 13(1):42-51. PubMed | Google Scholar
- United Stated(Food and Drug Administration). Food and Drug Administration (US). Silver Spring (MA): code of Federal Regulations. 20O9. Access November 2019
- Committee for medical products for veterinary (CVMP). Strategy on antimicrobials 2016-2020.Geneva: European Medicines Agency Press. 6 November 2015. PubMed | Google Scholar
- Couper MR. Strategies for the rational use of antimicrobials. Clin Infect Dis. 1997;24(Suppl 1):S154-6. PubMed | Google Scholar
- Center for Disease control and prevention. Antibiotic Awareness Week: Educational Resources. 2014. Access November 2019
- Goff DA, Van den Bergh D. A tool to improve healthcare professionals' awareness of antimicrobial resistance and antimicrobial stewardship. South Afr med journal. 2015; 105(5):420. PubMed | Google Scholar
- Carter RR, Montpetite MM, Jump RL. Mixed-methods pilot study to assess perceptions of antimicrobial stewardship in nursing homes. J Am Geriatr Soc. 2017; 65(5):1073-1078. PubMed | Google Scholar
- Ghana's national policy. National platform on antimicrobial resistance (NPAR) of Ghana. Access November 2019
- Food and Agriculture 0rganization of the United Nations (FAO). Reducing the spread of antimicrobial resistance on our farms and in our food. 2016.Access November 2019
- Antibiotic resistance worldwide and in Ghana. Antibiotic resistance worldwide and in Ghana. 2014. The ADMER Project Accra: Kenneth. Access November 2019
- Combating Antimicrobial Resistance [Internet]. Accra: Government of Ghana; c2017. Google Scholar
- Opintan JA, Newman MJ, Arhin RE, Donkor ES, Gyansa-Lutterodt M, Mills-Pappoe W. Laboratory-based nationwide surveillance of antimicrobial resistance in Ghana. Infect Drug Resist. 2015; 8: 379-389. PubMed | Google Scholar
- Project AMR. Surveillance Study in Ghana. 2016. Access November 2019
- Ghana Coalition of NGOs in Health. Ghana Coalition of NGOs in Health. 2016. Access November 2019
- Gelband H, Delahoy M. Policies to Address Antibiotic Resistance in Low- and Middle-Income Countries. National and International Action on Antimicrobial Resistance: the Center for Disease Dynamics, Economics and Policy (CDDEP) Discussion Paper. Washington DC: Center for Disease Dynamics, Economics and Policy. 2014; 26
- O'Neill J. Antimicrobial resistance: tackling a crisis for the health and wealth of Nations. London: HM Government Press. 2014;20 p.
- Okeke IN, Lamikanra A, Edelman R. Socioeconomic and behavioral factors leading to acquired bacterial resistance to antibiotics in developing countries. Emerg Infect Dis. 1999; 5(1):18-27. PubMed | Google Scholar
- Grace D. Review of evidence on antimicrobial resistance and animal agriculture in developing countries.2015. Nairobi: International Livestock Research Institute. Access November 2019
- Ministry of Health of the Republic of Ghana. Standard treatments Guidelines. Accra: Ministry of Health. Sixth Edition.2010
- Abrahams CA, Agbodaze D, Nakano T, Afari EA, Longmatey HE. Prevalence and antibiogram of Campylobacter jejuni in domestic animals in rural Ghana. Arch Environ Health. 1990; 45(1):59-62. PubMed | Google Scholar
- Addy PA. Susceptibility pattern of Neisseria gonorrhoeae isolated at the Komfo, Anokye Teaching Hospital, Ghana to commonly prescribed antimicrobial agents. East Afr Med J. 1994; 71(6):368-72. PubMed | Google Scholar
- Commey JO, Rodrigues OP, Akita FA, Newman M. Bacterial meningitis in children in southern Ghana. East Afr Med J. 1994; 71(2):113-7. PubMed | Google Scholar
- Darko R, Archampong EQ. The microflora of bile in Ghanaians. West Afr J Med. 1994; 13(2):113-5. PubMed | Google Scholar
- Dayie NT, Arhin RE, Newman MJ, Dalsgaard A, Bisgaard M, Frimodt-M�ller N et al. Multidrug-Resistant Streptococcus pneumoniae isolates from Healthy Ghanaian Preschool Children. Microb Drug Resist. 2015; 21(6):636-42. PubMed | Google Scholar
- Denno DM, Frimpong E, Gregory M, Steele RW. Nasopharyngeal carriage and susceptibility patterns of Streptococcus pneumoniae in Kumasi, Ghana. West Afr J Med. 2002; 21(3):233-6. PubMed | Google Scholar
- Duplessis C, Puplampu N, Nyarko E, Carroll J, Dela H, Mensah A et al. Gonorrhea surveillance in Ghana, Africa. Mil Med. 2015; 89(7):17-22. PubMed | Google Scholar
- Enweronu-Laryea CC, Newman MJ. Changing pattern of bacterial isolates and antimicrobial susceptibility in neonatal infections in Korle Bu Teaching Hospital, Ghana. East Afr Med J. 2007; 84(3):136-40. PubMed | Google Scholar
- Holliman RE, Liddy H, Johnson JD, Adjei O. Epidemiology of invasive pneumococcal disease in Kumasi, Ghana. Trans R Soc Trop Med Hyg. 2007; 101(4):405-13. PubMed | Google Scholar
- Homolka S, Meyer CG, Hillemann D, Owusu-Dabo E, Adjei O, Horstmann RD et al. Unequal distribution of resistance-conferring mutations among Mycobacterium tuberculosis and Mycobacterium africanum strains from Ghana. Int J Med Microbiol. 2010; 300(7):489-95. PubMed | Google Scholar
- Lawn SD, Frimpong EH, Al-Ghusein H, Acheampong JW, Uttley AH, Butcher PD et al. Pulmonary tuberculosis in Kumasi, Ghana: presentation, drug resistance, molecular epidemiology and outcome of treatment. West Afr J Med. 2001; 20(2):92-7. PubMed | Google Scholar
- Meybeck A, Calbet J, Ruimy R, Mashri K, Jachym M, Joly V et al. Multidrug-resistant tuberculous meningitis in an HIV-positive patient: a challenging disease. AIDS Patient Care STDS. 2007; 21(3):149-53. PubMed | Google Scholar
- Newman MJ, Addo KK, Aboagye S, Bonsu FA, Caulley P, Hesse IF et al. Culture and sensitivity of mycobacterial isolates from cases of pulmonary tuberculosis classified as treatment failures in a teaching hospital. West Afr J Med. 2007; 26(2):131-3. PubMed | Google Scholar
- Ohene A. Bacterial pathogens and their antimicrobial susceptibility in Kumasi, Ghana. East Afr Med J. 1997;74(7):450-5. PubMed | Google Scholar
- Petit PL, Haarlem JV, Poelman M, Haverkamp MC, Wamola IA. Bacteraemia in patients presenting with fever. East Afr Med J. 1995; 72(2):116-20. PubMed | Google Scholar
- Sackey BA, Mensah P, Collison E, Sakyi-Dawson E. Campylobacter, Salmonella, Shigella and Escherichia coliin live and dressed poultry from metropolitan Accra. Int J Food Microbiol. 2001; 71(1):21-8. PubMed | Google Scholar
- Tsai AY, Dueger E, Macalino GE, Montano SM, Tilley DH, Mbuchi M et al. The US military's Neisseria gonorrhoeae resistance surveillance initiatives in selected populations of five countries. MSMR. 2013; 20(2):25-7. PubMed | Google Scholar
- Van der Werf TS, Groothuis DG, Van Klingeren B. High initial drug resistance in pulmonary tuberculosis in Ghana. Tubercle. 1989; 70(4):249-55. PubMed | Google Scholar
- Voros S, Sackey JQ, Salles CA, Marbell EC, Grant FC, Nimo PK et al. Sensitivity test on Neisseria meningitidis strains to antimicrobial drugs. Ghana Med J. 1975; 14(3):188-92. PubMed | Google Scholar
- Wilkens J, Newman MJ, Commey JO, Seifert H. Salmonella bloodstream infection in Ghanaian children. Clin Microbiol Infect. 1997; 3(6):616-620. PubMed | Google Scholar
- Ameko E, Achio S, Alhassan S. Effects of antibiotic self-Medication on the efficacy of four antibiotics commonly used in Ghana on clinically isolated micro Organisms. Int J Pure Appl Sci Technol. 2012; 10(2): 62-70. Google Scholar
- Ayeh-Kumi PF, Tetteh-Quarcoo PB, Duedu KO, Obeng AS, Addo-Osafo K, Mortu S et al. A survey of pathogens associated with Cyperus esculentus L (tiger nuts) tubers sold in a Ghanaian city. BMC Res Notes. 2014;7:343. Google Scholar
- Donkor ES, Foster-Nyarko E, Enweronu- Laryea CC. Relationship between antibiotic resistance and sickle cell anemia: preliminary evidence from a pediatric carriage study in Ghana. Infect Drug Resist. 2013; 6:71-7. PubMed | Google Scholar
- Donkor ES, Newman MJ, Yeboah-Manu D. Epidemiological aspects of non-human antibiotic usage and resistance: implications for the control of antibiotic resistance in Ghana. Trop Med Int Health. 2012;17(4):462-8. PubMed | Google Scholar
- Edoh D, Alomatu B. Comparison of antibiotic resistance patterns between laboratories in Accra East, Ghana. AJST. 2007; 8(1):1-7. Google Scholar
- Egyir B, Guardabassi L, Esson J, Nielsen SS, Newman MJ, Addo KK et al. Insights into Nasal Carriage of Staphylococcus aureus in an Urban and a Rural Community in Ghana. PLoS One. 2014; 9(4): e96119. Google Scholar
- Labar AS, Millman JS, Ruebush E, Opintan JA, Bishar RA, Aboderin AO et al. Regional dissemination of a trimethoprim-resistance cene Cassette via a successful transposable element. PLoS one. 2013; 7(5): e38142. Google Scholar
- Labi AK, Obeng-Nkrumah N, Addison NO, Donkor ES. Salmonella blood stream infections in a tertiary care setting in Ghana. BMC Infect Dis. 2014; 14:3857. Google Scholar
- Lerbech AM, Opintan JA, Bekoe SO, Ahiabu MA, Tersb�l BP, Hansen M et al. Antibiotic Exposure in a Low-Income Country: screening Urine Samples for Presence of antibiotics and antibiotic resistance in coagulase negative staphylococcal contaminants. PLoS One. 2014;9(12): e113055. PubMed | Google Scholar
- Mills-Robertson F, Addy ME, Mensah P, Crupper SS. Molecular characterization of antibiotic resistance in clinical Salmonella Typhi isolated in Ghana. FEMS Microbiol Lett. 2002; 215:249-253. Google Scholar
- Mills-Robertson F, Crupper SS, Addy ME, Mensah P. Antibiotic resistance and genotyping of clinical group B Salmonella isolated in Accra, Ghana. J Appl Microbiol. 2003; 94(2):289-294. Google Scholar
- Namboodiri SS, Opintan JA, Lijek RS, Newman MJ, Okeke IN. Quinolone resistance in Escherichia coli from Accra, Ghana. BMC Microbiol. 2011; 11:44. Google Scholar
- Newman MJ, Seidu A. Carriage of antimicrobial resistant Escherichia coli in adult intestinal flora. WAJM. 2002; 21(1):48-50. Google Scholar
- Obeng-Nkrumah N, Twum-Danso K, Krogfelt KA, Newman MJ. High levels of extended-spectrum beta-lactamases in a major teaching hospital in Ghana: the need for regular monitoring and evaluation of antibiotic resistance. Am J Trop Med Hyg. 2013;89(5):960-4. PubMed | Google Scholar
- Odarkor Mills R, Twum-Danso K, Owusu-Agyei S, Donkor ES. Epidemiology of pneumococcal carriage in children under five years of age in Accra, Ghana. Infect Dis (Lond). 2015;47(5):326-31. Google Scholar
- Opintan JA, Newman MJ, Nsiah-Poodoh OA, Okeke IN. Vibrio cholerae O1 from Accra, Ghana carrying a class 2 integron and the SXT element. J Antimicrob Chemother. 2008; 62(5):929-933. PubMed | Google Scholar
- Opintan JA, Newman MJ. Distribution of serogroups and serotypes of multiple drug resistant Shigella isolates. Ghana Med J. 2007; 41(1): 8-29. PubMed | Google Scholar
- Tetteh-Quarcoo PB, Donkor ES, Attah SK, Duedu KO, Afutu E, Boamah I et al. Microbial carriage of cockroaches at a tertiary care hospital in Ghana. Environ Health Insights. 2013; 7:59-66. PubMed | Google Scholar
- Yeboah-Manu D, Kpeli GS, Ruf MT, Asan-Ampah K, Quenin- Fosu K, Owusu-Mireku E et al. Secondary bacterial infections of Buruli Ulcer lesions before and after chemotherapy with Streptomycin and Rifampicin. PLoS Negl Trop Dis. 2013; 7(5): e2191. PubMed | Google Scholar
- Dayie NTKD, Arhin RE, Newman MJ, Dalsgaard A, Bisgaard M, Frimodt-M�ller N et al. Penicillin resistance and serotype distribution of Streptococcus pneumoniae in Ghanaian children less than six years of age. BMC Infect Dis. 2013; 13:490. Google Scholar
- Donkor ES, Adegbola RA, Wren BW, Antonio M. Population biology of streptococcus pneumoniae in West Africa: multilocus sequence typing of serotypes that exhibit different predisposition to invasive disease and carriage. PLoS One. 2013; 8(1): e53925. PubMed | Google Scholar
- Gross U, Amuzu SK, Ciman R, Kassimova I, Gross L, Rabsch W et al. Bacteremia and antimicrobial drug resistance over time, Ghana. Emerg Infect Dis. 2011;17(10):1879-82. PubMed | Google Scholar
- Newman MJ, Frimpong E, Donkor ES, Opintan JA, Asamoah-Adu A. Resistance to antimicrobial drugs in Ghana. Infect Drug Resist. 2011; 4:215-20. PubMed | Google Scholar
- Owusu-Dabo E, Adjei O, Meyer CG, Horstmann RD, Enimil A, Kruppa TF et al. Mycobacterium tuberculosis Drug Resistance, Ghana. Emerg Infect Dis. 2006. Google Scholar
- Nys S, Okeke IN, Kariuki S, Dinant GJ, Driessen C, Stobberingh EE. Antibiotic resistance of faecal Escherichia coli from healthy volunteers from eight developing countries. J Antimicrob Chemother.2004; 54, 952-955. Google Scholar
- Dekker DM, Krumkamp R, Sarpong N, Frickmann H, Boahen KG, Frimpong M et al. Drinking water from dug wells in rural Ghana: Salmonella contamination, environmental factors, and genotypes. Int J Environ Res Public Health. 2015; 12(4):3535-46. Google Scholar
- Hamer DH, Darmstadt GL, Carlin JB, Zaidi AKM, Yeboah-Antwi K, Saha SK et al. Etiology of bacteremia in young infants in six countries. Pediatr Infect Dis J. 2015; 34(1):e1-8. PubMed | Google Scholar
- Opoku-Okrah C, Feglo P, Amidu N, Dakorah MP. Bacterial contamination of donor blood at the Tamale teaching hospital, Ghana. Afr Health Sci. 2009; 9(1):13-18. PubMed | Google Scholar
- Schwarz NG, Sarpong N, H�nger F, Marks F, Acquah SEK, Agyekum A et al. Systemic bacteraemia in children presenting with clinical pneumonia and the impact of non-typhoid salmonella (NTS). BMC Infect Dis. 2010;10:319. PubMed | Google Scholar
- Saba CKS, Escudero JA, Herrera-Le�n S, Porrero MC, Su�rez M, Dom�nguez L et al. First identification of Salmonella Urbana and Salmonella Ouakam in humans in Africa. J Infect Dev Ctries. 2013; 7(10):691-695. Google Scholar
- Yeboah-Manu D, Asante-Poku A, Bodmer T, Stucki D, Koram K, Bonsu F et al. Genotypic Diversity and Drug Susceptibility Patterns among M. tuberculosis complex isolates from South- Western Ghana. PLos One. 2011;6(7): e21906. PubMed | Google Scholar
- Gagneux S, Hodgson A, Ehrhard I, Morelli G, Genton B, Smith T et al. Microheterogeneity of serogroup A (subgroup III) Neisseria meningitidis during an outbreak in northern Ghana. Trop Med Int Health. 2000; 5(4):280-7. PubMed | Google Scholar
- Leimkugel J, Forgor AA, Gagneux S, Pfl�ger V, Flierl C, Awine E et al. An outbreak of serotype 1 Streptococcus pneumoniae meningitis in Northern Ghana with features that are characteristic of Neisseria meningitidis Meningitis Epidemics. J Infect Dis. 2005;192(2):192-9. PubMed | Google Scholar
- Brenyah RC, Ephraim RKD, Eghan BA, Asamoah J. Bacterial profile of diabetic foot ulcers of patients visiting a specialist diabetic clinic at Komfo Anokye Teaching Hospital, Kumasi, Ghana. Br J Med Med Res. 2007; 4:2231-0614. Google Scholar
- Feglo P, Adu-Sarkodie Y, Ayisi L, Jain R, Spurbeck RR, Springman AC et al. Emergence of a Novel Extended-Spectrum-β-Lactamase (ESBL)-Producing, Fluoroquinolone-Resistant Clone of Extraintestinal Pathogenic Escherichia coli in Kumasi, Ghana. J Clin Microbiol. 2013; 51(2):728. PubMed | Google Scholar
- Feglo PK, Gbedema SY, Quay SNA, Adu-Sarkodie Y, Opoku-Okrah C. Occurrence, species distribution and antibiotic resistance of Proteus isolates: a case study at the Komfo Anokye Teaching Hospital (KATH) in Ghana. Int J Pharm Sci Res. 2010; 1(9):347-352. Google Scholar
- Feglo P, Opoku S. AmpC beta-lactamase production among Pseudomonas aeruginosa and Proteus mirabilis isolates at the Komfo Anokye Teaching Hospital, Kumasi, Ghana. J Microbiol Antimicrob. 2014; 6(1):13-20. Google Scholar
- Djie-Maletz A, Reither K, Danour S, Anyidoho L, Saad E, Danikuu F et al. High rate of resistance to locally used antibiotics among enteric bacteria from children in Northern Ghana. J Antimicrob Chemother. 2008; 61(6):1315-1318. PubMed | Google Scholar
- George DF, Gbedema SY, Agyare C, Adu F, Boamah VE, Tawiah AA et al. Antibiotic Resistance Patterns of Escherichia coli Isolates from Hospitals in Kumasi, Ghana. ISRN Microbiol. 2012;2012:658470. PubMed | Google Scholar
- Tagoe DN, Gyande VK, Ansah EO. Bacterial contamination of mobile phones: when your mobile phone could transmit more than just a call. Webmed Central Microbiology. 2011; 2(10): WMC002294. Google Scholar
- Welcome.What is the public health research data forum.2014. Public Health Research Data Forum. Access November 2019
- Llor C, Bjerrum L. Antimicrobial resistance: risk associated with antibiotic overuse and initiatives to reduce the problem. Ther Adv Drug Saf. 2014 Dec; 5(6): 229-241. PubMed | Google Scholar
- Alawi MM, Darwesh BM. A stepwise introduction of a successful antimicrobial stewardship program: experience from a tertiary care university hospital in Western, Saudi Arabia. Saudi Med J. 2016 Dec; 37(12): 1350-1358. PubMed | Google Scholar
- Garcia-Vello P, Brobbey F, Gonz�lez-Zorn B, Saba CKS. A cross-sectional study on antibiotic prescription in a teaching hospital in Ghana. Pan Afr Med J. 2019 Jan. Unpublished.
- Laxminarayan R, Duse A, Wattal C, Zaidi AK, Wertheim HF. Antibiotic resistance-the need for global solutions. Lancet Infect Dis. 2013 Dec;13(12):1057-98. PubMed | Google Scholar