Molecular profile of breast cancers in Guinean oncological settings
Bangaly Traoré, Moussa Koulibaly, Aissatou Diallo, Malick Bah
Corresponding author: Bangaly Traoré, Surgical Oncology Unit, Donka National Hospital, Faculty of Medical Sciences and Technics, University Gamal Abdel Nasser of Conakry, Conakry, Guinea 
Received: 17 Jan 2019 - Accepted: 02 May 2019 - Published: 14 May 2019
Domain: Oncology
Keywords: Breast cancer, molecular subtypes, oncology
©Bangaly Traoré et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Bangaly Traoré et al. Molecular profile of breast cancers in Guinean oncological settings. Pan African Medical Journal. 2019;33:22. [doi: 10.11604/pamj.2019.33.22.18189]
Available online at: https://www.panafrican-med-journal.com//content/article/33/22/full
Molecular profile of breast cancers in Guinean oncological settings
Bangaly Traoré1,&, Moussa Koulibaly2, Aissatou Diallo3, Malick Bah1
1Surgical Oncology Unit, Donka National Hospital, Faculty of Medical Sciences and Technics, University Gamal Abdel Nasser of Conakry, Conakry, Guinea, 2Laboratory of Anatomo-Pathology, Donka National Hospital, Faculty of Medical Sciences and Technics, University Gamal Abdel Nasser of Conakry, Conakry, Guinea, 3Surgical Oncology Unit, Donka National Hospital, Faculty of Medical Sciences and Technics, University Gamal Abdel Nasser of Conakry, Conakry, Guinea
&Corresponding author
Bangaly Traoré, Surgical Oncology Unit, Donka National Hospital, Faculty of Medical Sciences and Technics, University Gamal Abdel Nasser of Conakry, Conakry, Guinea
Breast cancer is a complex disease characterized by the accumulation of multiple molecular alterations giving each tumor phenotype and an own evolutionary potential. This study aimed to describe the distribution of the profile and molecular subtypes of breast cancers followed at Surgical Oncology Unit of Donka National Hospital. This was retrospective and descriptive study on cases of breast cancer in which the hormone receptor status and expression of the HER2 oncogene have been performed from 2007 to 2016. We recorded 58 cases including 56 (96.6%) women and 2 (3.4%) men. The average age was 48.2 ± 10.9. Invasive ductal carcinoma accounted for 50 (86.2%) cases. The SBR grade was II in 31(53.4%) cases, III in 21 (36.2%) cases and I in 6 (10.3%) cases. The tumor was classified as T4 in 36 (62.1%) cases; it was metastatic in 11(19.0%) cases. Estrogen receptors were positive in 29 (50.0%) cases, progesterone receptors positive in 25 (43.1%) cases, the Her2 oncogene was overexpressed in 22 (39.3%) cases. The distribution of molecular sub-types was: 20 (34.5%) luminal A, 15 (25.9%) triple negative, 13 (22.4%) Her2 overexpressed, 8 (13.8%) luminal B and 2 (3.2%) undetermined. This preliminary study showed the poor accessibility of immunohistochemistry for the molecular diagnosis of breast cancer in our country. Luminal A subtypes and triple negatives were more common. The determination of molecular subtypes is a rational basis for hormone therapy and targeted therapy, thus personalizing the treatment of breast cancer.
Breast cancer is a major global public health problem. About 1,671,149 cases are diagnosed each year, including 521,907 deaths worldwide [1]. The number of cases reached 53,000 and more than 11,000 deaths in France [2]. In Africa, the incidence is 133,390 new cases with 63,160 deaths [1]. In North and Middle East Africa, breast cancer is the first cancer in women. It accounts for 14 to 42% of all female cancers with an exponential increase [3]. In Guinea, the age-standardized incidence is 14.5 new cases per 100,000 and the mortality rate is 7.9 per 100,000 [1]. It represents the leading cause of consultation at the Surgical Oncology Unit (SOU) of Donka National Hospital and represents 26% of all cancers [4]. In sub-Saharan countries, the patients are relatively younger and the stages are aggressive [5, 6]. Breast cancer now appears as a complex disease characterized by the accumulation of multiple molecular alterations that give each tumor a phenotype and a potential for evolution. The study of genomic alterations of tumor cells revealed their relation with the prognosis and the effectiveness of the treatments; thus requiring making a personalization in the therapeutic management. The early distinction of responder and non-responder patients is to avoid the use of ineffective chemotherapies for some patients. The progress of molecular biology techniques and gene sequencing allowed understanding the breast cancer genesis. Perou et al. [7] analyzed in 2000 the gene expression of breast cancer by micro-array and were able to highlight five molecular groups. This molecular classification distinguishes the luminal A, luminal B, Her2 positive, basal-like, and triple-negative subtypes [8]. Indeed, immunohistochemistry is used to define biological prognostic factors and especially to make a target therapy. This target therapy has been made possible through the use of monoclonal antibodies and hormone therapy. In our country, immunohistochemistry (IHC) is not available and there are very little data on the molecular profile of breast cancer [4, 9]. This study aimed to determine the first trends in the molecular profile of breast cancers followed at the surgical oncology unit of Donka National Hospital.
This was retrospective and descriptive study on cases of breast cancer in which the hormone receptor status and expression of the Her2 oncogene have been reported. These patients were followed at the Surgical Oncology Unit (SOU) of the Donka National Hospital, University Hospital Center of Conakry, from 2007 to 2016. Age, sex, menopausal status for women and existence of comorbidity were recorded. The histological diagnosis of cancer was confirmed in the two pathology laboratories at Donka National Hospital and China-Guinean Kipe Hospital. The breast cancer characteristics were studied through the histological type, Scarff Bloom Richardson (SBR) grade, initial clinical classification and the Union for International Cancer Control (UICC) tumor, node and metastasis (TNM) stage. For immunohistochemistry, paraffin blocks were sent to the Cerba laboratory (Paris) via Dakar (Senegal) in the majority of cases. The status of estrogen (ER) and progesterone (PR) receptors and human epidermal receptor (Her2) oncogene expression were used to establish the classification of molecular subtypes of breast cancer (Table 1). The index ki-67, if available in the patient's file was used. The molecular subtype was considered indeterminate if any of the three elements (ER, PR, Her2) were not reported. We reviewed the indications of hormone therapy and targeted therapy (trastuzumab) according to the hormonal status and expression of the oncogene Her. Statistical analysis was performed using SPSS software (SPSS Inc., Chicago, IL). Categorical variables were shown as the frequency and percentage (%) and continuous variables were presented as the mean and standard deviation (±SD). In this retrospective study, data were collected anonymously and confidentially. Patients signed the consent form for the use of data contained in their records.
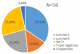
From 2007 to 2016, IHC performed in 58 patients (10.19%) out of a total of 569 breast cancers (Figure 1). The average age was 48.25 ± 10.9 years with extremes of 31 and 80 years. There were 56 women (96.6%) for 2 men (3.4%). There were 30 postmenopausal women (51.7%). Invasive ductal carcinoma was the most common histological type with 86.2%, an intra-ductal component was associated in 8 cases (13.8%). The Scarff Bloom and Richardson grades were SBR II in 31 cases (53.4%), SBR III in 21 cases (36.2%) and SBR I in 6 cases (10.3%). The tumor stage was less than or equal to T2 in 14 cases (24.1%), and greater than T2 in 44 cases (75.9%). There were axillary and/or subclavicular lymph nodes in 36 cases (62.1%). Locally advanced and metastatic stages were most common in 28 cases (48.3%) and 11 cases (19.0%). Table 2 presents the patients and breast cancer characteristics. Of the 58 cases, 29 (50.0%) had positive hormone receptors (HR+). ER were positive in 29 cases (50.0%) while PR were positive in 25 cases (43.1%). In 56 cases, the Her2 oncogene was overexpressed in 22 cases (39.3%). The Ki67 index was known for 14 cases including 4 (6.8%) less than 15% and 10 (17.2%) greater than or equal to 15%. The distribution of the molecular sub-types was 34.5% luminal A, 25.9% triple negative, 22.4% Her2 + and 13.8% luminal B. The tumors of undetermined molecular profile were 3, 4%. Chemotherapy was used in 94.8% of cases. Hormone therapy was used in 18 patients (72.0%). Only 4 (18.2%) patients with Her 2+ were treated with trastuzumab.
This study highlights the dire need for IHC in sub-Saharan Africa. In this retrospective study, IHC was performed for 10.2% of breast cancers diagnosed from 2007 to 2016. Access to IHC remains limited, as it accounts for less than 30% of breast cancers in West African countries [10, 11]. It is often during study projects, whether monocentric [12] or multicenter [13], that the molecular profile is better performed in different sub-Saharan Africa countries. On the other hand, there is better access in the Maghreb countries [14]. IHC is not available in our country as in most countries of sub-Saharan Africa. For the cases in this study, the histological diagnosis is made in Conakry, the paraffin blocks are sent to other countries or when the patients stay there for treatment. Despite the small number of cases, this study has established the first trend of hormone receptor expression, Her2 oncogene and molecular sub-types of breast cancer in Guinea. Breast cancer is a complex and heterogeneous disease associated with clinicopathological and biological behaviors that vary from one population to another [15]. These prognostic factors are essential for the management of breast cancer. In addition, the molecular classification is now an important tool to guide breast cancer management. In this study, the average age was 48.2 years. This age is similar to that found in a previous study of all breast cancers at the SOU of Donka [5]. The relatively younger age of women with breast cancer is already reported in several studies in Africa [12, 16] unlike developed countries where the age is more advanced, beyond 65 [17]. Gakwaya et al. [18] in Uganda reported in their study a high frequency in the 30-39 age groups. Age at diagnosis is an important prognostic factor since tumors diagnosed at younger ages are generally more aggressive and/or less response to treatment. This young age is related to the age of the female population in Guinea, whose under-40s represent 83.1% [19]. The anatomo-clinical characteristics of breast cancer in this study are the same as those of breast cancer reported in a previous study [5]. They are characterized by tumors larger than T2 (75.9%), stage III and IV (70.9%), high grade (SBR II and III) (89.6%), and metastatic (19.0%).
Intensive Care Consortium (ICC) was the common histological type in more than 80% as in several other studies in Africa and elsewhere [14, 13, 20]. However, many efforts still have to be made by Guinean pathologists as to the description of histological types. In western countries, the majority of breast tumors are less than 2cm, reflecting early detection of the disease [21]. We did not take into account the tumor size in this study but 75.9% were classified T3-T4. These same proportions are found in multicentric studies in sub-Saharan Africa [22]. The advanced stage of diagnosis in our patients could be explained by the young age, the delay of consultation, the absence of mass screening in the population, the bad orientation and an insufficient reference system of the cancer patients. HRs were positive in half of the patients in our study. The positivity of HR in breast cancer remains very varied and heterogeneous in countries. The results are similar in South Africa [23] and Ghana [11]. HR positivity is very high in the Maghreb and the Western countries, possibly exceeding 70% [24]. These differences would be related to the young age of our patients even though patients with HR positive were relatively older than patients with HR negative. HR positivity has been shown to be greater in older patients than in the elderly [25]. HR is different depending on the types of receptors. In our study, ER were positive in 50.0% while PR were positive in 43.1%. This receptor positivity appeared to be higher than in Mali where Ly et al. [26] showed that ER and PR were positive in 39% and 29% respectively. In a multicentric study including 507 patients from Senegal and Nigeria, Huo et al. [27] demonstrated a low HR positivity rate, in the order of 24 and 20% for ER and PR. The HR status is the best indicator for hormone therapy. The small size of the samples published in different African countries makes it difficult to determine the factors that influence the expression of hormone receptors. This study justified tamoxifen hormone therapy in 72.0% of patients in our study who were RH positive.
Oncogene HER2 was hyper-expressed in 39.3% of patients in our study. This expression is lower than that of hormone receptors. The Her2 oncogene is less overexpressed, with a proportion of around 17% in Senegal and Nigeria [28]. Although being a target therapy, the overexpression of Her2 is associated with poor prognostic factors correlated to the stage of breast cancer diagnosis. Data showed that expression of the Her2 increased according to stage, 22% in the early stage, 35% in the locally advanced stage and 40% in inflammatory breast cancers [28, 29]. The distribution of molecular subtypes found a high frequency of luminal A subtypes (34.5%), followed by triple negatives (25.9%), Her2 + types (22.4%) and luminal B (13.8%). In sub-Saharan Africa, luminal A is more common in some countries [12, 30], while in others it is the triple negative that is more common [26, 27]. Apart from locoregional (by surgery and radiotherapy) and systemic (by chemotherapy) control, triple negative breast cancers are very difficult to manage in case of resistance to chemotherapy due to the lack of therapeutic target. The numbers are small, which could be used to highlight the lack of services in Guinea. The number of patients treated with hormone therapy (72.0%) and trastuzumab (18.2%) are small, which could be used to highlight the lack of services in Guinea.
This preliminary study showed the poor accessibility of immunohistochemistry for the molecular diagnosis of breast cancer in our country. Luminal A subtypes and triple negatives were more common. The determination of molecular subtypes is a rational basis for hormone therapy and targeted therapy, thus personalizing the breast cancer treatment.
What is known about this topic
- Breast cancer in sub-Saharan African women is aggressive: triple negatives and high grades.
What this study adds
- We noted a difference in the distribution of molecular subtypes: luminal A more frequent than triple negative, but still high grade.
The authors declare no competing interests.
All authors contributed to the design and writing of this study. All authors also state that they have read and approved the final version of the manuscript.
We thank Mr Balde Thierno and all the team of Surgical Oncology Unit for the good preservation of patient records.
Table 1: classification of molecular subtypes
Table 2: patients and breast cancer characteristics
Figure 1: distribution of molecular subtypes
- Globocan. Estimated cancer incidence, mortality and prevalence worldwide. 2012. Consulté le 25 Décembre 2016.
- Espié M. The management of breast cancer. Diagnostic and Interventional Imaging. July-August 2014; 95(7-8): 753-757. Epub 2014 May 22. PubMed | Google Scholar
- Belkacémi Y, Boussen H, Hamdi-Cherif M et al. Epidemiology of breast cancer in young women in North Africa. 32nd Meeting of the French Society of Senology and Breast Pathology. Journées, Strasbourg, FRA. Novembre 2010; 56-68.
- Traore B, Barry Sonna M, Dassy E et al. Over Review of Three Years (2007-2009) Activities of the Unit of Surgical Oncology, National Hospital of Donka. Guinée Médicale. 2011; 72: 11-18.
- Traoré B, Keita M, Diane S, Dankoro A, Kabba IS, Keita N. Clinicopathological study of breast diseases presenting to the surgical oncology unit of Donka University Hospital in Conakry, Guinea. West Afr J Med. 2012; 31(4): 227-31. PubMed | Google Scholar
- Elima JA, Adébamowo C, Isabel DSS. Stage at diagnosis of breast cancer in sub-saharan Africa: a systematic review and méta analysis. Lancet Glob Health. 2016; 4(12): e923-e935. PubMed | Google Scholar
- Perou CM, Sřrlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. Molecular portraits of human breast tumors. Nature. 2000 Aug 17; 406(6797): 747-52. PubMed | Google Scholar
- Boisserie-Lacroixa M, Bullier B, Hurtevent-Labrota G, Ferron S, Lippa N, Mac Grogan G. Corrélation between imaging and prognostic factors: molecular classification of breast cancers. Journal of Diagnostic and Interventional Imaging. 2014; 95(2): 227-233. PubMed | Google Scholar
- Traore B, Toure A, Sy T, Mamadou Moustapha Dieng, Mamoudou Condé, Ahmadou Deme et al. Prognosis of Breast Cancer Patients Underwent Surgery in a Developing Country. Journal of Cancer Therapy. September 2015; 6(9): 803-810. Google Scholar
- Nfawor CC, Keshinro SO. Pattern of hormone receptors and human epidermal growth factor receptor 2 status in sub-saharan breast cancer cases: Private practice experience. Niger J Clin Pract. 2015; 18(4): 553-8. PubMed | Google Scholar
- Ohene-Yeboah M, Adjei E. Breast cancer in Kumasi. Ghana Ghana Med J. 2012; 46(1): 8-13. PubMed | Google Scholar
- Ahoua BE, Nguiessan AA, Baumaney SK et al. Immunohistochemical determination of estrogen and progesterone receptors in breast cancer: relationship with clinicopathologic factors in 302 patients in Ivory Coast. BMC Cancer. 2017; 17(1): 115. PubMed | Google Scholar
- Venderpuye V, Grover S, Hammad N, PoojaPrabhakar, Simonds H, Olopade F et al. An update on the management of breast cancer in Africa. Infect Agent Cancer. 2017; 12: 13. PubMed | Google Scholar
- Cherbal F, Gaceb H, Mehemmai C, Saiah I, Bakour R, Rouis AO et al. Distribution of molecular breast cancer subtypes among Algerian women and correlation with clinical and tumor characteristic: a population-based study. Breast Dis. 2015; 35(2): 95-102. PubMed | Google Scholar
- Blows FM, Driver KE, Schmidt MK, Broeks A, van Leeuwen FE, Wesseling J et al. Subtyping of breast cancer by immunohistochemistry to investigate a relationship between subtype and short and long term survival: a collaborative analysis of data for 10159 cases from 12 studies. Plos Med. 2010; 7(5):e1000279. PubMed | Google Scholar
- Fregene A, Newman LA. Breast cancer in sub-saharan Africa: how does it relate to breast cancer in African-American women? Cancer. 2005; 103(8): 1540-50. PubMed | Google Scholar
- The Northern Ireland Cancer Registry. Northern Ireland annual cancer incidence and survival statistics. May 2016. Consulté le 10 Oct 20.
- Gakwaya A, Kigula-Mugambe JB, Kavuma A, Luwaga A, Fualal J, Jombwe J et al. Cancer of the breast: 5years survival in a tertiary hospital in Uganda. Br J Cancer. 2008; 99(1):63-67. PubMed | Google Scholar
- Guinée. Enquęte démographique et de santé et ŕ indicateurs multiples (EDS-MICS). Santé reproductive dans l'EDS-MICS. 2012. consulted on December 30th, 2017.
- Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006; 295(21): 2492-502. PubMed | Google Scholar
- Blamey RW, Hornmark-Stenstam B, Ball G, Blichert-Toft M, Cataliotti L, Fourquet A et al. ONCOPOOL-European database for 16 944 cases of breast cancer. Eur J Cancer. 2010; 46(1): 56-71. PubMed | Google Scholar
- Jedy-Agba E, McCormack V, Olaomi O, Badejo W, Yilkudi M, Yawe T et al. Determinants of stage at diagnosis of breast cancer in Nigerian women: Sociodemographic, Breast cancer Awareness, Health Care access and clinical factors. Cancer Causes Control. 2017 Jul; 28(7): 685-697. Epub 2017 Apr 26. PubMed | Google Scholar
- McCormack VA, Joffe M, van den Berg E, Broeze N, Silva Idos S, Romieu I et al. Breast cancer receptor status and stage at diagnosis in over 1200 consecutive public hospital patients in Soweto, South Africa: a case series. Breast Cancer Res. 2013; 15(5): R84. PubMed | Google Scholar
- Lumachi F, Brunello A, Maruzzo M, Basso U, Basso SM. Treatment of estrogen receptor-positive breast cancer. Curr Med Chem. 2013; 20(5): 596-604. PubMed | Google Scholar
- Lisa KD, Mary AR, Christopher IL. Hormone receptor status, tumor characteristics and prognosis: a prospective cohort of breast cancer patients. Breast Cancer Res. 2007; 9(1): R6. PubMed | Google Scholar
- Ly M, Antoine M, Dembélé AK, Levy P, Rodenas A, Touré BA et al. High incidence of triple-negative tumors in sub-Saharan Africa: a prospective study of breast cancer characteristics and risk factors in Malian women seen in a Bamako university hospital. Oncology. 2012; 83(5): 257-63. Epub 2012 Sep 4. PubMed | Google Scholar
- Huo D, Ikpatt F, Khramtsov A, Dangou JM, Nanda R, Dignam J et al. Population differences in breast cancer: survey in indigenous African women reveals over-representation of triple-negative breast cancer. J Clin Oncol. 2009; 27(27): 4515-4521. Epub 2009 Aug 24. PubMed
- Seshie B, Adu-Aryee NA, Dedey F, Calys-Tagoe B, Clegg-Lamptey JN. A retrospective analysis of breast cancer subtype based on ER / PR and HER2 status in Ghanaian patients at the Korle Bu Teaching Hospital, Ghana. BMC Clin Pathol. 2015; 15: 14. PubMed | Google Scholar
- Zell JA, Tsang WY, Taylor TH, Mehta RS, Anton-Culver H. Prognostic impact of human epidermal growth factor-like receptor 2 and hormone receptor status in inflammatory breast cancer (IBC): analysis of 2014 IBC patient cases from the California cancer registry. Breast Cancer Res. 2009; 11(1): R9. Epub 2009 Feb 19. PubMed | Google Scholar
- Adebamowo CA, Famooto A, Ogundiran TO, Aniagwu T, Nkwodimmah C, Akang EE. Immunohistochemical and molecular subtypes of breast cancer in Nigeria. Breast Cancer Res Treat. 2008; 110(1): 183-88. Epub 2007 Aug 3. PubMed | Google Scholar