The prevalence and determinants of HIV seroconversion among booked ante natal clients in the University of Uyo teaching hospital, Uyo Akwa Ibom State, Nigeria
Nsikak Paul Nyoyoko, Augustine Vincent Umoh
Corresponding author: Nsikak Paul Nyoyoko, Department of Obstetrics and Gynaecology, University of Uyo Teaching Hospital, Uyo, Nigeria 
Received: 01 Apr 2015 - Accepted: 05 Dec 2016 - Published: 21 Dec 2016
Domain: Maternal and child health
Keywords: HIV, seroconversion, determinants, ante natal care, HCT, PMTCT, prevalence, window period, Uyo, Nigeria
©Nsikak Paul Nyoyoko et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Nsikak Paul Nyoyoko et al. The prevalence and determinants of HIV seroconversion among booked ante natal clients in the University of Uyo teaching hospital, Uyo Akwa Ibom State, Nigeria. Pan African Medical Journal. 2016;25:247. [doi: 10.11604/pamj.2016.25.247.6715]
Available online at: https://www.panafrican-med-journal.com//content/article/25/247/full
Original article 
The prevalence and determinants of HIV seroconversion among booked ante natal clients in the University of Uyo teaching hospital, Uyo Akwa Ibom State, Nigeria
The prevalence and determinants of HIV seroconversion among booked ante natal clients in the University of Uyo teaching hospital, Uyo Akwa Ibom State, Nigeria
Nsikak Paul Nyoyoko1,&, Augustine Vincent Umoh1
1Department of Obstetrics and Gynaecology, University of Uyo Teaching Hospital, Uyo, Nigeria
&Corresponding author
Nsikak Paul Nyoyoko, Department of Obstetrics and Gynaecology, University of Uyo Teaching Hospital, Uyo, Nigeria
Introduction: the Prevention of Mother to Child transmission (PMTCT) of HIV, an important intervention in the fight against HIV/AIDS can only be of benefit if the HIV status of the mother is known. Unfortunately, some women receive HIV counseling and testing (HCT) during the window period or prior to new infection in pregnancy and therefore, miss-out on the gains of PMTCT. A repeat HIV test would identify this later seroconversion and ensure early intervention. This study aimed at determining the prevalence of HIV seroconversion during pregnancy and assessing the factors associated with HIV seroconversion in clients who attended ante-natal clinic (ANC) in University of Uyo Teaching Hospital, Uyo.
Methods: a descriptive cross-sectional design was used to study 502 HIV negative clients who consecutively presented for ANC in the University of Uyo Teaching Hospital Uyo over a 3 months period (July -September 2013).
Results: fifteen (3%) of the 502 women who were HIV negative at booking visit tested positive (seroconverted) to HIV. Determinants of seroconversion at multivariate level were age bracket of 25-29 years (OR: 3.20; 95%CI: 2.39-4.29) clients� partners who had sexually transmitted infections, as evidenced by penile discharge (OR: 0.09; 95%CI: 0.01-0.45); Clients in polygamous setting (OR: 3.98; CI: 1.64-9.65); those taking alcohol (OR: 0.12; CI: 0.20-0.80) and those with partners taking recreational drugs (OR: 0.05, 95%CI: 0.002-0.995).
Conclusion: the study revealed a high seroconversion rate. Re-screening of pregnant women after the booking HCT is a very cost effective and beneficial strategy for the elimination of mother to child transmission of HIV.
The human immunodeficiency virus (HIV) pandemic is one of the most serious public health crises the world faces today. Acquired immunodeficiency syndrome (AIDS) has killed more than 25 million people since it was first discovered in 1981 [1]. In 2008, approximately 33.4 million people worldwide (1% of the global adult population aged 15-49 years) were infected with HIV with 67% of all people living with HIV worldwide residing in sub-Saharan Africa and 91% of all new infections among children occur there [2]. HIV/AIDS remains a threat to health in Nigeria and continues to strain the struggling health system as it has reversed many developmental gains of the recent past including maternal and under-five mortality rates. With an estimated population of 162,265,000 as at mid-2011 and an annual growth rate estimated to be 2.6% in 2012 [3], Nigeria, the most populated country in sub-Saharan Africa has the second largest number of people living with HIV in the world [4] with a national HIV prevalence of 4.1% and 3.4% in 2010 and 2012 respectively [5]. Akwa Ibom state HIV prevalence in both surveys were 10.9% and 6.5% respectively with a corresponding national position of second and sixth respectively [5]. It is pertinent to note however, that study of 2010 was a hospital based sentinel study while the 2012 study was a population based study. The prevention of mother-to-child transmission (PMTCT) of HIV focuses mainly on women who are HIV-positive at their first antenatal visit with uncertainty about the contribution of mothers who undergo sero-conversion after their first antenatal visit and during breastfeeding to the overall vertical transmission rate. Acute maternal HIV-1 infection during pregnancy and breast-feeding is associated with a very high rate of mother-to-child transmission (MTCT) of HIV-1. This rate can be over 50% in breast-feeding populations; approximately one third of infants whose mothers seroconvert following delivery will become infected through breast-feeding alone [6, 7]. Such a high transmission rate is understandable because maternal viral load is the most consistent predictor of MTCT of HIV infection [8], and acute HIV infection is associated with very high levels of HIV-1 RNA [9]. Most of this sero-conversion is unrecognized and therefore untreated. Despite recommendations from the (Nigerian) National Guidelines on PMTCT [10], repeat HIV testing during pregnancy is performed infrequently for women who have negative ante partum HIV test results. Women who sero-convert to HIV-positive status during pregnancy do not therefore receive prophylaxis because they are erroneously believed to be HIV-negative. Especially at risk are women who book early in pregnancy, increasing the interval between testing at booking visit and delivery. These women would also not receive counseling on the best method of infant feeding, thus increasing the risk of transmission to their infants. The risk of transmission of HIV to health workers attending to these women is also increased as they may still be treated as HIV negative. Repeat HIV testing during late pregnancy or labour could identify these cases and thus is very necessary. Defining co-factors for HIV infection during pregnancy and the postpartum period is a useful first step towards development of strategies to decrease HIV acquisition to both mothers and infants during this period [11]. Guidelines in resource-limited settings like ours recommend HIV testing as early as possible during pregnancy and then repeat testing toward the end of pregnancy (at about 36 weeks' gestation) or during labour; a strategy that may be cost-effective [12]. In their analysis of the cost-effectiveness of repeat HIV testing in pregnancy, Samson and colleagues [13] recommended that HIV testing be repeated during the third trimester in settings where HIV-1 incidence is 1.2 per 1000 person-years or higher. At that incidence, the costs of a second test are offset by averted medical costs. This study sought to determine the prevalence of HIV sero-conversion among women attending ANC in University of Uyo Teaching Hospital, Uyo and establish the factors associated with the sero-conversion. It is envisaged that the findings of this study would help capture mothers newly infected with HIV and recommend them for HIV care during pregnancy as well as decrease MTCT of HIV during pregnancy.
Study location: the Study was conducted in the maternity complex of the University of Uyo Teaching Hospital, Uyo (UUTH). This Hospital is the only tertiary healthcare facility in Akwa Ibom State, South-South geopolitical zone of Nigeria providing specialized health care services to the over 4 million people of the state and its environs including PMTCT intervention to HIV positive pregnant women. In the year 2011, there were 3,736 antenatal clients and 2,349 deliveries in the facility, with 604 deliveries in the 3 months of the study � July to September 2011.
�
Study population and technique: HIV sero-negative clients who booked for ANC at a gestational age earlier than 26 weeks gestation were consecutively recruited over a 12 weeks period until the sample size was obtained.�
Data collection and research instrument: a semi-structured interviewer-administered questionnaire was used to obtain the bio-data of clients and that of their partners/spouses; as well as the risk factors for HIV transmission. HIV Re-testing was done for the selected clients in labour, before induction of labour or prior to an elective caesarean section; according to the national HIV Rapid Testing Serial Algorithm.�
Data analysis: data obtained was analyzed using the statistical package for Social Sciences (SPSS) version 17. The level of significance was set at p-value less than 0.05.�
Ethical consideration: ethical approval was obtained from the ethical committee of UUTH, Uyo.
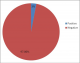
The total number of booked clients at delivery for the 3 months when the study was conducted was 604. Thirty six (36) of the 604 (6%) were known HIV positive clients, and were exempted from the study. Five hundred and two HIV negative clients were recruited into the study. Of these, 15 (3%) tested positive (seroconverted) when re-tested for HIV infection in labour as shown in Figure 1. The Majority of respondents were in the age bracket 25-29 years with a mean age of 29.46 + 3.99 years. Among those who seroconverted, majority (n=6; 40%) were within the 25-29 year age bracket. Most (n=440; 87.6%) respondents resided in urban areas. Four hundred and ninety two (98%) clients were married. Every client (100%) had a form of formal education with a higher proportion (n = 372; 74.1%) having tertiary (post-secondary) education. Almost an equal proportion were professionals (n=189; 37.6%) and unemployed (n=187; 37.3%). Majority (n=299; 59.6%) were Ibibios. Primigravid clients accounted for 43.4 % (n=218) of women recruited while only 2.8 % (n=13) were grandmultiparous. The knowledge of respondents concerning HIV transmission is displayed in Table 1. Two (0.4%) respondents were unaware of sexual transmission of HIV, 3 (0.6%) did not know it could be transmitted via blood transfusion and 25 (5%) were unaware of mother to child transmission of HIV infection. Risk factors for HIV sero-conversion analyzed is shown on Table 2. 16 (3.2%) clients had blood transfusion in index pregnancy. Thirteen (2.6%) had unprotected sexual intercourse with people other than their partners. Of the 153 (30.5%) clients who take alcohol, 26 (17%) had been drunk. Only 3 (0.6%) women smoke and none use recreational drugs. Relating sero-conversion to risk factors (as shown in Table 3) found in clients who became HIV positive at re-testing ,there was no statistically significant relationship between HIV status at delivery and lack of condom use in pregnancy (p =0.255),smoking habit(p =0.760) and partner�s alcohol intake (p= 0.423). However, unprotected intercourse with persons other than their partners (p =0.008), having partners with multiple sexual partners (0.001), alcohol intake (p = 0.002), and those who lived separate from their husbands (p = 0.001) were significantly at risk of HIV sero-conversion in pregnancy. Other statistically significant association includes partner with penile discharge, knowledge of partner�s HIV status, partner�s smoking habit, recreational drug use and, number of wives (p = 0.001) (Table 4). At multivariate analyses however, few factors found to be statistically significant in their association with HIV seroconversion were: clients of age group 25-29 years (OR: 3.20; 95%CI: 2.39-4.29; p < 0.001), client�s alcohol intake (p=0.03; OR=0.108), number of partner�s wives (p=0.02; OR=0.02), partner use of recreational drugs (p=0.05; OR =0.05) and penile discharge in partner (p=0.002; OR=0.036) (Table 5).
The recommendation of a double HIV antibody test algorithm [12, 14] (one in early pregnancy and a repeat in late pregnancy) to reduce missed opportunities for diagnosis and PMTCT interventions, may not be out of place for a country like Nigeria. This study was designed with this in view. This study documents a repeat HIV sero-prevalence of 3.0% among the booked antenatal attendees who were previously HIV sero-negative earlier in pregnancy. This figure conforms to the CDC's cut off of > 1.2% for high risk populations [14].This prevalence was higher than that in Jos University Teaching Hospital, Jos (JUTH) with a seroconversion rate of 2.1% (5 out of 235 women) [15]. The sample size of the Jos study, being smaller, may have contributed to this difference especially considering the fact that there are at least 3 referral health centres located in Jos metropolis catering for women in and around Jos. This is unlike in Uyo where UUTH is the only tertiary referral Centre. Also, Uyo (Akwa Ibom state) had a higher HIV prevalence than Jos (Plateau State) in the National HIV sentinel survey report 2012 [5] and expectedly, should have a higher HIV seroconversion rate in pregnancy. A recent study [16] closer to Uyo, in Nnewi �south east Nigeria revealed a repeat HIV sero-prevalence of 3.91% which is close to the result obtained here. It is important to note the sociocultural similarities between these 2 regions and the fact that both studies were carried out in tertiary health facilities may be responsible for the similar sero-conversion rate. In Eastern and Southern African countries, the seroconversion rates were varied with retest prevalence of 2.9% in Botswana [17], 2.6% in Kenya [11], 4.4% in Swaziland [18] and 3.0% in South Africa [19]. These studies produced results similar to this study, though they were multi-centered and carried out in both urban and rural areas. Also, the women were followed-up and re-tested at various times postpartum, 12 to 60 weeks after initial test. The length of follow-up affects the cumulative prevalence rate because the window period varies in individuals. For instance, a Zimbabwean study [20] where repeat tests were conducted at delivery, six weeks postpartum and at three monthly intervals until 24 months or on termination due to subsequent pregnancy, death or loss to follow up; gave a seroconversion rate as high as 17.7% (4.8 per 100 person years; 95% confidence interval [CI], 3.1 to 6.5).
The high HIV prevalence in some of the study areas affected the seroconversion rate. For instance, women in Western Kenya [21], where the HIV prevalence rate of 15% is over twice the national rate, were at especially high risk for seroconversion during pregnancy. In settings with high HIV incidence, new maternal infections may actually be responsible for a substantial proportion of HIV seroconversion and MTCT of HIV, particularly when MTCT prevention programs are effectively reducing transmission among mothers with known HIV infection. These women represent the missed opportunities for PMTCT of HIV. For example, it has been estimated that MTCT secondary to seroconversion during pregnancy could account for more than 40% of all ongoing MTCT in Botswana [17]. Investigators who conducted these studies above, acknowledge that some of the women who first tested positive late in pregnancy or in the early postpartum period may have had an initial false-negative result (or may have been in the "window period" for seroconversion). Nevertheless, a second true-positive test represents an important (albeit late) opportunity to intervene to prevent MTCT of HIV infection. In this study, the risk factors to HIV transmission common to the study area were analyzed. As already known, pregnancy in itself is a risk factor especially when conceived through heterosexual intercourse.
In the 2010 HIV survey in Nigeria [5], the infection was more prevalent among the 20-39 year age group and more specifically, mostly found in the 30-34 year age group. In the Uyo and a Tanzanian study [22], the 25-29 year age group was most affected. The Nnewi study [16] also showed a modal age group of 25-29 years. However, in Harare, Zimbabwe [20] women aged 17 years and below had the highest seroconversion incidence (6.25%) followed by those aged 18 to 19 years (5.42%). This suggests that most infections in women occur at a younger age, during the first few years after sexual debut. Biological factors such as immature genital tract and cervical ectopy which is common in young women might increase the risk. In 2010, HIV prevalence was highest in urban areas of Nigeria [5]. This study in UUTH conforms to the national findings, but this could simply be attributed to the fact that the study area, UUTH Uyo, is urban. In a study in KwaZulu-Natal, South Africa [19], among the 2377 HIV-negative women retested, 1099 (46.2%) and 1278 (53.4%) were tested at urban and rural health facilities, respectively; HIV incidence in pregnancy was higher but not statistically significant at the urban facilities (12.4/100 woman years versus 9.1/100 woman years). Univariate analysis showed an association between HIV seroconversion and unprotected sexual intercourse with multiple sexual partners (OR: 0.51, 95%CI: 0.3-0.86; p=0.012). This was similar to a study in northern Nigeria [23] (AOR 2.4, 95% CI= 0.97- 6.2) and Tanzania [22] (p < 0.001). However using multivariate analysis, this association was no more significant. Same pattern was noted when association was sought for client living with partner. In a South African study [24], lack of co-habitation was one of the five major factors associated with HIV seroconversion in pregnancy in over 80% of cases. Lack of co-habitation increases the risk of having multiple sexual partners, thus creating an avenue for HIV transmission. Smoking and vulval/vaginal ulcers were predictive of HIV seroconversion by univariate analysis, but not so with multivariate. This agrees with a study in Jos [23], Nigeria stating that HIV infection was not found to be associated with smoking in women and history of sexually transmitted infection.
�
In this study, there was a strong association between HIV seroconversion with clients who drink alcohol but none for partners who drink alcohol. This is contrary to the findings in Jos [23] where no association was found between HIV seroconversion and alcohol use in patients and their partner. In Uganda [25] however, individuals who had at any point, consumed alcohol experienced HIV prevalence twice that of those who had never done so: 10% vs 5% (OR 2.0, 95% CI: 1.5-2.8). Alcohol abuse has been reported to be associated with having a greater number of sex partners, history of condom failures, and lifetime history of sexually transmitted infections [26]. Use or non-use of condoms was not found to be associated with HIV seroconversion in this study. Contrary to this finding, in Harare [20], women who did not seroconvert during the time of pregnancy or follow up, were significantly more likely to have used a condom with their partners (OR = 0.68, 95% CI = 0.47 to 0.99). In a cochrane review [27] on condom use, 14 studies were included in the final analysis. The incidence of HIV infection among those who reported always using condoms was 1.14 per 100 person-years (95% CI= 0.56-2.04), while it was 5.75 per 100 person-years (95% CI= 3.16-9.66) among those who never used them. This gave an 80% reduction in the incidence of infection with condom use. It is however important to note that the meta-analysis was done using data from observational studies and the authors did not provide confidence intervals for their estimated effect of 80%.Observational studies inherently carry a risk of bias as Self-reported data always have the risk of being unreliable. Possible reasons for the absence of an association between HIV seroconversion with condom use in marriage, is that use of condoms in pregnancy is alien to our culture more so in pregnancy where contraception is unthinkable; and may lead to suspicion of infidelity. Cigarette smoking by a partner (AOR 3.0, 95% CI =0.95-9.4) was a significant predictor of HIV seroconversion in a rural hospital study in Northern Nigeria [23] following multivariate study, unlike in this study. Subjecting the study to Multivariate analysis, women of age group 25-29 years (OR: 3.20; 95%CI: 2.39-4.29) were most likely to seroconvert. Women married to men with sexually transmitted infections, as evidenced by penile discharge, were also associated with an increased risk of HIV seroconversion (OR: 09; 95%CI: 0.01-0.45; p =0.004). Same was the case of those in polygamous setting (OR: 3.98; CI: 1.64-9.65) in this study, which conforms to findings in the study in Northern Nigeria [23], South Africa [24] and Tanzania [22]. Polygamy remains a part of the African culture and Islam, as well as traditional religion. Polygamy is a �legalized� way of having multiple sexual partners and so increases the risk of HIV transmission. Recreational drugs pose a risk for transmission of HIV especially in intravenous drug users who may share needles. It also affects the individual�s judgment thereby reducing a person's ability to make informed choices concerning safe sex and protection from HIV infection. There was an association in this study between recreational drug use and seroconversion; much similar to that found in a Mauritian study [28].
The study revealed a seroconversion rate of 3% among booked, previously HIV negative clients, in labour. Eliminating mother to child transmission of HIV is still quite afar off. Predictors of sero-conversion in the study were patient�s age, alcohol intake, number of partner�s wives, partner�s use of recreational drugs and penile discharge in partners. Re-testing therefore becomes very necessary. Implementation of the recommendation concerning HIV retesting in labour as contained in the National Guidelines on PMTCT 2010, should be made top priority if elimination of MTCT is desired. All facilities in the country should be provided the wherewithal to implement this recommendation. A larger multicenter study involving urban and rural health facilities as well as the private health facilities, would give a clearer picture of the risk factors associated with HIV seroconversion.
What is known about this topic
- HIV seroconversion can occur during pregnancy in a woman who was previously HIV negative;
- Many factors have been implicated in HIV seroconversion during the antenatal period.
What this study adds
- The prevalence of HIV seroconversion among antenatal women in UUTH Uyo is significantly high to warrant routine HIV retesting in late pregnancy or labour;
- The common risk factors identified for HIV seroconversion among antenatal women in UUTH Uyo were patient�s age (25 -29 years), alcohol intake, number of partner�s wives, partner�s use of recreational drugs and penile discharge in partners;
- There is good knowledge (over 95%) of the mode of transmission of HIV among antenatal women in UUTH Uyo.
The authors declare no competing interest.
All the authors have contributed to this study in ways consistent with ICMJE authorship criteria. All authors have read and agreed to the final version of this manuscript and have equally contributed to its content and to the management of the case.
We appreciate Dr A.M Ekanem for his assistance with the analysis of this work. Special thanks also to Miss Affiong Edet Okon for assistance with the laboratory tests.
Table 1: knowledge of antenatal clients in UUTH on the modes of transmission of HIV/AIDS
Table 2: risk factors for HIV among antenatal clients in UUTH
Table 3: relationship between antenatal clients HIV status at delivery and risk factors for sero conversion - client factors
Table 4: relationship between antenatal clients HIV status at delivery and risk factors for sero conversion - partner factors
Table 5: univariate and multivariate analysis of variables that predicts HIV sero- conversion of HIV status of antenatal clients at delivery in UUTH, Uyo
Figure 1: prevalence of HIV seroconversion among booked antenatal clients at delivery in university of Uyo teaching hospital, Uyo, 2013
- Report on the global AIDS epidemic. Geneva, UNAIDS, 2006 http://www.unaids.org/en/HIV_data/2006GlobalReport/default.asp (Accessed 12 July 2006). Google Scholar
- Joint United Nations Programme on HIV/AIDS. AIDS epidemic update 2009. Available at http://bit.ly/iHUsZK. Accessed June 17, 2011. Google Scholar
- Population Reference Bureau. Nigeria PRB 2011 World Population Data Sheet 2011; Available at http://www.prb.org/DataFinder/Geography/Data.aspx?loc=269. Google Scholar
- National Agency for the Control of AIDS (NACA). Joint Annual Review of the National Response to HIV/AIDS. 2011; 201: p103. Google Scholar
- National Agency for the Control of AIDS (NACA). Federal Republic of Nigeria: Global AIDS Response Country Progress Report. Nigeria GARPR 2014. Abuja, Nigeria. 2014. Google Scholar
- Dunn DT, Newell ML, Ades AE, Peckham CS. Risk of Human Immunodeficiency Virus Type 1 transmission through breastfeeding. Lancet. 1992 Sep 5; 340(8819): 585-8. PubMed | Google Scholar
- Liang K, Gui X, Zhang YZ, Zhuang K, Meyers K, Ho DD. A case series of 104 women infected with HIV-1 via blood transfusion postnatally: high rate of HIV-1 transmission to infants through breastfeeding. J Infect Dis. 2009 Sep 1;200(5):682-6. PubMed | Google Scholar
- Garcia PM, Kalish LA, Pitt J, Minkoff H, Quinn TC, Burchett SK et al. Maternal levels of plasma human immunodeficiency virus type 1 rna and the risk of perinatal transmission. Women and Infants Transmission Study Group. N Engl J Med. 1999 Aug 5;341(6):394-402. PubMed | Google Scholar
- Soogoor M, Daar ES. Primary Human Immunodeficiency Virus Type 1 Infection. Curr HIV/AIDS Rep. 2005 Jun; 2(2): 55-60. PubMed | Google Scholar
- Federal Ministry of Health, Nigeria. National Guidelines for prevention of mother-to-child transmission of HIV. 4th ed. Abuja: Federal Ministry of Health; 2010. Google Scholar
- John Kinuthia, J Kiarie, C Farquhar, B Richardson, R Nduati, D Mbori-Ngacha et al. Co-Factors For HIV Incidence during Pregnancy and the Postpartum Period. 17th Conference on Retroviruses and Opportunistic Infections (CROI), San Francisco, 2010. Oral Abstract 155.http://www.retroconference.org/2010/abstracts/38372.htm. (Accessed 20 November 2012). Google Scholar
- Udeh B, Udeh C, Graves N. Perinatal HIV transmission and the cost-effectiveness of screening at 14 weeks' gestation, at the onset of labour, and the rapid testing of infants. BMC Infect Dis. 2008 Dec 31; 8: 174. PubMed | Google Scholar
- Sansom SL, Jamieson DJ, Farnham PG, Bulterys M, Fowler MG. Human Immunodeficiency Virus retesting during pregnancy: costs and effectiveness in preventing perinatal transmission. Obstet Gynecol. 2003; 102(4): 782-790. PubMed | Google Scholar
- CDC. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR. 2006; 55(RR14): 1-17. PubMed | Google Scholar
- Sagay AS, Musa J, Adewole AS, Imade GE, Ekwempu CC, Kapiga S et al. Rapid HIV testing and counselling in labour in a Northern Nigerian setting. Afr J Reprod Health. 2006 Apr; 10(1): 76-80. PubMed | Google Scholar
- UmeononihuOs, Ikechebelu JI, Okonkwo JE, Udigwe GO, Mbachu II. The prevalence of HIV sero-positivity in late pregnancy among antenatal attendees with seronegative status in first half of pregnancy in Nnewi, South East Nigeria. J HIV Hum Reprod [serial online] 2013 [cited 2013 Dec 20]; 1:25-9. Available from: http://www.jhhr.org/text.asp?2013/1/1/25/116533. Google Scholar
- Lu L, Legwaila K, Motswere C, Smit M, Jimbo W, Creek T. HIV Incidence in pregnancy and the first postpartum year and implications for PMTCT programs. Program and Abstracts of the 16th Conference on Retroviruses and Opportunistic Infections (CROI). 2009; Montreal, Quebec, Canada. Abstract 91. http://www.medscape.com/viewarticle/718849.( Accessed 22 November 2012). Google Scholar
- Kieffer MP, Hoffman H, Nlabhatsi B, Mahdi M, Kudiabor K, Wilfert C. Repeat HIV testing in labor and delivery as a standard of care increases ARV provision for women who seroconvert During Pregnancy. 17th Conference on Retroviruses and Opportunistic Infections (CROI), Sanfrancisco. 2010. Oral Abstract 156 retroconference.org/2010/abstracts/37241.htm (Accessed 20 November 2012). Google Scholar
- Moodley D, Esterhuizen TM, Pather T, Chetty V, Ngaleka L. High HIV incidence during pregnancy: compelling reason for repeat HIV testing. AIDS. 2009 Jun 19;23(10):1255-9. PubMed | Google Scholar
- Mbizvo MT, Kasule J, Mahomed K, Nathoo K. HIV-1 seroconversion incidence following pregnancy and delivery among women seronegative at recruitment in Harare, Zimbabwe. Cent Afr J Med. 2001 May; 47(5): 115-8. PubMed | Google Scholar
- Amornkul PN, Vandenhoudt H, Nasokho P, Odhiambo F, Mwaengo D et al. HIV prevalence and associated risk factors among individuals aged 13-34 years in rural Western Kenya. PLoS ONE. 2009; 4(7): e6470. PubMed | Google Scholar
- Msuya SE, Mbizvo E, Hussain A, Uriyo J, Sam NE, Stray-Pedersen B. HIV among pregnant women in Moshi Tanzania: the role of sexual behavior, male partner characteristics and sexually transmitted infections. AIDS Res Ther. 2006 Oct 17; 3: 27. PubMed | Google Scholar
- Etukumana EA, Thacher TD, Sagay AS. HIV Risk factors among pregnant women in a rural Nigerian Hospital. West Indian Med J. 2010; 59 (4): 424-8. PubMed | Google Scholar
- Wand H, Ramjee G. Combined impact of sexual risk behaviors for HIV seroconversion among women in Durban, South Africa: implications for prevention policy and planning. AIDS Behav. 2011 Feb; 15(2): 479-86. PubMed | Google Scholar
- Mbulaiteye SM, Ruberantwari A, Nakiyingi JS, Carpenter LM, Kamali A, Whitworth J. Alcohol and HIV: A Study among sexually active adults in rural Southwest Uganda. Int J Epidemiol. 2000 Oct; 29(5): 911-5. PubMed | Google Scholar
- Morojele NK, Kachieng'a MA, Mokoko E, Nkoko MA, Parry CD, Nkowane AM et al. Alcohol use and sexual behaviour among risky drinkers and bar and shebeen patrons in Gauteng province, South Africa. Soc Sci Med. 2006 Jan; 62(1): 217-27. PubMed | Google Scholar
- Weller S, Davis K. Condom effectiveness in reducing heterosexual HIV transmission. Cochrane Database Syst Rev. 2002; (1): CD003255. PubMed | Google Scholar
- UN Office for Drug Control and Crime Prevention. Civil Society Leader Gives Victims A Voice. Substance Abuse and HIV/AIDS in Africa. Available at: http://www.unodc.org/pdf/newsletter_2002-03-01_1.pdf (Accessed 13 September, 2012). Google Scholar