Extraoviarian granulosa cell tumor: a case report
Saad Rifki Jai, Robleh Hassan Farah, Adil Boussabir, Rachid Boufettal, Farid Chehab
Corresponding author: Robleh Hassan, Service de Chirurgie Visc�rale Aile 3, Centre Hospitalier Universitaire Ibn Rochd, Universit� Hassan II, Casablanca, Maroc 
Received: 27 Sep 2015 - Accepted: 06 Oct 2015 - Published: 15 Mar 2016
Domain: Clinical medicine
Keywords: Granulosa cell tumor, retroperitoneum, extraovarian
©Saad Rifki Jai et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Saad Rifki Jai et al. Extraoviarian granulosa cell tumor: a case report. Pan African Medical Journal. 2016;23:86. [doi: 10.11604/pamj.2016.23.86.8048]
Available online at: https://www.panafrican-med-journal.com//content/article/23/86/full
Extraoviarian granulosa cell tumor: a case report
Saad Rifki Jai1, Robleh Hassan Farah1,&, Adil Boussabir1, Rachid Boufettal1, Farid Chehab1
1Service de Chirurgie Visc�rale Aile 3, Centre Hospitalier Universitaire Ibn Rochd, Universit� Hassan II, Casablanca, Maroc
&Corresponding author
Robleh Hassan, Service de Chirurgie Visc�rale Aile 3, Centre Hospitalier Universitaire Ibn Rochd, Universit� Hassan II, Casablanca, Maroc
Extraovarian granulosa cell tumor (GCT) is a very unusually tumor, assumed to arise from the ectopic gonadal tissue along the embryonal route of the genital ridge. One such rare case of extraovarian granulosa cell tumor was encountered in a 60-year-old female patient who presented with a large intra-abdominal mass. Computerized tomography revealed a large retroperitoneal mass measuring 11 x 10 x 8cm in size. Her past medical history was irrelevant. She underwent exploration laparotomy and the mass was excised. Histopathological examination of the excised mass showed features of adult-type granulosa cell tumor.The tumor showed positivity for inhibin while epithelial membrane antigen was negative thus confirming the diagnosis of granulosa cell tumor. A diagnosis of extraovarian granulosa cell tumor can only be done after excluding any previous history of granulosa cell tumor of the ovary. Immunostains help to differentiate granulosa cell tumors from other neoplasms.
Granulosa cell tumors (GCT) of the ovary are rare tumor, comprising around 2-5% of ovarian neoplasms [1-5]. Adult granulosa cell tumors are considered as sex cord-stromal of the ovary [2]. Historically, Busby considered all tumors currently classified as sex cord-stromal tumors to be “mesenchymomas”, derived from the embroyologic genital ridge [2]. The current classification of ovarian tumors, however, reflects the contention that granulosa cell tumors arise specifically from the proximal precursors of granulosa cells, the “sex cords” [3]. It can recur or metastasize many years after initial treatment and rarely develop at an extraovarian site, even in an oophorectomized patient [4]. Primary extraovarian GCT is an extremely rare tumor. Until 2014, only twelfth cases of such tumors have been reported in the English literature [5]. Our case is the thirteen.
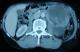
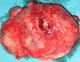
A 60 year old female patient was admitted to Centre Hospitalier universite Ibn Rochd, Casablanca, Maroc, with two months of a localized left upper quadrant abdominal pain, dull aching nature and associated with occasional bouts of vomiting and a history of non-documented weight loss. She had past surgical history of transvaginal resection of uterine fibroid trans-vaginally “hysteroscopy resection of uterine fibroid” (no document). Abdominal examination revealed a palpable and tender intraabdominal mass in the left upper abdominal quadrant; it was about 11cm x 6 cm in its greatest diameter. The laboratory investigations including hematocrit, leukocytes, platelets, kidney function tests, electrolytes and liver function tests were within normal range. Abdominal-pelvic computer tomography imaging shows a retroperitoneal and well-defined cystic mass (11 x 10 x 8cm), with peripheral uptake of the contrast and anteriorly located in relation to the left kidney .The mass had no partitioning and forced back to the contra lateral side the intestinal loops in (Figure 1). The patient underwent exploratory laparotomy with the presumptive diagnosis of intra-abdominal hydatid cyst. The intraoperative findings revealed a large bulging cystic tumor arising from the retroperitoneal space posterolateral to the descending colon, inferior to the spleen and anterior to the left kidney and attached to the splenic flexure by a highly vascularized tissue band, giving the impression of a retroperitoneal sarcoma as shown in (Figure 2). Externally, it was grayish red and smooth with a row of adipose and fibrous tissue tags along one surface. The mass was dissected free and excised (Figure 3). The patient had an uncomplicated postoperative recovery course and she was free of disease four months later. Grossly, the mass was grayish brown in color, soft and cystic with hemorrhagic area measuring 11cm x 6 cm. Cut section revealed solid homogenous grayish tumor with multiple small cystic areas and area of hemorrhage. Microscopic finding, showed small round to oval neoplastic cells with diffuse and trabecular patterns. The cells showed scanty cytoplasm and round to oval nuclei with nuclear grooves and Call-exner bodies (Figure 4). With this typical histopathological feature, a diagnosis of extrovarian GCT was made. The tumor was positive for inhibin and negative for EMA, confirming the diagnosis extraovarian GCT.
�
Post-operative course was unremarkable and patient discharged on 6th day. The histopathological studies with Immunohistochemistry analysis showed a diffuse adult type of a tumor of the granulosa with effraction of its capsule (Figure 5). The patient was called again for a planned second operation which consist hysterectomy with bilateral adnexectomy with hysterectomy, but she refused the operation and any other adjuvant treatment.
Granulosa cell tumor (CGT), an uncommon ovarian cancer [6].It can be of adult or juvenile type based on different clinical and histological features. The more common adult type of CGT usually presents during the perimenopausal or the early menopausal period,the median age being 50-54 years in most series [6,7]. The estimated incidence of GCT in the United States is 0.99/100,000, whereas the reported incidence in other developed countries ranges from 0.4 to 1.7 per 100,000. A number of putative risk factors have been associated with the development of GCT, although most are not reproducible and have unknown clinical or biologic significance [7].In adult women,they may be associated with endometrial hyperplasia ,cystic disease of the breast , endometrial carcinoma[8]. Patients with CGT require long-term follow-up with history, physical examination and tumor markers studies because 17% of relapses occur more than 10 TO 20 years after diagnosis [6,8]. Distant metastasis are rare, but have been reported in many sites [6]. The most common site of recurrence is in the pelvis [5]. Rarely, extraovarian GCT can develop, only twelfth cases have been reported so far in the English literature untill 2014 [5]. It can develop in the retroperitoneum, broad ligament mesentry, omentum, liver or adrenals [5, 6]. Recently, one case of GCT arising a mullerian cyst of the broad ligament has been reported [9]. Our case represents a very rare extraovarian granulosa cell tumor arising in the retroperitoneal space.
�
Typical histological findings of a GCT include small, pale, round to oval granulosa cells with characteristic "coffee-bean" nuclei. The characteristic amorphous globular structures called Call-Exner bodies are present in some GCT[10]. The histogenetic origin of extraovarian sex-cord stromal tumors is thought to be from the ectopic gonadal stromal tissue [2,5,11] with the sex-cord originating from the mesonehros [11]. A dualistic theory of both the coelomic epithelium and the mesonephros in the origin of the pregranulosa cells has also been proposed [6]. Accordingly, the mesonephros itself or at least its functional influence seems to be necessary for creating the sex cords. This is consistent with gonad formation being limited to the gonadal ridge and may explain why the sites of the origin of extraovarian sex cord stromal tumors or supranumerary ovaries are limited to the broad ligament, retroperitoneum, adrenal gland, all of which differentiate close to the mesonephros and mesonephric duct [3].�
The morphological differential diagnosis of GCT includes undifferentiated carcinoma, small cell carcinoma and endometrial stroma sarcoma [7].However, characteristic histological findings and immunostains are helpful in definitive diagnosis.�
Extraovarian GCT should be differentiated from other metastatic carcinomas of ovary that have similar morphology. Inhibin and EMA can help in differentiating of these tumors. GCT is positive for inhibin and negative for EMA [12].It should also be differentiated from other tumors such as small cell carcinoma, undifferentiated carcinoma, endometrial stromal sarcoma, carcinoid, and lymphoma .These tumors do not show positivity for inhibin. IHC for CK, EMA, LCA, CD99 and Chromogranin can help in diagnosing and differentiating these tumors. GCT does not show positivity for EMA, LCA, and Chromogranin [12]. In our present case, strong positivity for inhibin with a negative EMA stain confirmed the diagnosis of GCT. No biologic assay for these tumors markers content of our patient?s urine or blood was realized. Recently, activin B has been introduced as a new marker for post-operative follow-up of patient suffering from GCT [13].�
In the present case, the patient presented with left sided abdominal mass, associated with abdominal pain, the hormonal studies were not done, as the diagnosis of GCT was not suspected. The histopathology features of the tumor are typical of GCT, small pale, round oval nuclei with nuclear groove, and the tumor showed positivity for inhibin. This case is reported for its rarity, and difficulty of diagnosis, and to describe its relevance to the histogenic origin. The diagnosis is made by characteristic histological features. Immunostains like inhibin help in definitive diagnosis.
GCT of the ovary is a rare tumor, and the extraovarian GCT is extremely rare and can present as a retroperitoneal mass. A diagnosis of extraovarian GCT can be made by excluding any previous history of GCT of the ovary. It is Imperative that patients who have been given this diagnostic have lifelong follow up.
The authors declare no competing interests.
All authors have read and approved the final manuscript.
Figure 1: abdominal CT scan, shows abdominal-pelvic mass, well-defined measuring 11/10cm
Figure 2: abdominal-pelvic computer tomography scanning exhibiting a cystic mass with peripheral uptake of the contraste
Figure 3: resected tumor with a smooth and vascularized surface, measuring 11/10cm. the capsule was intact
Figure 4: diffuse and intense staining with vimentin
Figure 5: positive and diffuse immunostaining with cd 56, typical to tumors of the granulosa
- Singh-Ranger G, Sharp A, Crinnion JN. Recurrence of granulosa cell tumour after thirty years with small bowel obstruction.Int Semin Surg Oncol. 2004 May 11;1 (1):4. PubMed | Google Scholar
- Robinson JB, Im DD, logan L, McGuire WP, Rosenshion NB. Extraordinaire granulosa cell tumor; case report. Gyneco oncol. 1999 Jul;74(1):123-7. PubMed | Google Scholar
- Keitoku M, Konish I, Nanbu K, Yamamoto S, Mandai M, Kataoka N et al. extraovarien sex cord stromal tumor: case report and review of the literature. Int J Gyneco pathol. 1997 Apr;16 (2):180-5. PubMed | Google Scholar
- Paul PC, Chakraborty J, Chakrabarti S, Chattopadhyay B. Extraovarian granulosa cell tumor. Indian J Pathol Microbiol. 2009 Apr-Jun;52 (2):231-3. PubMed | Google Scholar
- Ali Abuseini, Nabeh Al-Kaisi .Extra-Ovarian Granulosa Cell Tumor: a Case Report. JRMS. September 2014; 21(4):66-68. PubMed | Google Scholar
- Kim SH, Park HJ, linton JA, shin DH, Yang WI, Chung WY et al. Extraovarian granulosa cell tumor. yonsei Med J. 2001 Jun;42(3):360-3. PubMed | Google Scholar
- Schumer ST, Cannistra SA. Granulosa cell tumor of the ovary. J Clin Onclo. 2003 Mar 15;21(6):1180-9. PubMed | Google Scholar
- Cronj� HS, Niemand I, Ban RH, Woodruff JD. Review of the granulosa-theca cell tumors from the emil Novak ovarian tumor registry. Am J Obstet Gyneco. 1999 Feb;180 (2 Pt 1):323-7. PubMed | Google Scholar
- Sakai Y. Granulosa cell tumor arising in the wall of m�llerian cyst of the broad ligament: report of case and immunohistochemical study. Arch Gynecol Obstet. 2007 Feb;275 (2):145-8. PubMed | Google Scholar
- Lal A, Bourt EP, Nayar R, Defrias DV. Cytologic feartures of granulosa cell tumors in fluids and fine needle aspiration specimens. Acta Cytol. 2004 May-Jun;48(3):315-20. PubMed | Google Scholar
- Griffith LM, Carcangiu ML. Sex cord tumor annulor tubules associated endometrios of the fallopian tube. Am J Clin Pathol. 1991 Aug;96(2):259-62. PubMed | Google Scholar
- Naniwadekar MR, Patil NJ. Extraovarian granulosa cell tumor of mesentery: a case report. Patholog Res Int. 2010 Mar 4; 2010:292606. PubMed | Google Scholar
- Vihko KK, Bl��erm, Puistola U, Tuohimaa P. Activin B in patients with granulosa cell tumors: serum levels in comparison to inhinbin. Acta Obstet Gyneco Scand. 2003 Jun;82(6):570-4. PubMed | Google Scholar