Radiological findings in Behþet disease
Mohame Chraa, Kissani Najib, Lamia Essaadouni
Corresponding author: Mohame Chraa, Neurology Department, Mohamed VI university hospital, lot Maatalah, Mhamid, Marrakesh, 40000, Morocco 
Received: 10 Dec 2014 - Accepted: 08 Jan 2015 - Published: 20 Jan 2015
Domain: Epidemiology
Keywords: Meuro-behçet disease, MRI, central nervous system, brainstem
©Mohame Chraa et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Mohame Chraa et al. Radiological findings in Behþet disease. Pan African Medical Journal. 2015;20:51. [doi: 10.11604/pamj.2015.20.51.5928]
Available online at: https://www.panafrican-med-journal.com//content/article/20/51/full
Radiological findings in Behþet disease
Mohamed Chraa1,&, Kissani Najib1, Lamia Essaadouni2
1Neurology Department, Mohamed VI university hospital, Marrakesh, Morocco, 2Internal medicine department, Mohamed VI university hospital, Marrakesh, Morocco
&Corresponding author
Mohame Chraa, Neurology Department, Mohamed VI university hospital, lot
Maatalah, Mhamid, Marrakesh, 40000, Morocco
Between 5 and 30% of patients with Behþet's disease will present neurological signs during the course of their illness. In order to evaluate the radiological signs on neuro-behþet disease, we studied consecutive patients in whom the diagnosis of this disease was retained, and who referred from January 2004 to December 2011 to the neurology and internal medicine departments in Mohammed VI universitary hospital in Marrakesh. Using 1.5T magnetic resonance imaging (MRI) with axial and coronal T2- weighted, axial Fluid Attenuated Inversion Recovery (FLAIR), pre- and post-contrast axial, coronal and sagittal T1-weighted sequences. The final number of patients in whom the diagnosis criteria of behþet disease were fulfilled and in whom an MRI was performed was 68 cases. Among these patients, 52 had parenchymal form of neuro-behþet with abnormalities in the MRI, 12 had vascular form and 4 patients had normal MRI. The brainstem, cerebral white matter, basal ganglia, internal capsule, thalamus and spinal cord were involved in forty four, thirty one, thirty, twenty nine, seventeen and four patients, respectively. The cerebral peduncle was the brainstem structure mainly involved with thirty cases followed by the pons with, twenty cases. Midbrain involvement interested forty patients. Brainstem atrophy was seen in eighteen cases. Finally, control MRI were obtained in four cases only, and showed changes in lesions size and shape.
Behþet┤s Disease (BD) is a rare idiopathic chronic relapsing multisystem vascular-inflammatory disease. It was initially described by the Turkish dermatologist Dr. Hulusi Behþet in 1937 who described the triad of oral aphthous ulceration, genital ulcers and relapsing uveitis/iritis [1]. Other symptoms include CNS involvement, arthritis or arthralgia, gastrointestinal features, epididymitis, arterial occlusion or aneurysms, thrombophlebitis and deep vein thrombosis. Neurological involvement, which occurs in up to 49% of all cases, is often quite severe with a high rate of mortality and morbidity [2]. Radiologically, neuro-behþet disease (NBD) usually involves the white matter, brainstem, basal ganglia and thalamus [3]. Typical cerebral MRI often reveals T1 iso/hypointense and T2 hyperintense lesions [2, 4]. This study describes the pattern of radiological finding in NBD.
Authors retrospectively studied the brain magnetic resonance imaging (MRI) findings of 68 patients diagnosed with NBD at the departments of neurology and internal medicine in Mohammed VI universitary hospital in Marrakesh from January 2004 to December 2011. The diagnosis of BD was performed by a senior neurologist or internal medicine physician, our patients fulfilled the criteria of the International Study Group for BD [1], the 68 patients included in this work all presented with neurological syndrome. Information extracted from the folders included age, gender, clinical form, neurological and extra neurological features, brain MRI finding as well as the clinical outcome and the MRI evolution of lesion after treatment if available (only 4 patients performed control MRI giving the lack of financial resources). Of the 68 patients, 4 patient┤s MRI was normal but the NBD diagnosis was clinically certain, furthermore, cerebrospinal fluid (CSF) study was abnormal in all these 4 cases. Clinically, patients who had MRI abnormalities were divided into parenchymal and non-parenchymal groups, with 52 and 12 respectively. The course of the disease was either relapsing-remitting in 27 cases, progressive in 30 cases or monophasic in 11 cases. The MRI sequences included axial and coronal T2- weighted, axial Fluid Attenuated Inversion Recovery (FLAIR), pre- and post-contrast axial, coronal and sagittal T1-weighted sequences. Number; location; size; MR signal abnormalities; contrast enhancement; and presence of cerebral or brainstem atrophy were evaluated in patients with parenchymal involvement.
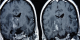
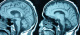
In our series, 68 patients developed neurological symptoms related to BD. The main neurological symptoms revealing neurobehþet in our patients were in order of frequency; pyramidal signs, dysarthria, cerebellar signs with 49, 35, and 24 respectively, followed by other neurological symptoms (Table 1). Intracranial hypertension was the main neurological symptom in patients with extraparenchymal NBD. Other systemic manifestations included; oral in all patients, genital, ocular, dermatological, vascular and articular involvement (Table 2). The clinical presentation in our patients was monophasic in 11 cases, relapsing-remitting in 27 cases and progressive in 30 cases. Of all patients, we found 52 who presented with parenchymal MRI lesions, 12 who had extraparenchymal involvement (deep venous thrombosis), and in four cases the MRI was normal. Of those with parenchymal signs, the brainstem, cerebral white matter, basal ganglia, internal capsule, thalamus and spinal cord were involved in forty four, thirty one, thirty, twenty nine, seventeen and four patients, respectively (Table 3). Typically, the lesions were in hypersignal in T2 and FLAIR weighted MRI sequences, and in hyposignal T1 weighted sequences, they were not limited to one region but interested at least two structures, mainly an association of brainstem and midbrain structures (Figure 1). In 15 cases we found atypical MRI presentations like periventricular and white matter non specific lesions or confusing results when there is an association of vascular factors like hypertension (Figure 2). The brainstem lesions were ventrally located in thirty six up of the forty four patients who had MRI lesions in this region, and interested the cerebral peduncle in thirty cases followed by the pons with twenty cases (Figure 3). The lesions size was medium to large sized in the majority of cases, with pseudotumoral forms in three cases (Figure 4). In thirty three patients we had contrast enhancement after gadolinium injection, this was heterogeneous enhancement particularly present when the lesions are large. We also observed brainstem atrophy in eighteen cases and a cortico or/and subcortical one in six cases (Figure 5), we found these patients to have a chronic form in sixteen cases and a relapsing one in eight cases. In patients with extraparenchymal form, MR angiography which was performed in all patients confirmed the diagnosis. All our patients underwent a treatment associating intravenous bolus of corticoids (1 g per day for three days) in association cyclophosphamid (1 g per day for several months). The clinical evolution is summarized in Table 5. Four patients underwent MRI control after the treatment was stopped and showed improvement in lesions size and shape.
BD is a systemic vasculitis which involves the central nervous system in up to 50 % of cases [5], and is a devastating form in this case because of its high morbidities. Neurological symptoms are usually correlated with radiological findings. NBD can be separated into two forms; parenchymal and extraparenchymal (cerebro-vascular thrombosis) presentations. The extraparenchymal form occurs in up to 35% of cases [6], in our series we had 12 cases out of 68 (17.6%). MRI has a major role in asserting the diagnosis of neurobehþet disease as well as in understanding its physiopathology [7], especially because of the difficulties to obtain histopathological studies. In the parenchymal form of NBD, the lesions are usually multiple small foci of high intensity on T2 weighted sequences, although in our series we found them to be medium to large in the majority of cases. They are usually extensive, confluent and distributed over white matter without predilection for the periventricular regions in opposition to multiple sclerosis (MS) [8-11], besides in NBD the basal ganglia are involved which is atypical in MS. In another hand brainstem lesions we observed are ventrally localized, which was the case in other observation [12], in opposition to MS where the floor of the fourth ventricle and the middle cerebellar peduncle are frequently concerned [13], we observed an involvement of the later in only three cases. The lesions interests the white matter (70%), the brainstem (60%), the basal ganglia and the thalamus (40%) [13], in our series the brainstem was the first involved, followed by the white matter, but the majority of our patients had an association of brainstem and midbrain structure involvement. So we can say that MRI lesions of the mesocephalo-diancephalic junction are highly suggestive of NBD. Another differential diagnosis with NBD is lupus erythematous, here brainstem lesion can help differentiate between the two giving the fact that its involvement is rare in the last one [14], and the grey matter is more frequently involved in lupus and other vasculitis [13]. One remarkable finding is the frequency of contrast enhancement lesions in NBD (19 out of 52). Finally, the follow up showed a reversibility of lesions size in the four patients in whom a control MRI had been performed, which may reflect a reversible breakdown in the blood-brain barrier [15-18], this is inconsistent with other results where the authors had either an increase in lesion burden [7] or a lack of reversibility of the lesions [13].
Although a retrospective study, our work offer results that are important in terms of MRI lesions characteristics in NBD (topography, size, contrast enhancement, atrophy). MRI was a powerful tool that help us in the diagnosis of NBD, the pattern of lesions found in this work can be highly suggestive of this diagnosis. Furthermore, the reversibility of the lesions in control MRI is a proof of the necessity to initiate an aggressive treatment when the diagnosis of NBD is suggested.
The authors declare no competing interests.
All the authors have contributed to this study in ways that comply to ICMJE authorship criteria. All the authors have read and approved the final version of the manuscript.
Table 1: neurological symptoms in order of frequency
Table 2: system organs involvement
Table 3: localisation of MRI lesions
Table 4: clinical evolution of our patients
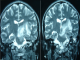
Figure 1: T2 weighted MRI sequences showing typical presentation of NBD with an association of brainstem and midbrain hypersignal lesions
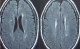
Figure 2: flair weighted MRI sequences of two patients showing atypical presentations of neurobehþet disease
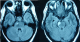
Figure 3: flair weighted MRI sequences showing brainstem lesions with ventral localization
Figure 4: pseudotumoral forms of neurobehþet disease
Figure 5: T1 weighted MRI sequences showing brainstem atrophy in a patient with NBD
- International Study Group for Behþet's Disease. Criteria for diagnosis of Behþet's disease. Lancet. 1990 May 5;335(8697):1078-80. PubMed | Google Scholar
- Borhani Haghighi A, Pourmand R, Nikseresht AR. Neuro-Behþet disease- A review. Neurologist. 2005 Mar;11(2):80-9. PubMed | Google Scholar
- Borhani Haghighi A, Samangooei S, Ashjazadeh N et al (2006). Neurological manifestation of Behþet's disease. Neurosciences (Riyadh). 2006 Oct;11(4):260-4. PubMed | Google Scholar
- Wechsler B, Dell'lscla B, Vidailhet M, et al. MRI in 31 patients with Behþet's disease and neurological involvement: prospective study with clinical correlation. Journal of Neurology Neurosurgery and Psychiatry. 1993 Jul;56 (7):793-8.. PubMed | Google Scholar
- Siva A, Altintas A, Saip S. Behþet's syndrome and the nervous system. Curr Opin Neurol. 2004 Jun;17 (3):347-57. PubMed | Google Scholar
- Wolf SM, Schotland DL, Phillips LL. Involvement of nervous system in Behcet's syndrome. Arch Neurol. 1965; 12:315-25. PubMed | Google Scholar
- O'Duffy JD, Goldstein NP. Neurologic involvement in seven patients with Behcet's disease. Am J Med. 1976 Aug; 61 (2):170-8. PubMed | Google Scholar
- Essaadouni L, Jaafari H, Abouzaid CH, Kissani N. Les manifestations neurologiques de la maladie de Behþet : Útude de 67 patients. Revue Neurologique. 2010 Aug-Sep;166 (8-9):727-33. PubMed | Google Scholar
- Afshin Borhani Haghighi, Sirous Sarhadi, Siamak Farahangiz. MRI findings of neuro-Behcet's disease. Clin Rheumatol. 2011 Jun;30(6):765-70. PubMed | Google Scholar
- Vidaller A, Carratala J, Moreno R, Arbizu T, Rubio F. Magnetic resonance imaging in neuro-Behset's disease. Br J Rheumatol. 1988 Feb;27(1):79-80. PubMed | Google Scholar
- Siva, A, Necdet, V, et al. Neuroradiological finding in neuro-Behþet syndrome. In: Behþet's disease, basic and clinical aspects (ed. JD O'Duffy, E Kokmen), Marcel Dekker, New York. 1991; 323- 329. Google Scholar
- Zuheir Al Kawi M, Bohlega S, Banna M. MRI findings in neuro-Behþet's disease. Neurology. 1991 Mar;41(3): 405-8. PubMed | Google Scholar
- Banna M, El Ramahl K. Neurologic involvement in Behþet disease: imaging findings in 16 patients. Am J Neuroradiol. 1991 Jul-Aug;12(4):791-6. PubMed | Google Scholar
- Sang Hoon Lee et Al. MRI Findings in Neuro-Behþet's Disease. Clinical Radiology. 2001 Jun;56 (6):485-94. PubMed | Google Scholar
- SuzukiK, Hara M, Nakajima S, et al. Analysis of systemic lupus erythematosus (SLE) involving the central nervous system by magnetic resonance imaging (MRI). Ryumachi. 1989 Apr;29 (2):88-96. PubMed | Google Scholar
- Willeit J, Schmutzhard E, Aichner F, Mayr U, Weber F, Gerstenbrand F. CT and MR imaging in neuro-Behþet disease. J Comput Assist Tomogr. 1986 Mar-Apr;10(2):313-5. PubMed | Google Scholar
- Patel DV, Neuman MJ, Hier DB. Reversibility of CT and MR findings in neuro-Behpet disease. J Comput Assist Tomogr. 1989 Jul-Aug;13(4):669-73. PubMed | Google Scholar
- Gerber S, Biondi A, Dormont D, Wechsler B, Marsault C. Long-term MR follow-up of cerebral lesions in neuro-Behþet's disease. Neuroradiology 1996 Nov;38(8):761-8. Google Scholar