Environmental surveillance of poliovirus and non-polio enterovirus in urban sewage in Dakar, Senegal (2007-2013)
Abdou Kader Ndiaye, Pape Amadou Mbathio Diop, Ousmane Madiagne Diop
Corresponding author: Abdou Kader Ndiaye, Centre National de R�f�rence des Ent�rovirus, Unit� de Virologie M�dicale, Fondation Institut Pasteur de Dakar, S�n�gal 
Received: 03 Nov 2013 - Accepted: 23 Jul 2014 - Published: 04 Nov 2014
Domain: Other
Keywords: Enteroviruses, poliovirus, sewage
©Abdou Kader Ndiaye et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Abdou Kader Ndiaye et al. Environmental surveillance of poliovirus and non-polio enterovirus in urban sewage in Dakar, Senegal (2007-2013). Pan African Medical Journal. 2014;19:243. [doi: 10.11604/pamj.2014.19.243.3538]
Available online at: https://www.panafrican-med-journal.com//content/article/19/243/full
Original article 
Environmental surveillance of poliovirus and non-polio enterovirus in urban sewage in Dakar, Senegal (2007-2013)
Environmental surveillance of poliovirus and non-polio enterovirus in urban sewage in Dakar, Senegal (2007-2013)
Abdou Kader Ndiaye1,&, Pape Amadou Mbathio Diop1, Ousmane Madiagne Diop1
1National Reference Center for Enteric Viruses, Medical Virology Unit, Institut Pasteur de Dakar, Senegal
&Corresponding author
Abdou Kader Ndiaye, Centre National de R�f�rence des Ent�rovirus, Unit� de Virologie M�dicale, Fondation Institut Pasteur de Dakar, S�n�gal
Introduction: Global poliomyelitis eradication initiative relies on (i) laboratory based surveillance of acute flaccid surveillance (AFP) to monitor the circulation of wild poliovirus in a population, and (ii) vaccination to prevent its diffusion. However, as poliovirus can survive in the environment namely in sewage, environmental surveillance (ES) is of growing importance as the eradication target is close. This study aimed to assess polioviruses and non polio enteroviruses circulation in sewage drains covering a significant population of Dakar.
Methods: from April 2007 to May 2013, 271 specimens of raw sewage were collected using the grab method in 6 neighborhoods of Dakar. Samples were processed to extract and concentrate viruses using polyethylene glycol and Dextran (two-phase separation method). Isolation of enteroviruses was attempted in RD, L20B and Hep2 cell lines. Polioviruses were identified by RT-PCR and Elisa. Non Polio Enteroviruses (NPEVs) were identified by RT-PCR and microneutralisation tests.
Results: polioviruses and NPEVs were respectively detected in 34,3% and 42,8% sewage samples. No wild poliovirus neither circulating vaccine-derived Poliovirus (cVDPV) was detected. Neutralization assays have identified 49 non polio enteroviruses that were subsequently classified in 13 serotypes belonging to HEV-A (22, 4%), HEV-B (12, 24%), HEV-C (26, 53%) and HEV-D (6, 12%) species.
Conclusion: this study is the first documentation of enteroviruses environmental detection in Senegal. It shows the usefulness of environmental surveillance for indirect monitoring of the circulation and distribution of enteroviruses in the community.
Human enteroviruses (HEVs) are ubiquitous, infecting ≈1 billion persons worldwide usually by fecal-oral route [1, 2]. They are associated with a wide spectrum of acute diseases from minor febrile illness to severe ones (aseptic meningitis, encephalitis, paralysis, myocarditis, neonatal enteroviral sepsis, etc). Enteroviruses could be related to chronic human disease like diabetes type 1 or cardiomyopathy [3, 4]. The risk of infection is directly correlated with poor hygiene and sanitation and overcrowding especially among population with bad quality of immunization. HEVs are classified in 4 species (HEV-A, HEV-B, HEV-C and HEV-D) covering more than 100 serotypes. Polioviruses (with 3 serotypes named PV1, PV2, and PV3) belong to HEV-C [5]. In 1988, WHO launched the Global Poliomyelitis Eradication Initiative based on effective immunization of children with efficient polio vaccines and monitoring of wild poliovirus (WPV) transmission through surveillance of Acute Flaccid Paralysis among children less than 15 years. The program mainly relied on mass immunization with the trivalent oral poliomyelitis vaccine. In 1998, paralytic poliomyelitis was endemic in more than 125 countries in 5 continents and was affecting more than a thousand children every day. In 2013, only three countries remain endemic for indigenous transmission of WPV (Afghanistan, Nigeria, and Pakistan) and the number of cases declined by more than 99 % (from 350,000 in 1988 to 223 as of 23 October 2013 including 217 cases in endemic countries). As the Global Poliomyelitis Eradication Initiative is getting closer to the target date of 2015, timeliness and accuracy for detecting wild poliovirus circulation in population is critical as the proportion of AFP cases caused by polioviruses will approach zero, and there will be a loss of sensitivity of AFP surveillance [6]. Notwithstanding presence or absence of clinical symptoms, the poliovirus is continually replicated in intestinal lymphatic tissue for several weeks and is excreted and shed in environment through faces. This excretion may contaminate surface water sources for drinking water, recreational activities, aquaculture and irrigation. The amount of virus so excreted can reach 107 infectious dose/day per person. Environment might lead to poliovirus reintroduction in certified "polio-free-area" especially in population groups with low immunization coverage. Oral poliovirus vaccine strains, vaccine-derived polioviruses (VDPV), and even wild-type poliovirus strains may remain infectious for as long as 2 months in sewage depending upon environmental factors (inactivation by sunlight, high temperature, etc.). Circulation of EVs in sewage is a proven indicator of their presence in given communities [7]. Environmental surveillance can provide valuable supplementary information, particularly in urban populations with bad quality of surveillance, particularly in context of virus circulation or re-introduction [8].
�
Several studies have demonstrated the usefulness of environmental surveillance as an additional tool to determine the epidemiology of viruses circulating in given communities [9-24]. In Nigeria, in 2012, wild poliovirus type 1(WPV1) was isolated from multiple sewage samples from Kano and Sokoto state [25]. In Egypt, WPV1 was isolated from two samples collected in Cairo in December 2012. The WPV1 sequences from these isolates were similar to WPV1 circulating in northern Sindh, Pakistan. WPV has not been detected in persons with AFP in Egypt since 2004 [26]. Israel has detected 67 wild poliovirus type 1 (WPV1) positive sewage samples from 24 sampling sites, collected from 3 February 2013 to 4 August 2013 [27-30]. WHO has confirmed an outbreak of at least ten cases of polio in Syria, where vaccination coverage has dramatically decreased during the civil war.�
The World Health Organization has included environmental Poliovirus surveillance in the new Strategic Plan of the Global Polio Eradication Initiative, as a supplement to Acute Flask Paralysis (AFP) surveillance. In that perspective enhanced AFP surveillance combined with environmental surveillance of polioviruses by examining sewage samples which has been shown to be more sensitive for detection of low circulation of WPV and VDPV should be an interesting strategy to implement for eradication. Indeed many viral agents including polioviruses and non polio enteroviruses have been recovered from none or inefficiently treated sewage water. In Senegal, thousands of doses of OPV are delivered every year to control PV circulation. Hence, WPV was not detected from 1998 to 2010 when an outbreak with 18 AFP cases with WPV occurred between January and April following separate introductions from Mauritania and Guinea. Since then, no WPV was detected. We initiated sampling of sewage in selected sewage plants in Dakar as pilot study for feasibility of environmental surveillance, to detect early introduction and/or silent transmission of wild poliovirus or VDPV and then facilitate a rapid response/control.
Sampling sites
�
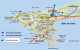
Dakar is the westernmost city on Africa mainland and is located on the Cap-Vert Peninsula in the Atlantic Ocean. It is divided into 19 administrative areas and the population is approximately 3,000,000 (Figure 1). Six sites were selected based on size of population, socioeconomic status, accessibility, immunization coverage, sanitation, hygiene conditions and proximity with industrial areas to avoid drainage of chemical wastes. Since 1989, Dakar wastewater system is divided in 2 parts: a) The grey waters of the northern part of Dakar (concerning around 1,600,000 people) are partly drained to the sea after pre-treatment at Camberene sewage treatment plant. A portion of treated water is utilized for irrigating agricultural areas and providing the major part of vegetables sold in several markets in Dakar. b) For the Western and Southern zones of the peninsula the untreated grey water is directly drained to the sea after mechanical removal of main floating objects (rags, plastics, sticks, cans, etc.) (Figure 1).�
Sewage collection and processing�
From April 2007 (Week 18) to May 2013 (Week 20) raw sewages were collected between 09.00 am and 11.00 am using the grab method as described in the WHO Guidelines for Environmental Surveillance of Poliovirus. Briefly, 1 liter of sewage (after decantation within the plant or directly in trenches) was collected using a bucket and with strict compliance to safety requirements. Sewage samples were transferred into clean and sterile labeled glass bottles, stored in a cooler and transported to the Polio Laboratory at Institut Pasteur de Dakar for processing within 1 h after collection. The concentration of wastewater samples was performed according to the method described previously by Lewis and Metcalf [31]. Briefly, the sewage sample was centrifuged at 2,500 rpm (1000 � g) for 10 min at 4�C in a centrifuge using swinging rotor. The supernatant was then collected and the pH adjusted to 7 -7.5.The pellet was stored at 4�C for further use. For each sample, 35 mg of Sodium chloride 5M, 287 ml of polyethylene glycol 6000 at 29% (i.e. 60 g per liter) and 39, 5 ml of Dextran T40 at 22% were added to 500 ml of sewage supernatant. The mixture was stirred with a magnetic stirrer for 60 min at 4�c and poured into a sterile separation funnel for each sample and left to stand overnight at +4�C to allow phase separation. The entire lower phase was harvested and the interphase collected slowly into a sterile 50ml centrifuge tube. The initially saved pellet was then resuspended into the harvested concentrate. Twenty percent chloroform was added to it, mixed thoroughly by vortexing and then centrifuged at 4�C for 20 min at 1500g. Fungizone (0,5%), Penicillin G (100 UI/ml) and Streptomycin (100 mg/ml) were added after chloroform extraction and aliquots were stored at -20�c until inoculation into cells.�
Viral isolation�
Isolation of Poliovirus was carried out in low-passaged RD (rhabdomyosarcoma), L20B (cells genetically engineered to express the human poliovirus receptor [32,33], and Hep2 (Human Caucasian larynx carcinoma epithelial cells) cell-lines. Flat-sided plastic tubes seeded with RD, L2OB, and Hep2 cells were inoculated in duplicate with 0.2ml of specimen concentrate, incubated at 36�C for 5 days. A culture was considered "positive for virus replication" when at least 75% of the cell monolayer presents CPE (cytopathic effect). All positive tubes were kept at -20�C while any culture negative in RD, L20B, and Hep2 was passaged on fresh cells and examined for 5 more days. Sample showing CPE in L20B was classified as "suspected poliovirus". Any culture negative in L20B but positive in RD or Hep2 was classified as "non polio enterovirus".�
Poliovirus typing and intratypic differentiation (ITD)�
All cell culture positive samples collected from 2007 to 2010 were characterized using a RT-PCR kit (provided by Centers of Disease Control and Prevention, Atlanta, USA) for the ITD of polioviruses including separate reactions with pan-enterovirus and pan-poliovirus primers. Confirmed polioviruses were further characterized using serotype specific, and Sabin type 1, 2, and 3 specific primers. Amplified products were loaded on 10% polyacrylamide gels (Bio-Rad) and electrophoresed at 100 V. The PCR products were visualized under UV lights after staining for 5 min in 2 �g/ml ethidium bromide. Cell culture positive samples collected from 2011 to 2013 were submitted to a rRT-PCR assay from Centers of Disease Control and Prevention (CDC) which amplifies the VP1 region of the poliovirus genome. Two assays were successively run: Real-time ITD Assay using six set of primers: Pan Enterovirus, Pan Poliovirus, Poliovirus serotype 1, 2 and 3, and multiplexed targeting Sabin 1, 2 and 3); Real-time VDPV Screening Assay (targeting known Sabin 1, 2 and 3 regions involved in Sabin viruses reversion to neurovirulence) Poliovirus isolates showing discrepancies between rRT-PCR ITD test and VDPV screening may represent Vaccine-Derived Polioviruses (VDPVs) i.e. drifted viruses from parent OPV strains. They were further characterized by sequencing of the full VP1 region of the genome [34].�
Identification of non-polio enteroviruses isolates�
Micro-neutralization tests with Lim Benyesh-Melnick (LB-M) pools of mixed equine antisera specific for common EV serotypes were used for identification of selected non-polio enteroviruses according to WHO standard protocols.Viruses that are non typable by microneutralization were classified as untypable.�
Intratypic differentiation of poliovirus strains by enzyme-linked immunosorbent assay�
As polioviruses mixture is frequent in sewage specimens and that homotypic mixtures cannot be excluded consequently, All type 1 and type 3 poliovirus isolates obtained after microneutralisation were tested on Elisa as described according to Who standards protocols [35].
From the 271 samples of raw sewage collected in all sites, 216 (79, 7%) were positive at least on one cell line. CPE on the 3 cell lines was observed in 43 samples (15, 9 %), whereas 73 samples (27%) showed CPE in only one cell line (49 on RD only and 24 on Hep-2c only). Polioviruses (PV) were detected in 93 samples (34, 3 %) and all came out as Sabin-Like strains (N=110) after intratypic differentiation by rRT-PCR. Repartition within the three serotypes was as follows: 29 PV1 (31.18%), 58 PV2 (62.37 %), and 28 PV3 (30, 11%) (Of note, 22 samples came out as mixtures of Sabin Like Polioviruses (Table 1). Non polio enteroviruses only were isolated from 116 samples i.e. 42, 8 % of the collected samples. Twenty three poliovirus (PV) Sabin Like 2 and 1 PV Sabin Like 3 polioviruses showed discordances between the 2 PCR assays used. They were sequenced in VP1 and compared to Sabin viruses in order to see if they met VDPV definition i.e. > 5 nucleotides difference for PV2 and > 9 nucleotides difference for PV1 and PV3 VDPV. The nucleotides changes were not significant between the virus sent for sequencing and the homotypic Sabin virus. None vaccine-derived poliovirus (VDPV) was detected in our study. No double reactive result, i.e. homotypic mixture of wild and sabin viruses, was noticed. Out of the 116 isolates negative on L20B and positive on Hep2 and/or RD cell lines, 49 non polio enteroviruses (42,2%), confirmed by Pan EV RT-PCR were randomly selected for identification by microneutralization tests. From the 33 serotypable EVs, 14 different serotypes of non polio enteroviruses were detected (Figure 2): 11 HEV-A (33, 3 %), 6 HEV- B (18.2 %), 14 HEV-C (42.4 %) and 2 HEV-D (6 %).Of note, 2 parechoviruses were identified. Fourteen non polio enteroviruses (28, 6 % of the selected NPEVs) were non typable by antisera pools. No seasonal patterns were noted, particularly for 2008 with a year round environmental surveillance. 3 peaks are noted for February, June and October.
A wide variety of EVs belonging to the four human enterovirus species were found in collected samples, with an isolation rate (79, 7%) consistent to what was described in different studies [36, 37]. Precedent surveys showed a total isolation rate of 65,15% in Yopougon (Abidjan) [38]. In Iran, Enteroviruses and Non-Polio Enteroviruses were respectively isolated from 49 (56.98%) and 46 (53.49%) of specimens [39]. In an another study in three Iranian provinces (Sistan & Baluchestan, Tehran and Fars) in 2009, authors isolated from specimens respectively 49 enteroviruses (56.98%), 38(60.32%) and 11(22.92%) [40]. In Finland, 72 enteroviruses (77, 4%) were identified positives out of 93 studied from sewage. HEV-C species was predominant in our study (42, 4%), followed by HEV-A (33, 3%), HEV-B (18,2%) and HEV-D (6%) [41].
�
In Singapore, a study detected same prevalence in environmental surveillance with76,5% of HEV-C (coxsackieviruses A-1, A-11, A-17, A22 and A-24), followed by HEV-A species (64,7%) with enterovirus 71 and 89, coxsackievirus A2, A5 and A16. HEV-B species represented 41,2%. HEV-D species were no detected in this study [42]. In our study, Coxsackieviruses represented the majority (75% of serotyped viruses) with coxsackie A-7 and coxsackie A-11 being the more represented. Some of these EVs are associated with diseases in human; EV68 who have both properties of enteroviruses and rhinoviruses is related to severe respiratory diseases [43, 44], EV 70 and Cox-A24 are the major etiological agents involved in acute hemorrhagic conjunctivitis (AHC) outbreaks worldwide, the first AHC outbreak was described in 1969 in Ghana, West Africa, and was called Apollo disease. Since this first reports, the infection has been described in numerous other countries, (China, India, Egypt, Cuba, Singapore, Taiwan, Japan, Pakistan, Thailand, United States, etc.) Massive outbreaks of AHC periodically occur in tropical areas and involve large populations [45-48] EV-19 EV-21 and are implicated in congenital and perinatal infection and recently associated to acute flaccid paralysis and fulminant hepatitis [49,50]. CV-A3, CV-A4, CV-A11, CV-A13 and CV-A17 are associated with herpangina and Hand Foot and Mouth Disease. 2 echoviruses 23 (now reclassified as parechovirus) were identified; Those viruses are recognized to cause mild gastro intestinal and respiratory illness, myocarditis and encephalitis mainly in children less than 5 years old [51,52].�
In Iran, previous study in environment showed a wide variety of Enterovirus Non Polio with EV-11 (31.52%), CV-B (27.58%), EV-7 (17.73%) and EV-4 (21.67%) [53]. 28, 6% of Enterovirus Non Polio were non typable in our study by microneutralization using two pools of antisera, 77% were non typable by using one pool of anti sera in Iranian observation [53]. We can observe large fluctuations in circulation over time. In temperate climates, enteroviral infections are most common in the summer and early fall, but seasonal patterns are less evident in tropical areas where circulation trends are year round. In a Ghanaian study, the peaks of NPEVs' infection were between July and August and in a lower level from November to January.
This study is the first one depicting enteroviruses detection in sewage specimens in Senegal. Even though some disease-specific programmes, like the polio eradication programme, has proven to be efficient in detecting EVs circulating within the population, very little is known about implications of EVs in a vast array of diseases in tropical settings. Results gathered from environmental surveillance of EVs can help targeting specific disease-associated pathogens in the community within the catchment area of sampling sites. Most importantly, poliovirus were found in approximately one third of the samples, proving that the sampling sites can be successfully used to sensitively monitor poliovirus (wild, VDPV and Sabin) circulation within a population pre- and post-eradication. Moreover, this will add value on AFP surveillance, not only in Senegal but also in west Africa as Dakar is an important hub for travelers/migrants from other west-african countries. Moreover, 42% of EVs that were identified belongs to HEV-C species that are known to recombine with PV and increase the probability of VDPV emergence. [54] On another perspective, enteroviruses may be a source of contamination in community through utilization of improperly treated wastewater for drinking, bathing, and irrigating especially in developing countries where resources in water are limited. This study set the basis for an agreement between the National Polio laboratory and ONAS (Office National de l'Assainissement du Senegal), the company managing sewage treatment plants, to use this protocol for (i) assessing effectiveness of water treatment that aim to ride off viral agents and (ii) monitoring of treated water that is further used for agricultural purposes.
The authors declare no competing interests.
A.KNDIAYE and P.A.M.DIOP participated equally to coordinate the work and drafted the manuscript. O.M DIOP, designed and partly coordinated the study, and reviewed the manuscript. All authors have read and agreed to the final version of this manuscript.
The study was financially supported by Institut Pasteur International Network. We are grateful to (i) Dave Kilpatrick, from US CDC, Atlanta, who provided molecular reagents for poliovirus detection, and (ii) Harrie Van Der Avoort, from RIVM, the Netherlands, who provided antisera and Elisa reagents. We are indebtful to Nicksy Gumede and Raffaella Williams from NICD, South-Africa who carried out the sequencing of suspected VDPVs. This study was made possible by Directors and laboratory technicians of ONAS (Office National de l'Assainissement du Senegal) who provided administrative authorizations and technical assistance, respectively, for sewage samples collection. Special thanks to Mohamet Dia for technical assistance for wastewaters concentration in our laboratory.
Table 1: polioviruses isolated from sewage samples
Figure 1 : map of Dakar indicating sampling sites from which the specimens have been collected for environmental surveillance for enteroviruses
Figure 2 : identification of NPEVs isolates
- Pallansch M, Roos R. Enteroviruses: polioviruses, coxsackieviruses, echoviruses, and newer enteroviruses. In: Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B, et al. Fields virology. 5th ed. Philadelphia: Lippincott Williams & Wilkins; 2007 ; 839-93. PubMed | Google Scholar
- Khetsuriani N, Parashar UD. Enteric viral infections. In: Dale DC, Federman DD, eds. Scientific American medicine. New York, NY: Web MD, Inc.; 2003:1758-66. PubMed | Google Scholar
- World Health Organization (2002). Enteroviruses-non polio. Media centre. Available at: http://www.who.int/ mediacentre/ factsheets/fs174/en Accessed on May 2014. PubMed | Google Scholar
- Sinha S. (2006). Enteroviruses. eMedicine from webMD. Available at: http://www. emedicine.com/MED/topic681.htm. Accessed 2 May 2014. PubMed | Google Scholar
- http://www.picornaviridae.com. Accessed 17 Avril 2014. PubMed | Google Scholar
- http://www.polioeradication.org. Accessed July 2014. PubMed | Google Scholar
- Wyn-Jones AP. and Sellwood J. Enteric viruses in the aquatic environment. J Appl Microbiol 2001 Dec; 91(6): 945-962. PubMed | Google Scholar
- WHO/V&B/03.03 Guidelines for environmental surveillance of Poliovirus circulation. 2003. http://www.who.int/vaccines-documents/ Accessed on July 2014. PubMed | Google Scholar
- 9.Hovi T. Surveillance for polioviruses. Biologicals. 2006 Jun; 34(2): 123-6. PubMed | Google Scholar
- Gary HE, Sanders R, Pallansch MA. A theoretical framework for evaluating the sensitivity of surveillance for detecting wild poliovirus: factors affecting detection sensitivity in a population with circulating wild poliovirus. J Infect Dis. 1997 Feb; 175(Suppl 1): S141-5. PubMed | Google Scholar
- Dowdle W, Van Der Avoort HG, De Gourville E et al. Containment of polioviruses after eradication and OPV cessation: characterizing risks to improve management. Risk Anal. 2006 Dec; 26 (6):1449-69. PubMed | Google Scholar
- Bottiger M, Herrstrom E. Isolation of polioviruses from sewage and their characteristics: experience over two decades in Sweden. Scand J Infect Dis. 1992; 24(2):151-5. PubMed | Google Scholar
- Deshpande JM, Shetty SJ, Siddiqui ZA. Environmental surveillance system to track wild poliovirus transmission. Appl Environ Microbiol. 2003 May; 69 (5): 2919-27. PubMed | Google Scholar
- Shulman LM, Manor Y, Sofe D, Handsher R, Swartz T, Delpeyroux F, Mendelson E. Neurovirulent vaccine-derived polioviruses in sewage from highly immune populations. PLoS One. 2006 Dec 20; 1: e69. PubMed | Google Scholar
- Vinj� J, Gregoricus N, Martin J, Gary HE Jr, Caceres VM, Venczel L, Macadam A, Dobbins JG, Burns C, Wait D et al. Isolation and characterization of circulating type 1 vaccine-derived poliovirus from sewage and stream waters in Hispaniola. J Infect Dis. 2004 Apr 1; 189 (7): 1168-75. PubMed | Google Scholar
- Kargar M, Sadeghipour S, Nategh R. Environmental surveillance of Non-Polio Enteroviruses in Iran. Virol J. 2009 Sep25; 6: 149 149. PubMed | Google Scholar
- Roivainen M, Blomqvist S, Al-Hello H, Paananen A, Delpeyroux F, Kuusi M, Hovi T. Highly divergent neurovirulent vaccine-derived polioviruses of all three serotypes are recurrently detected in Finnish sewage. Euro Surveill. 2010 May 13; 15(19)2010: pii/19566.. PubMed | Google Scholar
- La Rosa G, Pourshaban M, Laconelli M, Muscillo M. Quantitative realtime PCR of enteric viruses in influent and effluent samples from wastewater treatment plants in Italy. Ann Ist Super Sanita. 2010; 46(3): 266-273. PubMed | Google Scholar
- Hewitt J, Leonard M, Greening GE, Lewis GD. Influence of wastewater treatment process and the population size on human virus profiles in wastewater. Water Res. 2011 Nov 15; 45 (18): 6267-76. PubMed | Google Scholar
- Hovi T, Cantell K, Huovilainen A, Kinnunen E, T Kuronen, Lapinleimu K, Poyry T, Roivainen M, Salama N, Stenvik M et al. Outbreak of paralytic poliomyelitis in Finland: widespread circulation of antigenically altered poliovirus type 3 in a vaccinated population. The Lancet. 1986 21 June; 327(8495): 1427-32. PubMed | Google Scholar
- Ranta J, Hovi T, Arjas E. Poliovirus surveillance by examining sewage water specimens: studies on detection probability using simulation models. Risk Anal. 2001 Dec;21(6): 1087-96. PubMed | Google Scholar
- Cernakova B, Sobotova Z, Rovny I, Blahova S, Roivainen M, Hovi T. Isolation of vaccine-derived polioviruses in the Slovak Republic. Eur J Clin Microbiol Infect Dis. 2005 Jun; 24(6): 438-9. PubMed | Google Scholar
- Blomqvist S, Savolainen C, Laine P, Hirttio P, Lamminsalo E, Penttila E et al. Characterization of a highly evolved vaccine-derived poliovirus type 3 isolated from sewage in Estonia. J Virol. 2004 May; 78(9): 4876-83. PubMed | Google Scholar
- Hovi T. The efficiency and reliability of polio surveillance. Dev Biol (Basel). 2001; 105: 21-31. PubMed | Google Scholar
- CDC. Evaluating Surveillance Indicators Supporting the Global Polio Eradication Initiative. MMWR Morb Mortal Wkly Rep. 2013 Apr 12; 62 (14): 270-4. PubMed | Google Scholar
- El Bassioni L, Barakat I, Nasr E, De Gourville EM, Hovi T, Blomqvist S, Burns C, Stenvik M, Gary H, Kew OM, Pallansch MA, Wahdan MH. Prolonged detection of indigenous wild poliovirus in sewage from communities in Egypt. Am J Epidemiol. 2003 Oct 15; 158(8): 807-15. PubMed | Google Scholar
- WHO- Poliovirus detected from environmental samples in Israel - update. Global Alert and Response (GAR) 15/08/2013) available at: http://www.who.int/csr/don/2013_06_03/fr/index.html Accessed on Jully 2014. PubMed | Google Scholar
- Theodore Tulchinsky H, Asad Ramlawi, Ziad Abdeen, Itamar Grotto, Antoine Flahault. Polio lessons 2013: Israel, the West Bank, and Gaza. The Lancet. 2013 Nov 16; 382(9905): 1611-2. PubMed | Google Scholar
- WHO/Global Alert and Response (GAR) June 2013. Poliovirus detected from environmental samples in Israel. http://www.who.int/csr/don/2013_06_03/en/index.html Accessed on July 2014. PubMed | Google Scholar
- Manor Y, Handsher R, Halmut T, Neuman M, Bobrov A, Rudich H, Vonsover A, Shulman L, Kew O, Mendelson E. Detection of poliovirus circulation by environmental surveillance in the absence of clinical cases in Israel and the Palestinian authority. J Clin Microbiol. 1999 Jun; 37(6): 1670-5. PubMed | Google Scholar
- Lewis GD, and TG Metcalf. Polyethylene glycol precipitation for recovery of pathogenic viruses, including hepatitis A virus and human rotavirus, from oyster, water, and sediment samples. Appl Environ Microbiol. 1988 Aug; 54(8): 1983-88. PubMed | Google Scholar
- Pipkin PA, Wood DJ, Racaniello VR, and Minor PD. Characterization of L cells expressing the human poliovirus receptor for the specific detection of polioviruses in vitro. J Virol Methods. 1993 Mar; 41(3): 333-40. PubMed | Google Scholar
- World Health Organization: Polio Laboratory Manual. http://www.who.int/vaccines/en/poliolab/WHOPolio-Manual-9.pdf] webcite Department of Vaccines and Biologicals 2004 Accessed on July 2014. PubMed | Google Scholar
- Introduction of Real Time RT-PCR into the Global Polio Laboratory Network. http://www.polioeradication.org/Research/PolioPipeline/No2Autumn2008/IntroductionofRealTimeRTPCR Accessed 20 Feb 2012. PubMed | Google Scholar
- Van Der Avoort HG, Hull BP, Hovi T, Pallansch MA, Kew OM, Crainic R, Wood DJ. Comparative study of five methods for intratypic differentiation of poliovirus. J Clin Microbiol. 1995 Oct ; 33(10): 2562-66. PubMed | Google Scholar
- Hovi T, Stenvik M, Partanen H, Kangas A. Poliovirus surveillance by examining sewage specimens; Quantitative recovery of virus after introduction into sewerage at remote upstream location.Epidemiol Infect. 2001 Aug; 127(1):101-6.. PubMed | Google Scholar
- Hovi T, Blomqvist S, Nasr E, Burns CC, Sarjakoski T, Ahmed N, Savolainen C, Roivainen M, Stenvik M, Laine P, Barakat I, Wahdan MH, Kamel FA, Asghar H, Pallansch MA, Kew OM, Gary HE Jr, DeGourville EM, El Bassioni L. Environmental surveillance of wild poliovirus circulation in Egypt--balancing between detection sensitivity and workload. J Virol Methods. 2005 Jun; 126(1-2): 127-34. PubMed | Google Scholar
- Momou KJ , Akoua-Koffi C, Akre DS, Adjogoua EV , Tie�oulou L , Dosso M. Detection of enteroviruses in urban wastewater in Yopougon, Abidjan. Pathol Biol (Paris). 2012 Jun; 60 (3): e21-6. PubMed | Google Scholar
- Hamed Khodaei S, Kargar M , Tabatabaei H, Sarijlou M, Nategh R. Environmental Surveillance of Polio and Non-Polio Enteroviruses in Sistan and Balouchestan Province Iranian.J Publ Health. 2008; 37(3): 127-33. PubMed | Google Scholar
- Mohammad Kargar, Sara Sadeghipour and Rakhshandeh Nategh. Environmental surveillance of non-polio enteroviruses in Iran. Virol J. 2009 Septembre 25; 6: 149. PubMed | Google Scholar
- Poyry T, Stenvik M, Hovi T. Viruses in sewage waters during and after a poliomyelitis outbreak and subsequent nationwide oral poliovirus vaccination campaign in Finland. Appl Environ Microbiol. 1988 Feb; 54(2): 371-4. PubMed | Google Scholar
- Aw T and Gin KY-H. Environmental surveillance and molecular characterization of human enteric viruses in tropical urban wastewaters. Journal of Applied Microbiology. 2010 Aug; 109(2): 716-30. PubMed | Google Scholar
- Renois F et al. Enterovirus 68 in Pediatric Patients Hospitalized for Acute Airway Diseases. J Clin Microbiol. 2013 Feb; 51(2): 640-3. PubMed | Google Scholar
- Imamura T, Fuji N, Suzuki A, Tamaki R, Saito M, Aniceto R, Galang H, Sombrero L, Lupisan S, Oshitani H. Enterovirus 68 among children with severe acute respiratory infection, the Philippines. Emerg Infect Dis. 2011 Aug; 17 (8): 1430-5. PubMed | Google Scholar
- L�v�que N, Huguet P, Norder H, Chomel JJ. Enteroviruses responsible for acute hemorrhagic conjunctivitis. Med Mal Infect (in French). April 2010; 40 (4): 212-8. PubMed | Google Scholar
- Shukla D, Kumar A, Srivastava S, Dhole TN. Molecular identification and phylogenetic study of coxsackievirus A24 variant isolated from an outbreak of acute hemorrhagic conjunctivitis in India in 2010. Arch Virol. 2013 Mar; 158(3): 679-84. PubMed | Google Scholar
- Aubry C, Gautret P, Nougairede A, Dussouil AS, Botelho-Nevers E, Zandotti C, De Lamballerie X, Brouqui P, Parola P. 2012 outbreak of acute haemorrhagic conjunctivitis in Indian Ocean Islands: identification of Coxsackievirus A24 in a returned traveler. Eurosurveillance. 2012 May; 17(22): pii=20185. PubMed | Google Scholar
- Khan A, Sharif S, Shaukat S, et al. An outbreak of acute hemorrhagic conjunctivitis (AHC) caused by coxsackievirus A24 variant in Pakistan. Virus Res. 2008 Oct; 137(1): 150-2. PubMed | Google Scholar
- Kesson AM, Choo CM, Troedson C, Thorley BR, Roberts JA. Echovirus 19 associated with a case of acute flaccid paralysis. J Paediatr Child Health. 2013 Mar; 49(3): E239-42. PubMed | Google Scholar
- Cristina Pedrosa, Maria Jo�o Lage, Daniel Virella. Congenital echovirus 21 infection causing fulminant hepatitis in a neonate. BMJ Case Reports. 2013 Apr 9; pii:bcr2012008394. PubMed | Google Scholar
- Stanway G, Joki-Korpela P, Hyypi� T. "Human parechoviruses--biology and clinical significance". Rev Med Virol. 2000 Jan-Feb; 10 (1): 57-69. PubMed | Google Scholar
- Joki-Korpela P, Hyypi� T. "Parechoviruses, a novel group of human picornaviruses". Ann Med. 2001 Oct; 33 (7): 466-71. PubMed | Google Scholar
- Kargar M, Sadeghipour S, Nategh R. Environmental surveillance of Non-Polio Enteroviruses in Iran. Virol J. 2009 Sep 25; 6: 149. PubMed | Google Scholar
- Bessaud M, Joffret M-L, Holmblat B, Razafindratsimandresy R, Delpeyroux F. Genetic Relationship between Cocirculating Human Enteroviruses Species C. PLoS ONE. 2011; 6(9): e24823. PubMed | Google Scholar