Iatrogenic colorectal Kaposi sarcoma complicating a refractory ulcerative colitis in a human immunodeficiency negative-virus patient
Lamine Hamzaoui, Houda Kilani, Mahdi Bouassida, Moufida Mahmoudi, Emna Chalbi, Karima Siai, Heykel Ezzine, Hassen Touinsi, Mohamed MĺSaddak Azzouz, Sadok Sassi
Corresponding author: Mahdi Bouassida, MD, Department of Surgery, Mohamed Tahar Maamouri Hospital, 8000 Mrazga, Nabeul, Tunisia 
Received: 18 Jun 2013 - Accepted: 23 Aug 2013 - Published: 29 Aug 2013
Domain: Public Health
Keywords: Kaposiĺs Sarcoma, ulcerative colitis, Human Herpes Virus-8, Infliximab, immunosuppression
©Lamine Hamzaoui et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Lamine Hamzaoui et al. Iatrogenic colorectal Kaposi sarcoma complicating a refractory ulcerative colitis in a human immunodeficiency negative-virus patient. Pan African Medical Journal. 2013;15:154. [doi: 10.11604/pamj.2013.15.154.2988]
Available online at: https://www.panafrican-med-journal.com//content/article/15/154/full
Original article 
Iatrogenic colorectal Kaposi sarcoma complicating a refractory ulcerative colitis in a human immunodeficiency negative-virus patient
Iatrogenic colorectal Kaposi sarcoma complicating a refractory ulcerative colitis in a human immunodeficiency negative-virus patient
Lamine Hamzaoui1, Houda Kilani2, Mahdi Bouassida3,&, Moufida Mahmoudi1, Emna Chalbi2, Karima Siai1, Heykel Ezzine1, Hassen Touinsi3, Mohamed MĺSaddak Azzouz1, Sadok Sassi1
1Gastroenterology department, Mohamed Tahar Maamouri Hospital, Nabeul, Tunisia, 2Histopathology department, Mohamed Tahar Maamouri Hospital, Nabeul, Tunisia, 3General surgery department, Mohamed Tahar Maamouri Hospital, Nabeul, Tunisia
&Corresponding author
Mahdi Bouassida, MD, Department of Surgery, Mohamed Tahar Maamouri Hospital,
8000 Mrazga, Nabeul, Tunisia
Kaposi sarcoma is a mesenchymal tumor associated to a human herpes virus-8. It often occurs in human immunodeficiency virus-positive subjects. Colorectal localization is rare. We report the case of a colorectal Kaposi sarcoma complicating a refractory ulcerative colitis treated with surgery after the failure of immunomodulator therapy in a human immunodeficiency virus-negative heterosexual man.
Kaposi's sarcoma (KS) is a mesenchymal tumor, arising predominantly in the skin but which can affect any organ system. It's associated to human herpes virus-8 (HHV8) [1, 2]. Patients with inflammatory bowel disease, in particular ulcerative colitis (UC), are often treated with immunosuppressive therapy and can develop colorectal KS [3]. We report the case of a human immunodeficiency negative-virus (HIV) man, with a severe refractory UC, who was treated with steroids, azathioprine and infliximab (IFX). Failure of medical treatment indicated surgery. Histological examination of the colon revealed KS.
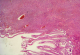
A 30-year-old heterosexual man, without familial and personal history, was diagnosed with rectosigmoid ulcerative colitis since 2010 revealed by chronic bloody diarrhea with abdominal pain and weight loss. He had a 3 pack-year history of smoking (stopped 3 years ago). He was initially treated with oral and local mesalamine with a good response. In 2011, he was treated 3 times with steroids (1mg/kg/day of prednisone) for a moderately active disease without a complete relief of diarrhea. In December 2011, at physical exam, his body mass index was 17kg/m▓. Abdominal exam was normal. There was no skin lesion. Iron deficiency anemia (11g/dL) and signs of inflammation: erythrocyte sedimentation rate (ESR) 37mm, c-reactive protein (CRP) 74mg/L were noted. Albumin level was 25g/L. Ileocolonoscopy showed large superficial ulcerations, spontaneous bleeding and pseudo-polyps. Lesions were mainly located in the rectosigmoid colon and the colitis was extended to the hepatic flexure. Histology found clear signs of active UC with no signs of malignancy. Oral corticosteroids were prescribed with a good initial response. At week 4 of corticosteroid therapy, we noted a relapse of bloody diarrhea, inflammation and aggravation of anemia (7,3g/dL). A left-sided colonoscopy didn?t show severe endoscopic signs. Copro-parasitological examinations were negative. Cytomegalovirus testing was negative too. Detection of Clostridium difficile toxins was not done. Intraveinous corticosteroids and parenteral nutrition were prescribed during 1week. At week 5 of corticosteroid therapy, another relapse occurred (7 to 8 stools daily, anemia 6g/dL, ESR 40mm, albumin 17g/L) and we had considered that it was a refractory severe UC. As the patient was reticent to surgery, medical treatment with immunomodulators was indicated. Assessement before immunosuppressive therapy was normal. He received blood transfusion, albumin infusion, parenteral nutrition and intravenous corticosteroids (with progressive decreasing doses). After improvement in health status, anemia (9g/dL) and albumin concentration (37g/L), azathioprine (2,5mg/kg/day, total of 3 months of treatment) and IFX (induction regimen with 5mg/kg, weeks 0, 2 and 6) were prescribed with unchanged disease activity. Drug failure led to a surgical treatment. A subtotal colectomy with double stomy of the ileum and of the sigmoid colon was performed. Colon macroscopic examination revealed multiple polypoid red lesions associated with a large ulcerations. Histologic examination of polypoid lesions showed a fusocellular spindle cells proliferation with a slit like vascular channels and extravased red blood cells consistent with the diagnosis of KS, associated with a typical features of UC (figure 1). Immunolabelling for HHV8 stained the nuclei of the spindle cells (figure 2,3). Involvement in 1 of the 25 perivisceral lymph nodes isolated was demonstrated. The patient underwent mucosal proctectomy and ileoanal anastomosis.
There are four clinical variants of KS with distinct clinical and epidemiological characteristics: classic, endemic, acquired immunodeficiency syndrome and iatrogenic [4]. Iatrogenic KS has been described in kidney and liver transplant recipients and also auto-immune diseases [5-6]. KS of gastrointestinal tract is rarely seen in the developed countries. Involvement by KS has been noted in 80% of patients with visceral disease in endemic areas [7]. Case studies have reported colorectal KS associated with inflammatory bowel disease in HIV-negative patients (since until now 11 observations). Most of cases were refractory UC on immunosuppressive therapy [8]. Our patient has received a triple immunosuppression (steroids, azathioprine and infliximab). It's the second case occurring into IFX. The diagnosis is difficult to establish in the absence of skin lesions, as in our patient. Nodular and polypoid lesions are observed in endoscopy and biopsies may fail to sample diagnostic tissue before tumor infiltration of the mucosa [8]. Intraluminal polyps are red or blue, due to high vascular and conjunctive tissue proliferation. Distinction from pseudo-polyps is difficult [9]. Seven months before diagnosis, the colonoscopy showed polypoid lesions, considered as inflammatory pseudo-polyps by the histologist. Immunohistochemistry of HHV-8 is helpful in the histological assessment of the lesions and may demonstrate infiltration of the mucosa by KS [8]. HHV-8 was positive in our case. Proctocolectomy associated to immunosuppressor withdrawal is usually effective to treat both UC and KS. After subtotal colectomy and confirmation of KS associated to UC, the patient will undergo proctectomy.
Although it is rare, it is important to consider a concomitant KS in patients with refractory severe ulcerative colitis, on immunosuppressor therapy, independently of HIV status.
There authors declare no competing interest.
All authors contributed in this work in ways that conform to ICMJE authorship criteria. All authors read and approved the final manuscript.
Figure 1: Histology (H and E stain x 40): Spindle cell proliferation with vascular channels infiltrating a colonic mucosa
Figure 2: HHV8 immunohistochemical stain: strong nuclear positivity in spindle tumor cells x200
Figure 3: Fig CD31 immunohistochemical stain: positivity in vascular channels x40
- Chang Y, Cesarman E, Pessin MS et al. Identification of herpesvirus-like DNA sequences in AIDSassociated Kasposi's sarcoma. Science. 1994; 266(5192): 1865-9. PubMed | Google Scholar
- Antman K, Chang Y. Kaposi's sarcoma. N Engl J Med. 2000; 342(14): 1027-38. PubMed | Google Scholar
- Meltzer SJ, Rotterdam HZ, Korelitz BI. Kaposi's sarcoma in association with ulcerative colitis. Am J Gastroenterol. 1987; 82(4): 378-81. PubMed | Google Scholar
- Martin RW III, Hood AF, Farmer ER. Kaposi sarcoma. Medicine. 1993; 72(4): 245-61. PubMed | Google Scholar
- GarcÝa- Astudillo LA, Leyva-Cobißn F. Human herpesvirus-8 infection and Kaposi┤s sarcoma after liver and kidney transplantation in different geographical areas of Spain. Transpl Immunol. 2006; 17(1): 86-9. PubMed | Google Scholar
- Klein MB, Pereira FA, Kantor I. Kaposi sarcoma complicating systemic lupus erythematosus treated with immunosuppression. Arch Dermatol. 1974; 110(4): 602-4. PubMed | Google Scholar
- Taylor FJ, Templeton AC, Vogel CL et al. Kaposi's sarcoma in Uganda: a clinicopathological study. Int J Cancer. 1971; 8(1): 122-35. PubMed | Google Scholar
- Girelli CM, Serio G, Rocca E, Rocca F. Refractory ulcerative colitis and iatrogenic colorectal Kaposi's sarcoma. Dig Liver Dis. 2009; 41(2): 170-4. PubMed | Google Scholar
- Tavassolie H, Mir-Madjlessi SH, Sadr-Ameli MA. The endoscopic demonstration of Kaposi's sarcoma of the colon. Gastrointest Endosc. 1983; 29(4): 331-332. PubMed | Google Scholar