Setting the stage for randomized controlled trials in Cameroon: a workshop report
Lawrence Mbuagbaw, Lehana Thabane, Pierre Ongolo-Zogo
Corresponding author: Lawrence Mbuagbaw, Department of Clinical Epidemiology and Biostatistics, McMaster University, Hamilton, ON, Canada 
Received: 23 Jul 2013 - Accepted: 06 Aug 2013 - Published: 21 Aug 2013
Domain: Epidemiology
Keywords: Clinical trials, capacity building, Cameroon, workshop
©Lawrence Mbuagbaw et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Lawrence Mbuagbaw et al. Setting the stage for randomized controlled trials in Cameroon: a workshop report. Pan African Medical Journal. 2013;15:144. [doi: 10.11604/pamj.2013.15.144.3134]
Available online at: https://www.panafrican-med-journal.com//content/article/15/144/full
Setting the stage for randomized controlled trials in Cameroon: a workshop report
Lawrence Mbuagbaw1,2,3,&, Lehana Thabane1,2,4,5,6, Pierre Ongolo-Zogo3,7
1Department of Clinical Epidemiology and Biostatistics, McMaster University, Hamilton, ON, Canada, 2Biostatistics Unit, Father Sean O�Sullivan Research Centre, St Joseph�s Healthcare - Hamilton, ON, Canada, 3Centre for Development of Best Practices in Health, Yaounde Central Hospital, Yaounde, Cameroon, 4Departments of Paediatrics and Anaesthesia, McMaster University, Hamilton, ON, Canada, 5Centre for Evaluation of Medicine, St Joseph�s Healthcare - Hamilton, ON, Canada, 6Population Health Research Institute, Hamilton Health Sciences, Hamilton, ON, Canada, 7Faculty of Medicine and Biomedical Sciences, University of Yaounde 1, Yaound�, Cameroon
&Corresponding author
Lawrence Mbuagbaw, Department of Clinical Epidemiology and Biostatistics, McMaster University, Hamilton, ON, Canada
In order to improve the generation and use of high quality health research evidence among Cameroonian researchers, the Centre for Development of Best Practices in Health (CDBPH) located in the Yaounde Central Hospital organised a three day workshop on clinical trials from the 29th April to the 1st May 2013. Sixteen participants form the Faculty of Medicine and Biomedical Science of the University of Yaounde 1 and the Ministry of Health attended this workshop. This report includes the material covered in the workshop, the readings and supplementary resources for clinical trials. The workshop was well received by the participants and resulted in significant gains in knowledge on clinical trials.
Randomized controlled trials (RCTs) are often regarded as the most rigorous method available for hypothesis testing about the effects of therapies, interventions or treatments in epidemiology and medicine [1]. In its simplest form, participants are randomly allocated to one of two interventions and followed up for a predefined duration over which to measure specific outcomes [1]. Since randomized controlled trials are experimental studies involving human subjects, specific design, ethical, administrative and organisational issues must be understood by health researchers. The Centre for Development of Best Practices in Health (CDBPH; www.cdbph.org) is a knowledge translation institution located in the Yaounde Central Hospital, Yaounde, Cameroon, that supports decision-makers, members of civil society organisations, journalist and health researchers in the collection, use and generation of health research evidence.
�
In December 2010, the CDBPH organised a workshop on systematic reviews which involved appraising RCTs [2]. Systematic reviews of RCTs (and other studies) inform evidence-based health care and are used to develop health care practice and policies. During this workshop, we noted significant limitations in understanding the concepts related to RCTs. For this reason, we organised another workshop introducing Cameroonian medical research scientists to clinical trials. The number of clinical trials conducted in Cameroon is relatively low, with only 34 registered trials on the www.clinicaltrials.gov website (search conducted 22nd June 2013), compared to other countries like Ghana (81 trials) or Malawi (99 trials) with similar population sizes [3]. This workshop responds to the urgent needs for health research capacity building in developing countries to promote evidence based health care [4, 5]. This lack of research capacity is often seen in the considerably small number of African-led trials within the continent [6].�
The purpose of this report is to provide a detailed account of the activities and the material covered during the workshop, such that it can be replicated and adapted to suit other trainers� needs.
Location
�
The CDBPH arranged and supported the workshop, which took place from the 29th April to the 1st May 2013, at the Laurence Vergne Room of the Yaounde Central Hospital, Yaounde, Cameroon.�
Aims�
The aims of this workshop were to build clinical trials skills in Cameroon by enabling participants to 1) distinguish clinical trials from other forms of clinical research, 2) describe the different phases of clinical trials, 3) list the steps involved in conducting a clinical trial, 4) understand key concepts in design, analysis and reporting of clinical trials, 5) understand the limitations of clinical trials and 6) describe variants in clinical trial design. These concepts were introduced at a basic level and followed-up in-depth according to participant interest.�
Participants�
The CDBPH invited lecturers from the Faculty of Medicine and Biomedical Sciences, of the University of Yaounde 1 and staff from the Division of Health Operations Research of the Ministry of Health.�
Facilitators�
The facilitators where chosen based on their methods expertise and experience with clinical trials in resource limited settings: a Canadian professor of biostatistics and epidemiology, originally from Lesotho, with extensive experience conducting international trials; a Cameroonian professor of radiology and epidemiology, with vast experience in evidence-based policy development and a Cameroonian doctor and epidemiologist with a strong background and experience in heath research methodology and clinical trials.�
Pre-workshop tasks�
Prior to the workshop, participants were expected to 1) complete one introductory reading on clinical trials [7], 2) prepare one research question for which a randomized clinical trial would be appropariate and 3) find one randomized clinical trial in their respective fields for appraisal.�
Program�
Over three days participants were introduced to the role of clinical trials in clinical research and how to set-up and run a clinical trial in Cameroon, using practical examples. Their clinical trial topics were discussed and revised according to standard recommendations for question formulation[8]. Table 1 is a list of trial topics proposed by the participants. The sessions were interactive, with engaging discussions between the staff from the Ministry of Health and individual researchers. Participants were also expected to take a pre- and post-workshop multiple choice questionnaire to evaluate their clinical trial knowledge. The questionnaire was tailored to assess if the objectives of the workshop were met.�
During the workshop, participants described the challenges they faced in conducting clinical research in Cameroon. As a compliment to the new material provided to participants during the workshop, discussions were held upon the difficulties they encountered in their own clinical research and solutions were proposed. For example, issues related to high drop-out rates were addressed during the session on attrition. The following challenges were identified:�
- Lengthy and complex consent forms
- Lengthy ethics approval procedures
- Difficulties in securing funding
- Poor access to high quality medical archives
- High drop-out rates
- Stigma preventing participants from taking part in research
- Difficulties in obtaining adequate comparators (placebos)
- Limited statistical competency
- Limited access to information regarding the regulation of research in Cameroon
�
Course material and readings�
The participants were provided with reading material relevant to each topic addressed. Table 2 is a summary of the topics covered in the workshop, the readings and other electronic resources. We sought to make the training relevant to the country by using some local readings and examples as often as possible.�
Evaluation�
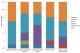
The results of the questionnaire were evaluated and compared. The questionnaire covered all topics discussed during the workshop. A total of 35 points could be obtained for responding correctly to 13 questions. The mean score (standard deviation) before the workshop was 16.5 (6.93) compared to 25.5 (5.05) after the workshop. This difference (+9) was statistically significant (t(30) =-4.2; p<0.001), showing a marked improvement in knowledge on clinical trials. Participants were also asked to rank the workshop in the following domains: quality of the lectures, quality of the practical exercises, quality of the reading material, pace of the course and the amount of subject material covered. Overall, the workshop was well received (Figure 1).�
Participants appreciated the interactive nature of the workshop; the practical exercises and the quality and timeliness of the presentations. They also noted some points for improvement such as: the need to develop a concrete trial proposal as part of the workshop, to reduce the amount of material covered and to allocate more time for practical sessions. In addition, they recommended a follow-up workshop with more French material.
The workshop was well received and resulted in significant knowledge gains amongst participants. The next steps will involve continuous support to all participants who wish to conduct RCTs and accrued efforts to deliver locally relevant material in terms of context and language.
This workshop was supported by the Effective Health Care Research Consortium which is funded by UKaid from the Department for International Development. It was organised by the Centre for Development for Best Practices in Health/Yaounde Central Hospital. We also wish to thank Professor Thabane from McMaster University for his availability during the workshop.
Table
1: Topics proposed by participants of clinical trials workshop
Table
2: Workshop outline
Figure 1: Overall
assessment of clinical trials workshop by participants (n=12)
- Porta M, editor. A dictionary of epidemiology. 5th ed. Oxford: Oxford University Press, Inc.; 2008.
- Mbuagbaw L, Wiysonge CS, Nsagha DS, Ongolo-Zogo P, Pantoja T. An introduction to systematic reviews and meta-analysis: a workshop report on promoting evidence based medical practice through capacity building in research synthesis. Pan Afr Med J. 2011;8:15. PubMed | Google Scholar
- USNIH. Clinicaltrials.gov 201 Available from: http://clinicaltrials.gov/ct2/search/browse?brwse=locn_cat_AF. Accessed 23 July 2013.
- Nchinda TC. Research capacity strengthening in the South. Soc Sci Med. 2002 Jun;54(11):1699-711. PubMed | Google Scholar
- Laabes EP, Desai R, Zawedde SM, Glew RH. How much longer will Africa have to depend on western nations for support of its capacity-building efforts for biomedical research?. Trop Med Int Health. 2011 Mar;16(3):258-62. PubMed | Google Scholar
- Andersson N. African Development of AIDS Prevention Trials capacities. South African Development Community HIV Prevention and Research Meeting; 9-10 October 2012; Johannesburg, South Africa. Google Scholar
- Smith A, Palmer S, Johnson DW, Navaneethan S, Valentini M, Strippoli GF. How to conduct a randomized trial. Nephrology (Carlton). 2010 Dec;15(8):740-6. PubMed | Google Scholar
- Thabane L, Thomas T, Ye C, Paul J. Posing the research question: not so simple. Can J Anaesth. 2009 Jan;56(1):71-9. PubMed | Google Scholar
- Goffin J, Gad SC. Introduction to Clinical Trials. Pharmaceutical Sciences Encyclopedia: John Wiley & Sons, Inc.; 2010.
- Kimmelman J. The social function of clinical equipoise. Clinical Trials. 2012 October 1, 2012;9(5):630-1. PubMed | Google Scholar
- Thabane L, Ma J, Chu R, Cheng J, Ismaila A, Rios LP, et al. A tutorial on pilot studies: the what, why and how. BMC Med Res Methodol. 2010;10:1. PubMed | Google Scholar
- Schulz KF, Grimes DA. Sample size calculations in randomised trials: mandatory and mystical. Lancet. 2005 Apr 9-15;365(9467):1348-53. PubMed | Google Scholar
- Downs M, Tucker K, Christ-Schmidt H, Wittes J. Some practical problems in implementing randomization. Clin Trials. 2010 Jun;7(3):235-45. PubMed | Google Scholar
- Schulz KF, Grimes DA. Generation of allocation sequences in randomised trials: chance, not choice. Lancet. 2002 Feb 9;359(9305):515-9. PubMed | Google Scholar
- Schulz KF, Grimes DA. Allocation concealment in randomised trials: defending against deciphering. Lancet. 2002 Feb 16;359(9306):614-8. PubMed | Google Scholar
- Schulz KF, Grimes DA. Blinding in randomised trials: hiding who got what. Lancet. 2002 Feb 23;359(9307):696-700. PubMed | Google Scholar
- Sackett DL. Commentary: Measuring the success of blinding in RCTs: don't, must, can't or needn't?. Int J Epidemiol. 2007 Jun;36(3):664-5. PubMed | Google Scholar
- J�ni P, Egger M. Commentary: Empirical evidence of attrition bias in clinical trials. International Journal of Epidemiology. 2005 February 1, 2005;34(1):87-8. PubMed | Google Scholar
- Dumville JC, Torgerson DJ, Hewitt CE. Reporting attrition in randomised controlled trials. BMJ. 2006 Apr 22;332(7547):969-71. PubMed | Google Scholar
- Schulz KF, Altman DG, Moher D. CONSORT 2010 statement: updated guidelines for reporting parallel group randomized trials. Ann Intern Med. 2010 Jun 1;152(11):726-32. PubMed | Google Scholar
- Mbuagbaw L, Thabane L, Ongolo-Zogo P, Lang T. The challenges and opportunities of conducting a clinical trial in a low resource setting: the case of the Cameroon mobile phone SMS (CAMPS) trial, an investigator initiated trial. Trials. 2011;12:145. PubMed | Google Scholar
- Bush T. Beyond HERS: some (not so) random thoughts on randomized clinical trials. Int J Fertil Womens Med. 2001 Mar-Apr;46(2):55-9. PubMed | Google Scholar
- Selby P, Brosky G, Oh PI, Raymond V, Ranger S. How pragmatic or explanatory is the randomized, controlled trial?. The application and enhancement of the PRECIS tool to the evaluation of a smoking cessation trial. BMC Med Res Methodol. 2012;12:101. PubMed | Google Scholar
- Treweek S, Zwarenstein M. Making trials matter: pragmatic and explanatory trials and the problem of applicability. Trials. 2009;10:37. PubMed | Google Scholar
- Knatterud GL. Management and conduct of randomized controlled trials. Epidemiol Rev. 2002;24(1):12-25. PubMed | Google Scholar