Effects of concurrent chloroquine and ethanol administration on the rat kidney morphology
Abdurrahman Abdulkadir, Ejikeme Felix Mbajiorgu, Trust Nyirenda
Corresponding author: Abdurrahman Abdulkadir, Department of Anatomy, College of Health Sciences, Federal University Birnin Kebbi, Kebbi State, Nigeria and Division of Histology and Embryology, School of Anatomical Sciences, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, Gauteng, South Africa 
Received: 10 Apr 2017 - Accepted: 19 Dec 2017 - Published: 18 Jan 2018
Domain: Anatomy,Cell biology,Pharmacology
Keywords: Chloroquine, ethanol, computed tomography, stereology, relative medullary thickness
©Abdurrahman Abdulkadir et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Abdurrahman Abdulkadir et al. Effects of concurrent chloroquine and ethanol administration on the rat kidney morphology. Pan African Medical Journal. 2018;29:49. [doi: 10.11604/pamj.2018.29.49.12471]
Available online at: https://www.panafrican-med-journal.com//content/article/29/49/full
Original article 
Effects of concurrent chloroquine and ethanol administration on the rat kidney morphology
Effects of concurrent chloroquine and ethanol administration on the rat kidney morphology
Abdurrahman Abdulkadir1,&, Ejikeme Felix Mbajiorgu2, Trust Nyirenda3
1Department of Anatomy, College of Health sciences, Federal University Birnin Kebbi, Kebbi State, Nigeria and Division of Histology and Embryology, School of Anatomical Sciences, Faculty of Health Sciences, University of The Witwatersrand, Johannesburg, Gauteng, South Africa, 2Division of Histology and Embryology, School of Anatomical Sciences, Faculty of Health Sciences, University of The Witwatersrand, Johannesburg, Gauteng, South Africa, 3Department of Anatomy and Physiology, Faculty of Medicine, National University of Science and Technology, Bulawayo, Zimbabwe and Division of Histology and Embryology, School of Anatomical Sciences, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, Gauteng, South Africa
&Corresponding author
Abdurrahman Abdulkadir, Department of Anatomy, College of Health Sciences, Federal University Birnin Kebbi, Kebbi State, Nigeria and Division of Histology and Embryology, School of Anatomical Sciences, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, Gauteng, South Africa
Introduction: the use of antimalarial chloroquine in malaria-endemic regions of Africa is rampant and it is not uncommon to find individuals taken the drug concurrent with alcohol. Effects of anti-malarial drug chloroquine (Chq) and ethanol (Et) combination on kidney volume and function using rat model was investigated.
Methods: 32 adult male rats were randomly distributed into four groups of 8 rats each. Group C serve as control and received vehicle only, while Q is Chq treated only, E is Et treated and QE is Et and Chq treated. Chq was administered intraperitoneally at 1mg/100g body weight weekly and 6% Et in drinking water provided ad libitum. Urine volume was collected before the treatment began and after the treatment. After eight weeks, all animals were euthanized; kidneys were harvested and fixed in 10% neutral formalin. The fixed left kidneys were scanned with computed tomography and the scan slices were used to estimate 3-dimensional kidney volume on ImageJ.
Results: total kidney volume was none significantly increased in Q, E and QE treated compared to control groups (p = 0.5150). Also, microscopic analysis showed increased proximal tubule diameter (p = 0.1426) and epithelial hypertrophy (p = 0.2530) and significant urinary space shrinkage (p = 0.00001). The initial urine volume was not significantly different between the control and treated groups (p = 0.9864) however, following treatment urine volume was significantly reduced in QE rats group (p = 0.0029).
Conclusion: the results suggest chloroquine and ethanol combination as potential cause of kidney injury through structural damage and function derangement.
Chronic drug administration can lead to cumulative drug toxicity to body organs especially liver and kidney. Chronic administration of chloroquine causes derangement of kidney function and histological abnormalities [1, 2]. Furthermore, concurrent chloroquine and alcohol ingestion is not uncommon in Africa, with the current increase in alcohol intake. It was reported by Musabayane et al [3] that chronic concurrent alcohol and chloroquine administration causes significant derangement in kidneys electrolyte handling and also observed dilatation of the urinary space and extensive damages to the proximal tubule and collecting duct on light microscopy. This evidence might suggest increased chloroquine toxicity to the kidney when the drug interacts with alcohol and a thus create the need to have more informed ways of monitoring the toxicity. Chloroquine is a synthetic antimalarial drug developed as a response to the quest for a drug to combat the scourge of malaria around the 1940s [4]. It was, however, abandoned two decades later due to resistant malaria [5] which significantly reduce the drug efficacy on malaria. Other reasons of chloroquine use are its application in treatment of some inflammatory conditions such as rheumatoid arthritis and systemic lupus erythematosus [6]. Furthermore, chloroquine is currently being investigated for application in viral therapy and cancer chemotherapy [7, 8]. These recent therapeutic applications of chloroquine require a long-term administration usually in combination therapy and toxicity is inevitable. Even so, the majority of the poor people in sub-Saharan Africa still resort to using chloroquine for any febrile episode considered to be malaria attack [9]. The situation created a repeated cycle of chronic chloroquine intake for malaria treatment. Also, the resurgence of chloroquine-sensitive malaria in some African countries [10, 11] makes a comeback for chloroquine use in malaria treatment.
It is facetious to not acknowledge the increase in alcohol ingestion in Africa [12] and perhaps concurrent intake of the beverage with any prescribed or over-the-counter drug with no exception to chloroquine. Both ethanol and chloroquine are metabolised by the liver cytochrome P450 enzymes and ethanol is notorious for competing with drugs for these enzymes and thereby delaying the drug metabolism and elimination [13]. This tends to increase the drug half-life and might lead to more toxicity of the drug to the body. Methods used in assessing kidney health status include transabdominal ultrasound used to check the state of kidney function and total kidney volume for hypertrophy or shrinkage [14, 15]. In addition to ultrasound, other methods of quantifying total kidney volume are the use of contrast-enhanced computed tomography (CT) [16] and also the stereological technique [17]. Stereology involved the exhaustive sectioning of the kidney and then selecting representative sections using systematic uniform random sampling, then using Cavalieri principle and point counting to estimate the area of each section. Thus, the product of sections area and section thickness provides the volume of the sample fraction. The volume of sample fraction is multiplied by the number of sections in each fraction to get the total volume. Urine concentrating ability of the kidney is also a marker of kidney function used [18]. Relative medullary thickness (RMT) was reported to have a significant direct relationship with the maximum urine concentrating ability of animals. This relationship has been reported between animal species [19] and also in a post-nephrectomy experiment on a goat [14]. RMT is by measuring the length of the medulla from the corticomedullary junction to the tip of papilla referred to as the medullary thickness (MT) relative to the kidney size (KS) using the Sperber formula [18].
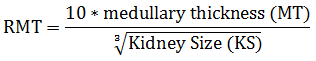
Animals and housing: all animals experiment and procedures were approved by the Animal Ethics Screening Committee (AESC) of the University of the Witwatersrand (AESC 2015/11/54C). A total of 32 adult male Sprague Dawley rats average weight 405 � 24g were bred and housed at the Central Animal Services of the University of the Witwatersrand, Johannesburg. The rats were individually housed in Perspex cages lined with saw dust and paper shreds with the temperature maintained at 21 � 1�C and a 12 hour light cycle, cages were changed twice a week. Standard rat food was provided ad libitum to all the rats.
�
Study design: the rats were acclimatised to the cage and handling for five days and then randomly distributed into four groups of 8 rats each. A control group (C) and three experimental groups, the experiment include a group with chloroquine treatment only (Q), a group with ethanol treatment only (E) and a group with both chloroquine and ethanol treatments (QE). All treatments lasted for eight weeks. Chloroquine phosphate salt (Sigma Aldrich, PHR1258) was purchased commercially and was reconstituted in our laboratory with 0.9% saline into 10mg/ml solution; fresh solution was made for each week. Chloroquine was administered weekly for eight weeks based on the weekly weight as an intraperitoneal injection at the dose of 0.1ml/100g (equivalent of 1mg/100g) body weight to the treatment group Q and QE, while C and E groups received 0.1ml/100g body weight of 0.9% saline. Absolute alcohol was acquired from our general lab which was then diluted with distilled water to our desired concentration of 6%, this concentration is approximate to the concentration of ethanol in commercial beer. Ethanol was administered ad libitum in distilled water at a concentration of 6% ethanol in distilled water (V/V) to the experimental groups E and QE, while groups C and Q received plain distilled water ad libitum for consecutive eight weeks of the experiment. At the end of the eight weeks treatment, all rats terminal weight were taken and then were euthanised with an overdose of sodium pentobarbitone (Euthanaze�) 20mg/100g body weight intraperitoneal injection. Blood samples were collected using 5 ml syringes with 21G needles by cardiac puncture into plain blood sample tubes to collect serum. Kidneys were excised through midline abdominal incisions and were fixed in 10% neutral buffered formalin for 10-14 days until further analysis. The right kidney was separated from the left at the point of collection for consistency of analyses. Both kidneys were weighted using RADWAG sensitive balance (RADWAG Wagi Elektroniczne, Poland) prior to fixation.�
Urine collection and volume estimation: urine volume was collected over 24 hours before the injections and after the 8 weeks of treatment. Rats were transferred in metabolic cages individually for 24 hours and then all the urine produced was collected and then the rats were returned to the normal cages. Urine volume was determined using measuring cylinder.�
Relative medullary thickness measurements: the right kidney was used for the RMT measurements; the first step was gross length, breadth and width measurements with a dial vernier calliper (Mauser Inox 0-150mm, 0.05mm). The second step is the medullary thickness (MT) measurement; this was done by dissecting the right kidney into two mid-sagittal halves. One of the halves was placed on a dissecting microscope 5x total magnification (Nikon SMZ1500, Japan) fitted with a digital camera (Nikon DSfi1) and capture screen (Nikon Digital sight) and digital images were acquired. On these images the cortex and medulla were clear in fixed kidney and a metric transparent ruler was introduced into the images for standardisation of the scale. The capture screen provides a text option to properly put a label to the images. The images were then imported into ImageJ image analysis software. On ImageJ scale was set using the metric ruler in the images and was standardised by comparing the length of the kidney measured with a vernier calliper to the length measured with the ImageJ software and it was 0.5mm accurate and was acceptable. The medullary thickness was then measured using the ImageJ software [21] from the corticomedullary junction to the tip of the papilla, together with the cortical thickness, and the results were exported from the ImageJ into Microsoft Excel 2010 for further analyses.�
Stereological volume estimation: the 10% neutral formalin fixed left kidney was scan with a microfocus computed tomography machine (Nikon X TH 225/320 LC) at a 50KV and 100�A. The kidney was stabilised with Styrofoam above and below in a transparent container which is then exposed to the X-ray source for scanning. The scanned images were then reconstructed to a three-dimensional VGI image file with CT 3D Pro XT 2.2 software which can now be analysed with VGStudio Max 3.0 (Volume Graphics GmbH, Germany). The VGStudio provide serial sections images of the kidney which met the criteria for unbiased stereological sectioning method. Serial sections were obtained using the VGStudio software and saved as image files; the sections exhaust the whole kidney at a 10�m interval. So, depending on the kidney size thousands of sections were generated, a sample fraction was calculated and then 10 sections were sampled based on a pilot using the systematic uniform random sampling (Figure 1). Significant coefficient of error (CE) was provided by the 10 sections that were chosen. The images were analysed with Volumest plugin [22] on ImageJ [21] and the volume (mm3) and the CE were obtained for the 10 sections. Total kidney volume is the product of the volume from the 10 sections multiplied by the sample fraction. The procedure was repeated twice for each kidney and an average of the results was taken.�
Microscopic morphometry: the left kidney was sliced into 3 equal parts and middle block was processed for routine histology. 5�m sections were cut on a rotary microtome and were stained with haematoxylin and eosin (H&E). Photomicrographs of the H&E stained sections were taken with Axioskop 2 microscope fitted with a camera Axiocam HRc2 at 63x magnification. Measurements were taken on the photomicrographs using the Zen lite analysis software. In each of the parameter for each of the treatment group and control group 60 measurements were done.�
Serum analyses: the blood collected in the tubes was centrifuged with Rotofix 32A (Hettich Zentrifugen, Germany) centrifuging machine at 1067 x g for 10 minutes. Serum was then collected as the supernatant into the 1ml Eppendorf tubes and stored in -80�C deep freezer until further analyses. Serum urea was analysed with Reflotron analyser (Boehringer Mannheim, Germany) and the creatinine was sent to Idexx Laboratories, Johannesburg, for analysis.�
Statistical analysis: all data were managed and graphs were constructed in Microsoft Excel 2010 and the statistical analyses were done with STATA 13.1, results are presented as Mean � SEM. The p-value of 0.05 is considered significant. Data was tested for normality using Shapiro-wilk test, normally distributed data is analysed with one-way analysis of variance (ANOVA) while Kruskal-wallis was used for non-parametric data. Bonferroni post-hoc was used for multiple comparisons in significant One-way ANOVA and Dunn's multiple comparisons for significant Kruskal-wallis test.
General findings: at the end of the eight week treatment, the control group gained more weight than the treatment groups without statistical significance (One-way ANOVA, p = 0.3063). Also, the food consumption was more in the control group than in the treatment group (Kruskal-wallis, p = 0.4190). The kidney weight was however increased by 2%, 5% and 4% in Q, E and QE groups respectively compared to the control group (One-way ANOVA, p = 0.7957). While the kidney weight relative to body weight was decreased by 3% and 4% in the Q and E groups respectively compared to the control group, it was increased by 1% in the QE group compared to the control group (One-way ANOVA, p = 0.3865) (Table 1).
�
24 hour urine volume: urine volume was not significantly different in both the control and treatment groups before the commencement of the experiment (One-way ANOVA, p = 0.9864), however, there was significant reduction of the urine volume (Dunns test, p = 0.0029) in QE group compared to the control group with non-significant increase in the urine volume in Q and E compared to the control group (Figure 2).�
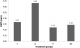
Relative medullary thickness: the relative medullary thickness is increased in Q compared to control (Figure 3). It was, however, significantly decreased in E and QE compared to the control (One-way ANOVA, p = 0.0001). Cortical thickness reduced in the treatment groups compared to the control group (Table 2).�
Total kidney volume using stereology: the total kidney volume was increased by 8%, 4% and 7% in Q, E and QE compared to the control group with no statistically significant differences (One-way ANOVA, p = 0.6533). The coefficient of error was 0.018 which is highly significant (Figure 4).�
Microscopic morphometry: there was significant shrinkage of the urinary space in the treatment groups compared to the control group (p = 0.00001), with non-significant decrease in the glomerular diameter (p = 0.5096) and the Bowman's capsule (p = 0.3212) (Table 3). The diameter and epithelial cells height were increased in the proximal tubules of all the treatment groups compared to the control group (p = 0.1426, p = 0.2530 respectively for diameter and epithelial cells height) (Table 4, Table 5). The distal tubules and the collecting ducts diameters were decreased in the treatment groups compared to the control group except in the E group which showed increased diameter of the distal tubule and collecting duct (p = 0.0925, p = 0.5537 respectively) (Table 4). The epithelial cells height were surprisingly increased despite the reduced diameter of collecting ducts of the treatment groups compared to the control (p = 0.0925), whereas there was decreased epithelial cells height of the distal tubule in treatment groups compared to the control except in the Q group (p = 0.5537) (Table 5).�
Biochemical analyses: the serum urea level was slightly increased in Q compared to control group, however, it was decreased in E and QE (One-way ANOVA, p = 0.9734) (Figure 5, A). Creatinine was increased in all the treatment groups compared to the control (One-way ANOVA, p = 0.1948) (Figure 5, B).
The results of this study suggested that chloroquine and ethanol concurrent administration causes loss of appetite evidenced by the decrease in food consumption and reduced weight gain in the treatment groups but not in the control rats. There was also increased kidney weight which is not unrelated to the increase in total volume of the organ observed in the treatment groups but not in the control rats. The increase organ weight might be due to the inflammatory response to the organ exposure to chloroquine and ethanol treatments, because chloroquine causes glomerular and tubular cell vacuolation [23], while ethanol promotes interstitial oedema and renal hypertrophy [24]. We observed renal hypertrophy in the Q, E and QE groups compared to the control group, this we believe to be the result of the proximal tubular cells hypertrophy. Alcohol is known to cause renal hypertrophy. Also, chloroquine and ethanol compromises the urine concentrating ability of the kidney as seen by significant reduction of the RMT in the treated group compared to the control. However, there was a non-significant increase in the Q group which indicates an increase in urine concentrating mechanism with chloroquine alone treatment. Ethanol was reported to cause a decrease in urine concentrating ability which conforms to our finding [25] but no such report on chloroquine alone. Some researchers [19] measure the urine osmolality in relation to RMT we, however, instead use the serum urea level which has a significant role [20] in the urine concentrating mechanism. The serum urea is seen to decrease in a similar pattern with the RMT itself, suggesting a decrease in urea level reduces the urine concentrating mechanism. Evidence from this result suggests that decrease in the urea level has a direct relationship with the RMT whereas the urine osmolality was reported to have inverse relationship. Creatinine clearance is a more clinical indicator of glomerular filtration capacity, the increase in serum creatinine level showed reduced creatinine clearance and a compromise of the glomerular filtration function. Our finding showed increased creatinine in all the treatment groups compared to the control, the increase creatinine level had an inverse relationship with the cortical thickness.
�
In a report by Yamashita et al [15] it was revealed that cortical thickness in a strong indicator of the renal function, as the cortical thickness tapered the function will be compromised. Significant reduction of the urine volume caused by the concurrent administration of chloroquine and ethanol is an indication of glomerular filtration compromise. Ethanol is known to increase urine volume by inhibiting the action of antidiuretic hormone (ADH) in the kidney or suppressing the blood levels of the hormone [24]. While, chloroquine is known to increase plasma level of ADH [26], the combination of chloroquine and ethanol significantly reduces urine volume suggest an intense chloroquine effect on the blood ADH levels. The general knowledge that chloroquine and ethanol both compete for the cytochrome P450 enzymes convince us that significant amount of chloroquine is maintained in the blood for a longer period when administered concurrently with ethanol. While most of the chloroquine is being metabolised by cytochrome P450 only, ethanol is metabolised by other enzymes as well [13]. With this knowledge, we believe more chloroquine will be available in the blood than alcohol, with only small amount of ethanol remaining to compete for the cytochrome enzymes. The results of concurrent chloroquine and ethanol administration showed that there is an increase in kidney weight and renal hypertrophy with compromised renal function indicated by the decrease in renal maximum urine concentrating ability and an increase in the serum creatinine level. This result provides evidence that there were actual structural changes in the kidney that altered the function and sodium water balance as reported by Musabayane et al [3]. The stereological technique used in estimation of the kidney volume is a non-bias morphometry which in itself eliminate the human bias, but also in addition to the stereology the application of microfocus computed tomography (�CT) eliminated the shrinkage factor occurring from tissue processing and also provide a more accurate sampling strategy. This method will provide more chances of using the tissue for other histological studies because after the �CT scanning the tissues were still intact.
Evidence from this study suggests renal morphological changes in relation to the compromise of the function seen with concurrent administration of ethanol and chloroquine in a rat.
What is known about this topic
- Renal fluid excretion in rats following ethanol and chloroquine administration;
- Electrolytes handling in rats following ethanol and chloroquine administration;
- Effects of ethanol and chloroquine concurrent administration on antidiuretic hormone.
What this study adds
- Quantitative microscopic morphometry on the effects of concurrent chloroquine and ethanol on rat kidneys;
- Gross morphometry of relative medullary thickness in rat kidney;
- Application of stereology to assess kidney volume changes.
The authors declare no competing interests.
Abdurrahman Abdulkadir, Contributes to conception of the idea, laboratory work and data collection, statistical analysis, preparation of the manuscript Ejikeme Felix Mbajiorgu, Idea conception, preparation and approval of the final manuscript. Trust Nyirenda, helps in laboratory work and preparation of manuscript. All the authors have read and agreed to the final manuscript.
Authors wish to acknowledge Mrs Hasiena Ali on the help in the laboratory work.
Table 1: general findings
Table 2: the classical linear gross morphometry of the kidney
Table 3: bowman's capsule and glomeruli measurements
Table 4: tubules diameter measurements
Table 5: tubular epithelial cells height
Figure 1: stereological method of sampling; the figure above shows representative scan slices of the kidney used on ImageJ to estimate total volume
Figure 2: 24 hour
urine volume; the figure shows the changes in the urine volume collected over
24 hours in the control and experimental groups, before commencement of treatment
and after the last dose of the drug
Figure 3: relative
medullary thickness (RMT); the figure shows the relative medullary thickness
measurements in the control and treatment groups
Figure 4: total kidney volume; the figure shows the total kidney volume estimated using stereological technique on ImageJ volumest plugin
Figure 5: biochemical
analyses of urea and creatinine; the figure shows results of serum urea (A) and
creatinine (B)
- Musabayane C, Windle R, Forsling M, Balment R. Arginine vasopressin mediates the chloroquine induced increase in renal sodium excretion. Trop Med Int Health. 1996; 1(4): 542-50. PubMed | Google Scholar
- Cooper R, Magwere T. Chloroquine: novel uses & manifestations. Indian J Med Res. 2008; 127(4): 305-16. PubMed | Google Scholar
- Musabayane CT, Cooper RG, Rao PVVP, Balment RJ. Effects of ethanol on the changes in renal fluid and electrolyte handling and kidney morphology induced by long-term chloroquine administration to rats. Alcohol. 2000; 22(3): 129-38. PubMed | Google Scholar
- Krafts K, Hempelmann E, Sk�rska-Stania A. From methylene blue to chloroquine: a brief review of the development of an antimalarial therapy. Parasitol Res. 2012; 111(1): 1-6. PubMed | Google Scholar
- Slater AF. Chloroquine: mechanism of drug action and resistance in Plasmodium falciparum. Pharmac Ther. 1993; 57(2): 203-35. PubMed | Google Scholar
- Rainsford K, Parke AL, Clifford-Rashotte M, Kean W. Therapy and pharmacological properties of hydroxychloroquine and chloroquine in treatment of systemic lupus erythematosus, rheumatoid arthritis and related diseases. Inflammopharmacol. 2015; 23(5): 231-69. PubMed | Google Scholar
- Savarino A. Use of chloroquine in viral diseases. Lancet Infect Dis. 2011; 11(9): 653-4. PubMed | Google Scholar
- Savarino A, Lucia MB, Giordano F, Cauda R. Risks and benefits of chloroquine use in anticancer strategies. Lancet Oncol. 2006; 7(10): 792-3. PubMed | Google Scholar
- Oluleye T, Babalola Y, Ijaduola M. Chloroquine retinopathy: pattern of presentation in Ibadan, Sub-Sahara Africa. Eye(Lond). 2016 Jan; 30(1): 64-7. PubMed | Google Scholar
- Kublin JG, Cortese JF, Njunju EM, Mukadam RAG, Wirima JJ, Kazembe PN et al. Reemergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine use in Malawi. J Infect Dis. 2003; 187(12): 1870-5. PubMed | Google Scholar
- Laufer MK, Thesing PC, Eddington ND, Masonga R, Dzinjalamala FK, Takala SL et al. Return of chloroquine antimalarial efficacy in Malawi. N Engl J Med. 2006; 355(19): 1959-66. PubMed | Google Scholar
- Pisa PT, Loots DT, Nienaber C. Alcohol metabolism and health hazards associated with alcohol abuse in a South African context: a review: review article. S Afr J Clin Nutr. 2010; 23(3): 4-10. Google Scholar
- Zakhari S. Overview: how is alcohol metabolized by the body. Alcohol Res Health. 2006; 29(4): 245-55. PubMed | Google Scholar
- Abdellatif AM, Hassan Y. Effects of Uninephrectomy on Morphological and Histological Measurements of the Remnant Kidney in a Goat (Capra hircus) Model. J Phys Pharm Adv. 2013; 3(3): 94-102. Google Scholar
- Yamashita SR, Atzingen ACv, Iared W, Bezerra ASdA, Ammirati AL, Canziani MEF, et al. Value of renal cortical thickness as a predictor of renal function impairment in chronic renal disease patients. Radiol Bras. 2015; 48(1): 12-6. PubMed | Google Scholar
- Almajdub M, Magnier L, Juillard L, Janier M. Kidney volume quantification using contrast-enhanced in vivo X-ray micro-CT in mice. Contrast Media Mol Imaging. 2008; 3(3): 120-6. PubMed | Google Scholar
- Nyengaard JR. Stereologic methods and their application in kidney research. J Am Soc Nephrol. 1999; 10(5): 1100-23. PubMed | Google Scholar
- Al-kahtani MA, Zuleta C, Caviedes-Vidal E, Garland Jr T. Kidney mass and relative medullary thickness of rodents in relation to habitat, body size and phylogeny. Physiol Biochem Zool. 2004; 77(3): 346-65. PubMed | Google Scholar
- Tejo Riquelme PA, Diaz Isenrath GB, Andino N, Borghi CE. Renal intraspecific variation along an aridity gradient detected by new renal indices in a desert herbivorous rodent. J Exp Zool. 2014; 321(6): 348-56. PubMed | Google Scholar
- Yang B, Bankir L. Urea and urine concentrating ability: new insights from studies in mice. Am J Physiol Renal Physiol. 2005; 288(5): F881-F96. PubMed | Google Scholar
- Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nature Methods. 2012; 9(7): 671-5. PubMed | Google Scholar
- Merzin M. Applying stereological method in radiology. Volume measurement: University of Tartu, Estonia. 2008. Google Scholar
- Albay D, Adler SG, Philipose J, Calescibetta C, Romansky SG, Cohen AH. Chloroquine-induced lipidosis mimicking Fabry disease. Mod Pathol. 2005; 18(5): 733-8. PubMed | Google Scholar
- Singh VP, Singh N, Jaggi AS. A review on renal toxicity profile of common abusive drugs. Korean J Physiol Pharmacol. 2013; 17(4): 347-57. PubMed | Google Scholar
- Assadi FK, Manaligod JR, Fleischmann LE, Zajac CS. Effects of prenatal ethanol exposure on postnatal renal function and structure in the rat. Alcohol. 1991; 8(4): 259-63. PubMed | Google Scholar
- Ahmed MH, Osman MM. Why does chloroquine impair renal function: Chloroquine may modulate the renal tubular response to vasopressin either directly by inhibiting cyclic AMP generation, or indirectly via nitric oxide. Med Hypotheses. 2007; 68(1): 140-3. PubMed | Google Scholar