What do you think of an unusual axillary mass?
Hafsa Benzzi, Majda Askour, Afaf Elouazani, Mouna Bouaddi, Badreddine Hassam, Amar Saidi
Corresponding author: Hafsa Benzzi, Dermatology Department, Mohamed V University, Rabat, Morocco 
Received: 11 Jul 2013 - Accepted: 02 Oct 2013 - Published: 13 Dec 2017
Domain: Clinical medicine
Keywords: Axillary mass, skin, apocrine adenocarcinoma
©Hafsa Benzzi et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Hafsa Benzzi et al. What do you think of an unusual axillary mass?. Pan African Medical Journal. 2017;28:309. [doi: 10.11604/pamj.2017.28.309.3092]
Available online at: https://www.panafrican-med-journal.com//content/article/28/309/full
What do you think of an unusual axillary mass?
Hafsa Benzzi1,&, Majda Askour1, Afaf Elouazani1, Mouna Bouaddi1, Badreddine Hassam1, Amar Saidi2
1Dermatology Department, Mohamed V University, Rabat, Morocco, 2Pathology Center United Nations, Rabat, Morocco
&Corresponding author
Hafsa Benzzi, Dermatology Department, Mohamed V University, Rabat, Morocco
Skin apocrine carcinoma is a rare malgnancy of epidermal adnexa, most frequent in axillary seat, where apocrine sweat gland are abundant, the neoplasm can arise in groin, anogenital, lips, eyelid, characterized by a plate or surface area of nodules hummocky. Etiology and incidence are not known. The prognosis is influenced by the risk of locoregional recurrence and metastatic evolution. We describe the case of 61-year-old man who presented a left axillary slow-growing mass since 2 years ago. The cutaneous biopsy objectified an apocrine adenocarcinoma. The paraclinic exams performed to detect primary breast were tumor negative, first step before confirming the diagnosis. Standard treatment is surgical excision with margins of 2 to 3cm for local tumor, for apocrine adenocarcinoma regional lymph node dissection if nodes were clinically positive is wide surgical excision. This kind of tumour is chemoresistant. In this case, adjuvant chemotherapy was indicated, before surgery to reduce tumoral volume. This case illustrates the importance clinicopathological correlation of skin cancer, particularly apocrine one. Clinical particularity and careful analyses histology helps diagnosis approach.
Skin apocrine carcinoma is a rare malignancy of epidermal adnexa, most frequent in axillary seat, where apocrine sweat gland are abundant, the neoplasm can arise in groin, anogenital, lips, eyelid, characterized by a plate or surface area of nodules hummocky. Etiology and incidence are not known. The prognosis is influenced by the risk of locoregional recurrence and metastatic evolution [1]. The slow evolution, painless charatacter leads to discover the tumor at systemic dissemination with locally invasive stage. The differential diagnosis between CAC and axillary skin metastasis adenocarcinoma, particularly breast is sometimes difficult. In the following report we present the case of a 61-year-old man with apocrine adenocarcinoma of the left axillary area with local lymph and distant metastases, which illustrates the difficulty.
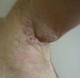
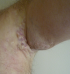
We describe the case of 61-year-old man without a medical history who consulted dermatology department presenting a left axillary slow-growing mass (Figure 1, Figure 2) since 2 years ago, painless at first, becoming painful since 6 months that conducts the patient to consult. Physical examination objectified a hummocky plate full of nodules measuring 10-6cm, erythematous, purple color, painful at mobilization, adherent, the plate is infiltrating surrounding tissue, there was no bleeding or serious discharge. The member was oedematous (Figure 3), superficial venous maze, without neither palpable mass of breast nor supernumerary nipple. There was homolateral nodes individualized clinically and the somatic examination was normal. The cutaneous biopsy objectified an apocrine adenocarcinoma. The paraclinic exams performed to detect primary breast were tumor negative. A thorough systemic workup for metastatic disease have performed, tomography showed lungs and nodes, scintigraphy showed bones metastases. In conlcusion, the patient presented an axillary adenocarcinoma apocrine with node, bone and lung metastases. After multi-disciplinary concertation a polychimiotherapy was indicated.
CAC constitutes a rare cutaneous adenocarcinoma, commonly seen in the axilla [1]. The primary cutaneous apocrine carcinomas are malignant adnexal tumor that develops in skin areas rich in apocrine glands. Presumptive progenitor cells for apocrine differentiation may be present along the lines joining the axillae, areolae and anogenital and they may be responsible for giving rise to some examples of extramammary Paget�s disease [2]. They arise in the form of plaques or nodules hummocky more or less confluent, painless. Evolution is the more often indolent, slowly progressive, but can be aggressive, with a risk of local recurrence or metastatic patterns including the pulmonary, brain and bone, which can lead to death [3]. One hundred cases of ACC [4,5], were described in literature, mainly located in the axillary level, but other locations are possible especially on the scalp, forehead, eyelids the upper lip, cheeks, the pubis, the nipple and fingers [6].
�
Before treating, the difficulty of differential diagnosis between histological CAC and axillary metastasis of lobular carcinoma breast was recently highlighted [7] or supernumerary breast carcinoma, some malignant tumor of sweat glands, the whole interest of a thorough clinical and histological study. Axilla is undoubtedly the most frequent location of apocrine tumor due to high density of apocrine glands, other locations are possible especially on the scalp, forehead, eyelids, the upper lip, cheeks, the pubis, the nipple and fingers [8]. Clinically, the macroscopic aspect is not specific, nodules or plaques, initially flesh-colored, with a slow growth.�
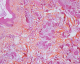
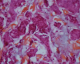
To discuss diagnostic of cutaneous metastases adenocarcinoma and therefore, we need imperatively complete morphological examination. Histologicaly the ACC are organized in tubular and papillary structures or massive basophils (Figure 4, Figure 5), the diagnosis of apocrine tumor has been mentied in presence of decapitation secretion images in a cystic structure or projections papillary [9], like our patient who had a sheet of cell floating in mucus, the nucleus is large, irregular and abundant cytoplasm. Sometimes there is variation in patterns of tumors. Obaidat et al. [10] described it as a �composition of tubular and ductal structures, papillary projections, subcutis infiltration, with necrotic area and decapitation of secretion�. In the other hand, Zelger et al. [1] reported that the histology of CAC is a �cobblestone pattern� of epithelium, vacuoles along the apical end of the epithelium, with variety of cell types and apocrine secretion. So both of descriptions insist on decapitation of secretion in eosinophilic epithelial cells and the second argument is attachment of neighbouring follicular epithelium.�
The presence of a positive immunostaining may help in diagnosis but is not a sufficient argument. The GCDFP-15 is expressed specifically by apocrine epithelium but may still be any positive mammary adenocarcinoma in differentiation associated with apocrine gland secretion and carcinomas of the salivary glands [11]. In fact, many of the reported CACs have shown weak and focal expression of GCDFP-15 or have failed to demonstrate any expression of the marker [7]. A immunostaining with anti-herceptin-2 [Her-2] can be used because it is sometimes positive in skin metastases a mammary adenocarcinoma, whereas carcinomas primary cutaneous apocrine or eccrine do not set the anti-Her-2 [12]. Hormone receptors, however, are not discriminatory to distinguish CAC and metastasis lobular breast carcinoma.�
In the past, negativity of ER and PR with positive expression of androgen receptor (AR) has shown to be relevant criteria for diagnosis of CAC [13]. Klein et al. [14] also suggested that the lack of expression of ER and PR is more important from a practical point of view than positivity of AR. In contrast, Robson et al. [15] demonstrated that 62% were positive for ER, 60% were positive for PR and 36% were negative for AR. For our patient, ER and PR were both positif. Ninety hundred percent of oestrogen receptor and 70% of progesterone receptor. Immunochemical analysis of CAC has a debatable aspect.�
Therefore, the hormone receptors are not discriminatory to distinguish CAC and metastasis lobular breast carcinoma, because apocrine glands expresses estrogen and progesterone receptors. It needs set of arguments. CK7 is not a discriminatory marking because it can be present in both CAC and in breast metastase. In this case, we opted for CAC due to clinical presentation in axilla of a hummoky tumor, the indolent clinical course and negativity explorations in search of another primitive neoplasy and decapitation secretion and attachment of neighborouing follicular epithelium caracteristis of apocrin glands in histology. Standard treatment is surgical excision with margins of 2 to 3cm for local tumor, for apocrine adenocarcinoma regional lymph node dissection if nodes were clinically positive is wide surgical excision. This kind of tumour is chemoresistant [16]. In this case, adjuvant chemotherapy was indicated, before surgery to reduce tumoral volume. Witold Kycler and al. [17] recommended chemotherapy (adriablastin and cyclophosphamide) and palliative treatment for metastatic apocrine adenocarcinoma of the axillary area.�
We don�t dispose of much data for evolution, but a study of series treating apocrine adenocarcinoma since 1951, showed 39 were located in axilla, 12 patient had local relapse, 23 had lymph nodes relapse, 12 patients with visceral metastases and 9 patient dead [18]. We need more prospective studies to define precisely prognosis of such tumors that are still rare, so we can adopt a efficient diagnostic and treatment strategy.
In conclusion, this case illustrates the importance clinicopathological correlation of skin cancer, particularly apocrine one. Clinical particularity and careful analyses histology helps diagnosis approach. More studies will help determining there is no treatment consensus. The worrying aspect in our histological patient should prompt careful clinical monitoring to detect a possible poor outcome.
The authors declare no competing interests.
All authors have read and agreed to the final manuscript.
Figure 1: an axillary hummoky tumor
Figure 2: burgeoning aspect of the tumor
Figure 3: edema of the upper limb
Figure 4: carcinomatous
proliferation separating the collagen fibers of the deep dermis: it is arranged
in tubes, in wires and of variable size in lobules (x4)
Figure 5: cells with increased volume in places, often vesicular nucleus and nucleolus and a hyperchromic also eosinophilic granular cytoplasm (x10)
- Zelger BG, Stelzmueller I, Dunst KM, Zelger B. Solid apocrine carcinoma of the skin: report of a rare adnexal neoplasm mimicking lobular breast carcinoma. J Cutan Pathol. 2008 Mar;35(3):332-6. PubMed | Google Scholar
- Murphy GP, Elder DE. Non-melanocytic tumors of the skin. Atlas of tumor Pathology, 1991, third series. Vol I, pp 61-153. Armed Forces Institute of Pathology, Washington DC.
- Shintaku M, Tsuta K, Yoshida H, Tsubura A, Nakashima Y, Noda K. Apocrine adenocarcinoma of the eyelid with aggressive biological behavior: report of a case. Pathol Int. 2002 Feb;52(2):169-73. PubMed | Google Scholar
- Baes H, Suurmond D. Apocrine sweat gland carcinoma - Report of a case. Br J Dermatol. 1970 Oct;83(4):483-6. PubMed | Google Scholar
- Robson A, Lazar AJ, Ben Nagi J, Hanby A, Grayson W, Feinmesser M et al. Primary cutaneous apocrine carcinoma: a clinico-pathologic analysis of 24 cases. Am J Surg Pathol. 2008 May;32(5):682-90. PubMed | Google Scholar
- Katagiri Y, Ansai S. Two cases of cutaneous apocrine ductal carcinoma of the axilla - Case report and review of the litterature. Dermatology. 1999;199(4):332-7. PubMed | Google Scholar
- Fernandez-Flores A. Primary cutaneous apocrine carcinoma versus metastasis, a plea to the dermatopathology community. Am J Dermatopathol. 2010 Dec;32(8):853-4. PubMed | Google Scholar
- Zehr KJ, Rubin M, Ratner L. Apocrine adenocarcinoma presenting as a large ulcerated axillary mass. Dermatol Surg. 1997 Jul;23(7):585-7. PubMed | Google Scholar
- Kim HK, Chung KI, Park BY, Bae TH, Kim WS, Lee TJ. Primary apocrine carcinoma of scalp: report of primary scalp cutaneous apocrine carcinoma indistinguishable from cutaneous metastasis of breast carcinoma. J Plast Reconstr Aesthet Surg. 2012 Mar;65(3):e67-70. PubMed | Google Scholar
- Obaidat NA, Alsaad KO, Ghazarian D. Skin adnexal neoplasms--part 2: an approach to tumors of cutaneous sweat glands. J Clin Pathol. 2007 Feb;60(2):145-59. Epub 2006 Aug 1. PubMed | Google Scholar
- Matsuo K, Fukutomi T, Hasegawa T, Akashi-Tanaka S, Nanasawa T, Tsuda H. Histological and immunohistochemical analysis of apocrine breast carcinoma. Breast Cancer. 2002;9(1):43-9. PubMed | Google Scholar
- Hiatt KM, Pillow JL, Smoller BR. Her-2 expression in cutaneous eccrine and apocrine neoplasms. Mod Pathol. 2004 Jan;17(1):28-32. PubMed | Google Scholar
- Cohen M, Cassarino DS, Shih HB, Abemayor E, St John M. Apocrine Hidradenocarcinoma of the scalp: a classification conundrum. Head Neck Pathol. 2009 Mar;3(1):42-6. PubMed | Google Scholar
- Klein W, Chan E, Seykora JT. Tumors of the epidermal appendages. Lever�s histopathology of the skin. 9th ed. Philadelphia, PA: Lippincott Williams & Wilikins. 2005. p867-e926.
- Robson A, Lazar AJ, Ben Nagi J et al. Primary cutaneous apocrine carcinoma: a clinico-pathologic analysis of 24 cases. Am J Surg Pathol. 2008 May;32(5):682-90. PubMed | Google Scholar
- Morabito A, Benlaqua P, Vitale S et al. Clinical management of case of apocrine gland carcinoma of the scalp: efficacy of a chemotherapy schedule with methotrexate and bleomycine. Tumori. 2000 Nov-Dec;86(6):472-4. PubMed | Google Scholar
- Witold Kycler, Konstanty Korski, Piotr Łaski, Elżbieta W�jcik, Danuta Bręborowicz. Metastatic apocrine adenocarcinoma of the axillary area. Rep Pract Oncol Radiother. 2006;11(6):299-302. Google Scholar
- Chamberlain RS, Huber K, White JC, Travaglino-Parda R. Apocrine gland carcinoma of the axilla: review of the literature and recommendations for treatment. Am J Clin Oncol. 1999 Apr;22(2):131-5. PubMed | Google Scholar