Clinical risk factors for in-hospital mortality in older adults with HIV infection: findings from a South African hospital administrative dataset
Kumeren Govender, Fatima Suleman, Yoshan Moodley
Corresponding author: Kumeren Govender, Nelson Mandela School of Medicine, University of KwaZulu-Natal, South Africa 
Received: 19 Oct 2016 - Accepted: 12 Feb 2017 - Published: 03 Mar 2017
Domain: Epidemiology
Keywords: HIV, hospital mortality, risk factors, older adult
©Kumeren Govender et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Kumeren Govender et al. Clinical risk factors for in-hospital mortality in older adults with HIV infection: findings from a South African hospital administrative dataset. Pan African Medical Journal. 2017;26:126. [doi: 10.11604/pamj.2017.26.126.11000]
Available online at: https://www.panafrican-med-journal.com//content/article/26/126/full
Original article 
Clinical risk factors for in-hospital mortality in older adults with HIV infection: findings from a South African hospital administrative dataset
Clinical risk factors for in-hospital mortality in older adults with HIV infection: findings from a South African hospital administrative dataset
Kumeren Govender1,2,&, Fatima Suleman2, Yoshan Moodley3
1Nelson R Mandela School of Medicine, University of KwaZulu-Natal, South Africa, 2Discipline of Pharmaceutical Sciences, University of KwaZulu-Natal, South Africa, 3Discipline of Anaesthesiology and Critical Care Medicine, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, South Africa
&Corresponding author
Kumeren Govender, Nelson R Mandela School of Medicine, University of KwaZulu-Natal,
South Africa
Introduction: the proportion of older South African adults (aged ≥50 years old) with HIV infection requiring hospitalization is likely to increase in the near future. Clinical risk factors for in-hospital mortality (IHM) in these patients are not well described. We aimed to identify clinical risk factors associated with IHM and their overall contribution towards IHM in older South African adults with HIV infection.
Methods: clinical data for 690 older adults with HIV infection was obtained from the hospital administrative database at the Hlabisa Hospital in KwaZulu-Natal, South Africa. Logistic regression was used to determine independent clinical risk factors for IHM. Population-attributable fractions (PAFs) were calculated for all independent clinical risk factors identified.
Results: male gender (p=0.005), CD4 count <350 cells/mm3 (p=0.035), unknown CD4 count (p=0.048), tuberculosis (p=0.033) and renal failure (p=0.013) were independently associated with IHM. Male gender contributed the most to IHM (PAF=0.22), followed by unknown CD4 count (PAF=0.14), tuberculosis (PAF=0.12), renal failure (PAF=0.06) and CD4 count <350 cells/mm3 (PAF=0.01).
Conclusion: although further research is required to confirm our findings, there is potential for these clinical risk factors identified in our study to be used to stratify patient risk and reduce IHM in older adults with HIV infection.
Dramatic scale-up in the provision of antiretroviral therapy (ART) for HIV-infection in sub-Saharan Africa continues to improve the quality of life and radically reduce early mortality associated with HIV [1]. In sub-Saharan Africa it is estimated that ART coverage reached 41% in 2014, up from less than 1% in 2000 [2]. This has led to a substantial increase of the HIV prevalence in sub-Saharan Africa with nearly 11.5 million people initiated on therapy as of 2015 [2]. Mills et al. demonstrated that the life expectancy of those who are HIV-infected can approach the life expectancy of those who are not HIV-infected provided ART is initiated early (CD4 count >250 cells/µL) [3]. Therefore, as scale-up of ART in sub-Saharan Africa continues to rapidly expand, the life expectancy of the HIV-infected population continue to improve, resulting in a temporal transition of the HIV epidemic toward the older age groups. In Africa, the World Health Organization (WHO) defines older age as 50 years or more, due to new roles and loss of previous roles in society [4]. An increase in the prevalence of HIV in persons older than 50 years of age has already been reported in several developed countries. In the United States ART was first introduced in 1996, after which the prevalence of HIV in older adults (≥ 50 years old) grew from 17% in 2001 to 30% in 2008 [5, 6].
In Italy the proportion of older adults living with HIV increased from 4.9% in the years 1982-1990 to 15.9% in the years 2000-2005 [7]. Older adults in 2012 represented 9.5% (608 000 persons) of the total HIV burden in South Africa [8]. A study in rural South Africa quantified the aging of the HIV epidemic and predicted that the number of HIV-infected older adults would increase by 100% between 2004 and 2025 [9]. Moreover there is an increasing trend in the incidence of HIV in older adults reported in rural settings where health services are now becoming accessible [8]. Inevitably a proportion of these older adults with HIV infection will require hospitalization for various HIV-related or HIV-unrelated conditions during their lifetime [10-12]. Studies on HIV-infected individuals from the United States and Greece have shown higher comorbidities, longer lengths of admission and more frequent hospitalizations in older patients compared to younger patients [5, 13, 14]. Furthermore studies suggest higher inpatient mortality in HIV-infected older adult patients when compared to their HIV-infected younger counterparts [14]. In-hospital mortality (IHM) is associated with higher health care expenditure and is also an important indicator of a public health system�s response to the HIV epidemic [14, 15]. Considering the progressively high levels of IHM in older adults infected with HIV and its potential impact on resource utilization within public health systems, it is important that methods such as risk stratification systems be developed, that aim to reduce IHM in these patients. Risk stratification systems involve the identification of independent clinical risk factors within a population at risk of a poor outcome [16]. However, clinical risk factors that contribute to IHM in older adults infected with HIV are not well described, particularly in the South African context where the burden of HIV in this sub-population is amongst the highest in the world. We therefore sought to identify clinical risk factors associated with IHM and their overall contribution toward this poor outcome in older adults with HIV infection admitted to a South African hospital.
Study design and setting: this study was a retrospective analysis of data collected at the Hlabisa Hospital between January 2011 and February 2015. The hospital provides services to over 230 000 people in the Hlabisa district, Mtubatuba and parts of the "Big Five" municipalities in the Northern KwaZulu-Natal Province, South Africa. The overall HIV incidence in the Hlabisa district has been estimated at 3.2 per 100 person years while the district prevalence of HIV in the older adult is estimated at 9.5% and predicted to further rise [8]. Other characteristics of the Hlabisa population are described elsewhere [17].
�
Data source and definitions: HIV-positive patients aged ≥ 50 years old who were admitted to the Hlabisa Hospital between January 2011 and February 2015 were identified through review of hospital administrative data collected as part of the Africa Centre Demographic Information System (ACDIS) [17]. Patients were considered HIV-infected if they had an HIV-positive serostatus recorded in the database. Demographic characteristics analysed included gender and age; and clinical characteristics analysed included HIV status, ART status, CD4 count, and co-morbidity. Patient comorbidities were identified from the discharge diagnoses fields listed in the administrative database using the : International Statistical Classification of Diseases and Related Health Problems- 10th edition (ICD-10) code groupings proposed by the American Healthcare Cost and Utilization Project (HCUP) [18]. The most prevalent co-morbidities were included in the final statistical analysis in accordance with other similar studies on IHM [19].�
Statistical analysis: univariate and multivariate statistical testing of the final dataset was undertaken. All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 23 (IBM Corp., Armonk, NY, USA).�
Univariate statistical methods: gender, ART status, CD4 count, and co-morbidities were converted to categorical variables. Categorical data was analysed using χ2 tests or Fishers Exact test, where appropriate. Age data was treated as a continuous variable. When tested for normality using the Shapiro-Wilk test, age data was not found to be normally distributed and was therefore analysed using a Mann-Whitney U test. Results of the univariate statistical testing are presented as frequencies and percentages, or medians and interquartile ranges (IQRs), where appropriate.�
Multivariate statistical methods: : logistic regression was used to identify independent associations between clinical characteristics and IHM. Clinical characteristics with p<0.2 from the univariate testing were included in the logistic regression analysis. This purposeful selection of clinical characteristics ensured that we did not potentially violate the "10 outcome events per single variable" rule of thumb for logistic regression analyses and also ensured that we would obtain a parsimonious regression model [20, 21]. A Hosmer-Lemeshow test was used to measure model fit. Results were presented as odds ratios (OR) with a confidence interval of 95% (CIs). Results with a p-value <0.05 were considered statistically significant.�
Population-attributable fraction (PAF): the PAF is defined as the proportion of cases in the population that would not have occurred in the absence of the exposure [22]. In this study the PAF was calculated for each independent risk factor identified from the binary logistic regression analysis (clinical characteristics with p<0.05 in the regression model) using conventional methods [22].�
Ethical approval: ethical approval for this study was obtained from the University of KwaZulu-Natal Biomedical Research Ethics Committee (Protocol EXM277/15 & Protocol BE281/16).
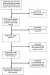
Following application of the relevant inclusion and exclusion criteria (Table 1), the final study population consisted of 690 older adult patients with HIV infection admitted to the Hlabisa Hospital between January 2011 and February 2015 (Figure 1). The cumulative incidence of IHM in the study population was 27.1% (95% CI: 23.9-30.5%). The baseline clinical characteristics of the entire study population are shown in Table 2. The median age of the entire study population was 58.0 (IQR: 52.0-61.0) years old. Approximately 51.2% of the entire study population was male. Of the entire study population, 45.4% were receiving ART, 53% were not receiving ART, and ART status could not be established in 1.6% of patients. Nine percent of patients in the entire study population had known CD4 counts <350 cells/mm3, 4.1% had CD4 counts ≥350 cells/mm3, while 87% of the entire study population did not have CD4 counts recorded. The ten most prevalent comorbidities in the entire study population were: Tuberculosis, acute bronchitis, acute and unspecified renal failure, intestinal infection, essential hypertension, anaemia, pneumonia (excluding that caused by tuberculosis), diabetes mellitus without complications, mycoses, and meningitis (excluding TB meningitis).
�
Univariate statistical associations between patient clinical characteristics and IHM are also reported (Table 2). Male gender (p=0.001), CD4 cell count (p=0.045), tuberculosis (p=0.017), renal failure (p=0.013) and essential hypertension (p=0.026) met the criteria for inclusion in the multivariate analysis (p<0.2 from the univariate analysis). Five factors (Male gender (p=0.005), CD4 count <350 cells/mm3, (p=0.035), unknown CD4 count (p=0.048), tuberculosis (p=0.033) and renal failure (p=0.013)) were found to be independently associated with a higher risk of IHM in the logistic regression analysis (Table 3). When entered into the logistic regression analysis, variables such as essential hypertension, pneumonia (excluding that caused by TB) and mycosis were not independently associated with an increased risk of IHM (Table 3). The Hosmer-Lemeshow test indicated adequate regression model fit (p>0.05). The PAF obtained for the independent risk factors obtained from the logistic regression analysis (Table 4) suggested male gender to have the highest contribution to IHM (PAF=0.22) followed by unknown CD4 cell count (PAF=0.14), tuberculosis (PAF=0.12), renal failure (PAF=0.06) and CD4 cell count <350 cells/mm3 (PAF=0.01).
Almost 1 in 4 older adults (27 in every 100 older adult patients) with HIV infection suffered IHM in our study. This estimate is far higher than that described in similar populations from developed world countries such as the USA [14], but still lies within the range of estimates reported in a study from another African country, Malawi (20-30 deaths per every 100 patients) [19]. Independent associations between several clinical characteristics (Male gender, CD4 count, tuberculosis, and renal failure) and a higher risk of IHM in older adults with HIV infection were noted in our study. Male gender was associated with a 67% higher risk of IHM when compared with female gender, and accounted for almost 1 in 5 patient deaths. Male gender is associated with delayed presentation to healthcare facilities [23], and this may have been the case in our study where far more patients who were male presented with advanced HIV disease and subsequently suffered poorer outcomes when compared with their female counterparts. CD4 count measurements are important for determining staging of HIV disease and were required to determine a patient's eligibility for antiretroviral therapy. Our findings with regard to a low CD4 count are in keeping with the findings of a systematic review by Wagjanga et al., which also identified low CD4 count measurements, a proxy for late presentation for care, as an important determinant of inpatient survival [24]. Our findings for patients with an unknown CD4 count suggest that a large proportion of these patients might potentially have low CD4 counts, and opportunities for intervention in these patients are missed. The PAFs obtained for the CD4 count categories suggests that a known low CD4 count contributes less to overall mortality than an unknown CD4 count measurement, which is likely due to known immunocompromised patients receiving subsequent treatment. These findings further highlight the importance of obtaining CD4 count measurements in patients with HIV infection.
�
HIV infection substantially increases the risk of tuberculosis, a concerning relationship considering the high burden of tuberculosis in the general South African population. UNAIDS reports that tuberculosis is among the leading causes of mortality in people living with HIV infection [25]. This is supported by a South African autopsy study by Cox et al., which found that a large proportion of deaths in patients with HIV infection were likely caused by tuberculosis [26]. Our findings suggest a 47% higher risk of IHM in HIV and tuberculosis co-infected patients, and do not seem out of place considering the high burden of tuberculosis and HIV in the South African setting, with 12 in every 100 inpatient deaths in older adults with HIV infection being attributed to tuberculosis. Active case finding and adequate treatment of tuberculosis among the HIV-infected population remains a key intervention for reducing morbidity and mortality in our setting. Persons with HIV may develop renal failure as a result of the nephrotoxic side effects of some antiretroviral therapy drugs, or through nephritis resulting from the direct infection of the kidney by HIV or opportunistic infections. Acute renal failure is estimated to occur in up to 30% of patients with HIV infection, and is much more common in this patient group when compared to HIV-uninfected patients [15, 27]. The implications of renal failure on subsequent patient outcomes are marked, often linked with higher morbidity and mortality in afflicted patients [28-30]. This might explain our findings for a 91% higher risk of IHM in patients with renal failure, with 6 in every 100 inpatient deaths in our study being attributed to renal failure.�
Our study did not identify independent associations between non-communicable comorbid disease (besides renal failure) and IHM. This finding may be related to infectious diseases contributing to mortality in a rural HIV-infected population rather than non-communicable diseases. This is further supported by the finding of communicable conditions comprising the majority of the most common patient comorbidities in our study. However, it is important to note that South Africa has begun a state of epidemiological transition, whereby higher disease burden appears to be shifting away from communicable diseases toward non-communicable diseases [31, 32]. It is likely that in the future non-communicable comorbid diseases will become more important in the context of patient outcomes in South Africa. Increasing age above 50 years old was also not associated with a higher risk of IHM in this study. Increasing age is usually associated with increased non-communicable disease comorbidity [33], which potentially increases the risk of future poor patient outcomes. However, we report a low burden of communicable disease in this study, despite the age of our study population. This might explain the observed lack of association between age and IHM in our study. No independent associations (either protective or harmful) with IHM could be established for anaemia and antiretroviral therapy status in our study. It is possible that anaemia was under-diagnosed, as anaemia is usually described as a common haematological abnormality in patients with HIV infection which is associated with poor survival [34]. In the general HIV-infected population, antiretroviral therapy use is associated with a reduction in mortality with HIV-infected individuals on ART now achieving near-normal life expectancies [3, 35]. Notably, almost half our study population was receiving antiretroviral therapy, which might explain our findings for this characteristic.�
Study limitations: data related to a number of potentially relevant patient clinical characteristics, for example body mass index, were not collected as part of the hospital administrative database. Furthermore, data related to medication use (other than antiretroviral therapy) and laboratory results (other than CD4 count measurements) were not collected. As such we were unable to investigate the impact of these clinical characteristics on the incidence of IHM. The specific pathological cause of death was indeterminable from the hospital administrative database, and we therefore describe all-cause mortality in this study. Future studies employing a prospective study design with an emphasis on collecting data on potentially relevant population-specific characteristics are required to overcome the aforementioned study limitations. There were also several limitations identified which can only be overcome through health system strengthening. For instance, this study limited its analysis to inpatient outcomes as patient survival status following hospital discharge could not be reliably ascertained. Patient tracking and use of community caregivers to conduct home visits might improve follow-up of discharge patients [36]. In addition, a large proportion of the CD4 count measurement data was missing. The challenge of missing CD4 count measurements has been identified elsewhere [37], and it once again represents a challenge which can only be overcome through health systems approaches such as the rollout of point of care CD4 count machines and the development of relevant checklists to improve CD4 count measurement recording [38]. The data used in our study was collected from a single, rural hospital and the findings of this study may not be generalizable to other South African hospital settings. Multisite studies, involving several institutions located in both rural and urban settings are required. While our study does have limitations, it still provides an important account of clinical risk factors for IHM in older patients with HIV infection, which can be used to develop future interventions aimed at reducing patient risk.
Almost 1 in 4 older adult patients with HIV infection in our study suffered IHM. Male gender, a CD4 count <350 cells/mm3, an unknown CD4 count, tuberculosis and renal failure were important predictors of a higher risk of IHM in our study. The proportion of IHM attributed to each of these clinical risk factors ranged from 0.01 for CD4 count <350 cells/mm3, to 0.22 for male gender. While the most prevalent non-communicable diseases investigated in this study did not appear to contribute to IHM when compared with communicable disease (with the exception of renal failure), it is likely that this situation will change in the near future. Further research is required not only to validate the findings of our study, but to also overcome limitations identified in our study. It is hoped that our findings will be used to develop interventions aimed at reducing the risk of IHM in older adults, such as risk stratification systems.
What is known about this topic
- The proportion of older South African adults (aged ≥ 50 years old) with HIV infection requiring hospitalization is likely to increase in the near future;
- Clinical risk factors for in-hospital mortality (IHM) in these patients are not well described.
What this study adds
- This study adds to the body of evidence that almost 1 in 4 older adults (27 in every 100 older adult patients) with HIV infection suffered IHM in our setting ;
- Male gender, a CD4 count <350 cells/mm3, an unknown CD4 count, tuberculosis and renal failure were important predictors of a higher risk of IHM in older HIV-infected adult patients;
- The study hopes that our findings will be used to develop interventions aimed at reducing the risk of IHM in older adults, such as risk stratification systems.
The authors declare no competing interest.
Kumeren Govender, Yoshan Moodley and Fatima Suleman participated in the design of the study, analysis and interpretation of the data, writing and critically revising the manuscript. All authors have read and agreed to the final version of this manuscript and have contributed to its content.
This work forms a component of the Masters degree studies of Kumeren Govender, supervised by Yoshan Moodley and Fatima Suleman. Kumeren Govender was supported by the UKZN Medical Education Partnership Initiative (MEPI): grant number: 5R24TW008863 from the President's Emergency Plan for AIDS Relief (PEPFAR), and the National Institutes of Health, U. S. Department of Health and Human Services. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the views of the funders or government.
Table 1: inclusion and exclusion criteria used to derive the study population
Table 2: clinical characteristics of the study population expressed as a frequency (%)
Table 3: results of multivariate analysis: Clinical characteristics independently/not independently associated with in-hospital mortality
Table 4: population-attributable fractions (PAFs) for clinical variables independently associated with in-hospital mortality
Figure 1: study profile
- Hontelez JA, de Vlas SJ, Baltussen R, Newell M-L, Bakker R, Tanser F et al. The impact of antiretroviral treatment on the age composition of the HIV epidemic in sub-Saharan Africa. AIDS. 2012;26(01). PubMed | Google Scholar
- World Health Organization. Global Health Sector Response to HIV, 2000-2015: Focus on Innovations in Africa: Progress Report. 2015 [cited 2017 Feb 12] Available from http://apps.who.int/iris/bitstream/10665/198065/1/9789241509824_eng.pdf. 2015 PubMed | Google Scholar
- Mills EJ, Bakanda C, Birungi J, Mwesigwa R, Chan K, Ford N et al. Mortality by baseline CD4 cell count among HIV patients initiating antiretroviral therapy: evidence from a large cohort in Uganda. AIDS. 2011;25(6):851-5. PubMed | Google Scholar
- World Health Organization. Definition of an older or elderly person. cited 2016 Jan 17. 2016. PubMed | Google Scholar
- Skiest DJ, Rubinstien E, Carley N, Gioiella L, Lyons R. The importance of comorbidity in HIV-infected patients over 55: a retrospective case-control study. Am J Med. 1996;101(6):605-11. PubMed | Google Scholar
- Moore RD, Chaisson RE. Natural history of HIV infection in the era of combination antiretroviral therapy. AIDS. 1999;13(14):1933-42. PubMed | Google Scholar
- Longo B, Camoni L, Boros S, Suligoi B. Increasing proportion of AIDS diagnoses among older adults in Italy. AIDS Patient Care STDS. 2008;22(5):365-71. PubMed | Google Scholar
- Wallrauch C, B�rnighausen T, Newell M. HIV prevalence and incidence in people 50 years and older in rural South Africa. S Afr Med J. 2010;100(12):812-3. PubMed | Google Scholar
- Hontelez JA, Lurie MN, Newell M-L, Bakker R, Tanser F, B�rnighausen T et al. Ageing with HIV in South Africa. AIDS.2011 Aug 24;25(13):1665-7. PubMed | Google Scholar
- Buchacz K, Baker RK, Moorman AC, Richardson JT, Wood KC, Holmberg SDet al. Rates of hospitalizations and associated diagnoses in a large multisite cohort of HIV patients in the United States, 1994-2005. AIDS. 2008;22(11):1345-54. PubMed | Google Scholar
- Berry SA, Fleishman JA, Moore RD, Gebo KA. Trends in reasons for hospitalization in a multisite United States cohort of persons living with HIV, 2001-2008. J Acquir Immune Defic Syndr. 2012;59(4):368. PubMed | Google Scholar
- Martinson NA, Omar T, Gray GE, Vermaak JS, Badicel M, Degiannis E et al. High rates of HIV in surgical patients in Soweto, South Africa: impact on resource utilisation and recommendations for HIV testing. Trans R Soc Trop Med Hyg. 2007;101(2):176-82. PubMed | Google Scholar
- Metallidis S, Tsachouridou O, Skoura L, Zebekakis P, Chrysanthidis T, Pilalas D et al. Older HIV-infected patients-an underestimated population in northern Greece: epidemiology, risk of disease progression and death. Int J Infect Dis. 2013;17(10):e883-e91. PubMed | Google Scholar
- Tadros A, Shaver E, Davis SM, Davidov DM. Hospitalizations of older patients with human immunodeficiency virus in the United States. J Emerg Med. 2012;43(6):1138-44. PubMed | Google Scholar
- Cleary S, Boulle A, Castillo-riquelme M, Mcintyre D. The burden of HIV/AIDS in the public healthcare system. S Afr J Econ. 2008;76(s1):S3-S14. PubMed | Google Scholar
- Sullivan LM, Massaro JM, D'Agostino Sr RB. Tutorial in biostatistics: presentation of multivariate data for clinical use: the Framingham Study risk score functions. Stat Med. 2004;23:1631-60. PubMed | Google Scholar
- Tanser F, Hosegood V, B�rnighausen T, Herbst K, Nyirenda M, Muhwava W et al. Cohort Profile: Africa centre demographic information system (ACDIS) and population-based HIV survey. Int J Epidemiol. 2008;37(5):956-62. PubMed | Google Scholar
- Agency for Healthcare Research and Quality, Rockville, MD. Healthcare Cost and Utilization Project (HCUP) [internet]. 2016 [cited 2017 Jan 15]. Available from: https://www.hcup-us.ahrq.gov/toolssoftware/ccs10/ccs10.jsp#download Google Scholar
- Akinkuotu A, Roemer E, Richardson A, Namarika D, Munthali C, Bahling A et al. In-hospital mortality rates and HIV: a medical ward review, Lilongwe, Malawi. Int J STD AIDS. 2011;22(8):465-70. PubMed | Google Scholar
- Bursac Z, Gauss CH, Williams DK, Hosmer D. Purposeful selection of variables in logistic regression. Source Code Biol Med. 2008;3(1):17. PubMed | Google Scholar
- Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR. A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol. 1996;49(12):1373-9. PubMed | Google Scholar
- Steenland K, Armstrong B. An overview of methods for calculating the burden of disease due to specific risk factors. Epidemiology. 2006;17(5):512-9. PubMed | Google Scholar
- Banks I. No man's land: men, illness, and the NHS. Br Med J. 2001;323(7320):1058. PubMed | Google Scholar
- Wajanga BM, Webster LE, Peck RN, Downs JA, Mate K, Smart LR et al. Inpatient mortality of HIV-infected adults in sub-Saharan Africa and possible interventions: a mixed methods review. BMC Health Serv Res. 2014;14(1):627. PubMed | Google Scholar
- Joint United Nations Programme on HIV/AIDS. UNAIDS Gap Report 2014 [internet]. 2014 [cited 2017 Jan 15]. Available from: http://www.unaids.org/en/.../unaids/.../unaidspublication/2014/UNAIDS_Gap_report_en.pdf. PubMed | Google Scholar
- Cox JA, Lukande RL, Lucas S, Nelson AM, Van Marck E, Colebunders R. Autopsy causes of death in HIV-positive individuals in sub-Saharan Africa and correlation with clinical diagnoses. AIDS Rev. 2010;12(4):183-94. PubMed | Google Scholar
- Kalim S, Szczech LA, Wyatt CM. Acute kidney injury in HIV-infected patients. Semin Nephrol. 2008 Nov;28(6):556-62. PubMed | Google Scholar
- Go AS, Chertow GM, Fan D, McCulloch CE, Hsu C-y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296-305. PubMed | Google Scholar
- Choi AI, Li Y, Parikh C, Volberding PA, Shlipak MG. Long-term clinical consequences of acute kidney injury in the HIV-infected. Kidney Int. 2010;78(5):478-85. PubMed | Google Scholar
- Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365-70. PubMed | Google Scholar
- Wagner K-H, Brath H. A global view on the development of non communicable diseases. Prev Med. 2012;54:S38-S41. PubMed | Google Scholar
- Steyn K, Sliwa K, Hawken S, Commerford P, Onen C, Damasceno A et al. Risk factors associated with myocardial infarction in Africa the INTERHEART Africa study. Circulation. 2005;112(23):3554-61. PubMed | Google Scholar
- Rodriguez-Penney AT, Iudicello JE, Riggs PK, Doyle K, Ellis RJ, Letendre SL et al. Co-morbidities in persons infected with HIV: increased burden with older age and negative effects on health-related quality of life. AIDS Patient Care STDS. 2013;27(1):5-16. PubMed | Google Scholar
- Mocroft A, Kirk O, Barton SE, Dietrich M, Proenca R, Colebunders R et al. Anaemia is an independent predictive marker for clinical prognosis in HIV-infected patients from across Europe. AIDS. 1999;13(8):943-50. PubMed | Google Scholar
- Herbst AJ, Cooke GS, B�rnighausen T, KanyKany A, Tanser F, Newell M-L. Adult mortality and antiretroviral treatment roll-out in rural KwaZulu-Natal, South Africa. Bull World Health Organ. 2009;87(10):754-62. PubMed | Google Scholar
- Dalal RP, MacPhail C, Mqhayi M, Wing J, Feldman C, Chersich MF et al. Characteristics and outcomes of adult patients lost to follow-up at an antiretroviral treatment clinic in Johannesburg, South Africa. J Acquir Immune Defic Syndr. 2008;47(1):101-7. PubMed | Google Scholar
- Vogt F, Tayler-Smith K, Bernasconi A, Makondo E, Taziwa F, Moyo B et al. Access to CD4 testing for rural HIV patients: findings from a cohort study in Zimbabwe. PloS one. 2015;10(6):e0129166. PubMed | Google Scholar
- Farham B. Point-of-care CD4 test improves retention and treatment initiation in Mozambique. SA J Contin Med. 2011;29(10):432. PubMed | Google Scholar