Renal oncocytoma: experience of Clinical Urology A, Urology Department, CHU Ibn Sina, Rabat, Morocco and literature review
Marwane Andaloussi Benatiya, Ghizlane Rais, Mounir Tahri, Ali Barki, Hachem El sayegh, Ali Iken, Yassine Nouini, Azzouz Lachkar, Lounis Benslimane, Hassan Errihani, Mohammed Faik
Corresponding author: Dr Rais Ghizlane, H�pital My Abdellah, Institut National d�Oncologie, Postal Code 10100, Rabat, Morocco 
Received: 15 Dec 2011 - Accepted: 12 May 2012 - Published: 24 Jul 2012
Domain: Clinical medicine
Keywords: Renal oncocytoma, tumor, diagnosis, treatment
©Marwane Andaloussi Benatiya et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Marwane Andaloussi Benatiya et al. Renal oncocytoma: experience of Clinical Urology A, Urology Department, CHU Ibn Sina, Rabat, Morocco and literature review. Pan African Medical Journal. 2012;12:84. [doi: 10.11604/pamj.2012.12.84.1428]
Available online at: https://www.panafrican-med-journal.com//content/article/12/84/full
Original article 
Renal oncocytoma: experience of Clinical Urology A, Urology Department, CHU Ibn Sina, Rabat, Morocco and literature review
Renal oncocytoma: experience of Clinical Urology A, Urology Department, CHU Ibn Sina, Rabat, Morocco and literature review
Marwane Andaloussi Benatiya1, Ghizlane Rais2,&, Mounir Tahri1, Ali Barki1, Hachem El sayegh1, Ali Iken1, Yassine Nouini1, Azzouz Lachkar1, Lounis Benslimane1, Hassan Errihani2, Mohammed Faik1
1Clinical Urology Department A, CHU Ibn Sina, Rabat, Morocco, 2Medical Oncology Department, National Institute of oncology, Rabat, Morocco
&Corresponding author
Dr Rais Ghizlane, H�pital My Abdellah, Institut National d�Oncologie, Postal Code 10100, Rabat, Morocco
Oncocytoma is a rare renal tumor first described by Zippel in 1942 [1]. Renal Oncocytomas (RO) are usually solitary masses and represent between 3% and 10% of all renal tumors [2,3]. Oncocytoma is considered to be a benign neoplasm in the majority of cases; this is the reason why there is only one documented case of liver metastasis in literature [4]. Clinically, oncocytoma may be asymptomatic, but symptomatic patients may present initial signs of haematuria, flank pain or palpable mass Oncocytomas usually appear to be unifocal, but multifocal and bilateral appearance and concomitant renal cell carcinoma (RCC) have been reported [2,4].
�
The diagnosis of these benign lesions is generally suggested by computed tomography (CT) or magnetic resonance imaging (MRI). The appearance of a typical central stellate scar can occasionally be mimicked by necrosis in a renal cancer, this feature is not considered specific [5-7]. Moreover, fine needle aspiration and biopsy are often not diagnostic due to oncocytoma having similar histopathologic characteristics as various eosinophilic variants of renal cell carcinoma (RCC) [8]. Histologically, oncocytomas consist of round-to polygonal-shaped cells with an abundant finely granular cytoplasm [2,9]. Careful attention to pathologic features and the adjunctive use of immunostains can aid in discriminating oncocytoma from other renal tumors characterized by granular, eosinophilic cytoplasm, especially chromophobe renal cell carcinoma [8]. Therefore, RO pose a special diagnostic and therapeutic challenge. Since no safe preoperative diagnostic differentiation between oncocytomas and RCC can be achieved, most patients undergo radical nephrectomy. However, nephron-sparing and laparoscopic surgical approaches (partial nephrectomy, enucleation or wegde resection) can be used to treat appropriately selected patients [4,9]. The primary aim of this analysis is to outline the clinico-epidemiological, therapeutic profile and outcomes of patients with renal oncocytoma in our institution.
We reviewed 130 primary nonurothelial epithelial renal neoplasms with primary resection at the Department of Urology A of the CHU IBN SINA (Rabat, Morocco) between the years 1990 and 2008 and classified 6 cases (4,6%) as renal oncocytomas. All patients underwent preoperative CT scan, in order to assign clinical stage according the TNM 2002 Staging System [10]. Pathological staging was assigned in accordance with the same staging system [10]. Data were collected from patient medical files and, clinicopathologic data including patient characteristics, clinical manifestations, surgical technique, pathologic findings and clinical outcome were analyzed. The detailed histopathological report was reviewed. Tumor size was evaluated by measuring the largest diameter of the tumor mass removed surgically. Every 6 months, patients underwent a periodical follow-up which included a physical examination, routine laboratory evaluation, ultrasounds and CT scan.
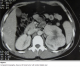
The study population was composed of a man and five women, and the mean age was 53 �9.7 years (range 34 to 61 years). Only One patient was asymptomatic at presentation and was diagnosed incidentally as renal tumor by sonography. Five patients (83%) had flank pain and two (33%) had macroscopic hematuria. Physical examination was normal in all patients. The tumor was right sided in 4 cases (66%) and left sided in 2 cases (33%). No bilateral or multifocal disease was observed. All patients underwent CT scan which showed a centrally located stellate area of low attenuation in three of them (Figure 1). Preoperatively, the clinical impression of oncocytoma had been considered in only 3 (50%) patients following various imaging studies. However, other radiologic features suspecting RCC were noted and patients underwent radical nephrectomy. Definitive diagnosis was assessed in all cases postoperatively.
�
All the tumors were well circumscribed but unencapsulated. Tumor size ranged from 6,5 cm to 12 cm in greatest dimension, with a mean tumor size of 8.75�2.04 cm. Four patients were classified in stage pT2 and two in stage p T1. Most of the pathological features of the studied oncocytoma were typical of this entity. Surgical specimens were well-circumscribed, with no encapsulated masses, mahogany brown or pale yellow in color. Predominant cell type was a typical oncocytoma with general low mitotic activity. No extension to perinephric fat tissue or lymphovascular invasion was observed. After a mean follow-up of 36 months (range 26-62), there was neither recurrence nor death from oncocytoma. Accordingly, the disease-specific survival was 100%. The demographic and clinical features of the patients are summarized in Table 1.
Renal oncocytomas (RO) occur with an overall incidence of 3�10% among all renal tumors and are detected by chance mostly [2,3]. Comparatively, our study shows that the incidence was 4.6% of the primary resectable renal masses in a 19-year period. RO occur more frequently in men than in women with male/female ratio of 2-3:1, and peak occurrence is between the ages of 40 and 60.have approximately the same mean age incidence as RCC [11]. The demographic characteristics of our population (Table 1) were slightly similar to those ones of previous reports [4,12,13]. Similarly, in our series, the mean age at diagnosis was 53 years; however there was a female predilection with a male-to-female ratio of 1:5.
�
Most of renal oncocytomas are asymptomatic at presentation and are discovered incidentally during evaluation for nonurological problems, whereas hematuria and pain occur in a minority of patients. The reported incidence of incidentally discovered masses varied from 50% [14] to 100% [15]. In our series, only one patient was asymptomatic, five patients had pain and two had hematuria (due probably to the large size of tumor at diagnosis).�
RO usually appear as solitary lesions measuring 4-8 cm. The mean size of tumor was 8,75cm in our report, which is quietly larger than previously reported in the literature [13-16]. Multifocal and bilateral appearance have been observed in 4�6% and 4%, respectively [3,12]. Accordingly, a multifocal and bilateral growth pattern is rare. No case of bilateral or multifocal oncocytoma was observed in our series.�
No safe preoperative diagnostic differentiation between oncocytomas and RCC can be achieved because of the lack of pathognomonic radiographic signs. CT-scans show oncocytomas to be more homogenous than RCC. It usually reveals a solid homogeneous lesion with a centrally located stellate area of low attenuation; however, this is considered nonspecific and occurs in only 33% of oncocytomas [4]. On MRI, most oncocytomas demonstrate low signal intensity relative to the renal cortex on T1-weighted images [2]. Twenty-seven percent of oncocytomas also demonstrate a central stellate scar on MRI [17]. Angiography sometimes reveals the typical spoke-wheel formation; Bioptic procedures contain the risk of seeding tumor cells and cannot establish a safe distinction as well [18]. In our series, 3 patients presented with typical CT findings especially with a centrally located stellate area. However, these characteristics may suggest but cannot definitively diagnose oncocytoma [5,19]�
Recently, few articles have reported the use of positron emission tomography (PET) for the diagnosis of oncocytoma [20,21]. Blake and al reported a case of RO displaying intense activity on 18F-fluorodeoxyglucose PET [20], but it is important to recognize that oncocytomas can yield false-positive results on 18F-FDG PET, and absolute radiologic differentiation of oncocytoma from RCC remains elusive. Finally, the novel imaging techniques were investigational, and further validated studies are required. We think that the diagnostic process may be complex and it could involve both imaging and bioptical assessment [2,6,7,22]. To date, controversies still remain as far as exact radiological characterization of renal masses. Even if preoperative distinction between oncocytoma and RCC is considered as a critical step to avoid aggressive surgical procedures, to date any imaging technique can really distinguish between oncocytoma and malignant lesions [2]. In facts, it is widely recognized that the presence of renal clear cell carcinoma (RCC) with oncocytic features may occur, and there may be a frequent coexistence of RCC in the same or contralateral kidney of patients [23].Thus, we believe percutaneous core biopsy or fine needle aspirations play a very limited role in identification of renal neoplasm, but in certain circumstances, biopsy could definitively establish diagnosis [22].�
Histologically, renal oncocytomas are typically well-circumscribed and often encapsulated [2,9]. A central white-colored scar is occasionaly observed, especially in larger tumors[2,4]. Microscopically, RO consist of a pure population of oncocytes, which are large well-differentiated neoplastic cells with intensely eosinophilic granular cytoplasm due to the large number of mitochondria [9,24-25]. Like chromophobe carcinomas, oncocytomas appear to originate from collecting duct cells [25]. In most cases, oncocytomas and different histological subtypes of RCC can be differentiated on gross inspection and from H&E-stained microscopic slides. Sometimes the differentiation is difficult, especially that among the eosinophilic variant of chromophobe RCC, the granular variant of conventional RCC, and oncocytoma [26,27].�
With regard to Hales�s colloidal iron stain, chromophobe RCC show strong diffuse positivity and oncocytoma show negative or weak focal staining. Recent data proposed that cytokeratin staining profiles may be useful for discriminating oncocytoma from its renal mimics: oncocytomas are typically CK7�, CK14+ and CK20+, while most chromophobe RCCs are positive for CK7; the various eosinophilic RCC are typically negative for CK14, and only 0�8% of RCCs are positive for CK20 [28,29]. In our series, no immunohistochemichal staning was realized, because histological features were typical and in accordance with other similar reports.�
Despite their benign behavior, oncocytomas should be monitored closely and treated if there is evidence of coexisting RCC, which occurs in 10% to 32% of reported patients [2], or if it had rapid growth with a risk of destroying the adjacent renal parenchyma [30].�
Surgical treatment of RO is still unclear. Although radical surgery has been employed in the past as principal therapy, more precise preoperative and peri-operative diagnosis should allow more frequent use of conservative surgery, such as partial nephrectomy or tumor excision. Currently, nephron-sparing surgery has been recommended as the standard of care for renal oncocytoma. These include nephronsparing surgery, cryoablation, radiofrequency ablation, high intensity focused ultrasound, microwave thermotherapy and interstitial photon irradiation. Actually, cryo and radiofrequency ablation are the most studied. They have been performed laparoscopically and percutaneously[11]. Renal oncocytoma was not specifically considered as a leading diagnostic differential in our series, because oncocytomas are fairly rare in our context. Therefore, all patients underwent radical nephrectomy, but we think that conservative surgery, as possible, should be adopted in future cases.
Renal oncocytoma has a benign clinical course with excellent long-term outcomes. In our series, it happened mostly in females and is more frequently symptomatic. As the overwhelming majority of cases behave in a benign fashion, conservative surgery is the mainstay of treatment, especially for patients with small tumors. However, as preoperative diagnosis based on imaging studies was uncertain, radical nephrectomy was warranted for all patients in our series.
The authors declare no competing interests.
MB and G.R were involved in the analysis of the data and the literature research, and also wrote the manuscript. AB helped with the patient management and revision of the manuscript. MT helped with the literature research. HS helped with the literature research. YN helped with modifications and revision of the manuscript. AI,YN, LB,AL, HE, MF approved the treatment and analyzed the literature data. All authors read and approved the final manuscript.
We would like to thank the Department of Radiology for their cooperation and help in making the slide and the printing thereof.
Table 1: Demographic characteristics and clinical features of the patients
Figure 1: Computed tomography, shows a left renal tumor with central stellate scar
- Zippel L. Zur kenntnis der onkocyten. Virchows Arch. 1942;308:360�82
- Chao DH, Zisman A, Pantuck AJ, Freedland SJ, Said JW, Belldegrun AS. Changing concepts in the management of renal oncocytoma. Urology. 2002 May;59(5):635-42. This article on PubMed
- Dechet CB, Sebo T, Farrow G, Blute ML, Engen DE, Zincke H. Prospective analysis of intraoperative frozen needle biopsy of solid renal masses in adults. J Urol. 1999 Oct;162(4):1282-4; discussion 1284-5. This article on PubMed
- Perez-Ordonez B, Hamed G, Campbell S, Erlandson RA, Russo P, Gaudin PB, Reuter VE. Renal oncocytoma: a clinicopathologic study of 70 cases. Am J Surg Pathol. 1997 Aug;21(8):871-83. This article on PubMed
- Davidson AJ, Hayes WS, Hartman DS, McCarthy WF, Davis CJ Jr. Renal oncocytoma and carcinoma: failure of differentiation with CT. Radiology. 1993 Mar;186(3):693-6. This article on PubMed
- Hilton S. Imaging of renal cell carcinoma. Semin Oncol. 2000 Apr;27(2):150-9. This article on PubMed
- Liu J, Fanning CV. Can renal oncocytomas be distinguished from renal cell carcinoma on fine-needle aspiration specimens? A study of conventional smears in conjunction with ancillary studies. Cancer. 2001 Dec 25;93(6):390-7. This article on PubMed
- Kuroda N, Toi M, Hiroi M, Shuin T, Enzan H. Review of renal oncocytoma with, focus on clinical and pathobiological aspects. Histol Histopathol. 2003; 18: 935-942 . This article on PubMed
- Morra MN and Das S. Renal oncocytoma: A review of histogenesis, histopathology, diagnosis and treatment. J Urol. 1993; 150:295-302. This article on PubMed
- Sobin L, Wittekind C. TNM classification of malignant tumors, 6th ed. New York: Wiley; 2002
- Ponholzer A, Reiter WJ, Maier U. Organ sparing surgery for giant bilateral renal oncocytoma. J Urol. 2002 Dec;168(6):2531-2. This article on PubMed
- Amin MB, Crotty TB, Tickoo SK, Farrow GM. Renal oncocytoma: a reappraisal of morphologic features with clinicopathologic findings in 80 cases. Am J Surg Pathol. 1997 Jan;21(1):1-12. This article on PubMed
- Davis Jr CJ, Mostofi FK, Sesterhenn IA. Renal oncocytoma. Clinipathological study of 166 patients. J Urogenet Pathol
- Frydenberg M, Eckstein RP, Saalfield JA, Breslin FH, Alexander JH, Roche J. Renal oncocytomas - An australian experience. Br J Urol. 1991 Apr;67(4):352-7. This article on PubMed
- Klein MJ, Valensi QJ. Proximal tubular adenomas of kidney with so-called oncocytic features. A clinicopathologic study of 13 cases of a rarely reported neoplasm. Cancer. 1976 Aug;38(2):906-14. This article on PubMed
- Romis L, Cindolo L, Patard JJ, Messina G, Altieri V, Salomon L, et al. Frequency, clinical presentation and evolution of renal oncocytomas: multicentric experience from a European database. Eur Urol. 2004 Jan;45(1):53-7; discussion 57. This article on PubMed
- Harmon WJ, King BF, Lieber MM. Renal oncocytoma: magnetic resonance imaging characteristics. J Urol. 1996 Mar;155(3):863-7. This article on PubMed
- Dechet CB, Sebo T, Farrow G, Blute ML, Engen DE, Zincke H. Prospective analysis of intraoperative frozen needle biopsy of solid renal masses in adults. J Urol. 1999 Oct;162(4):1282-4; discussion 1284-5. This article on PubMed
- Tikkakoski T, Paivansalo M, Alanen A, Nurmi M, et al. Radiologic findings in renal oncocytoma. Acta Radiol. 1991 Sep;32(5):363-7. This article on PubMed
- Blake MA, McKernan M, Setty B, Fischman AJ, Mueller PR. Renal oncocytoma displaying intense activity on 18F-FDG PET. AJR Am J Roentgenol. 2006 Jan;186(1):269-70. This article on PubMed
- Shriki J, Murthy V, Brown J. Renal oncocytoma on 1-11C acetate positron emission tomography: case report and literature review. Mol Imaging Biol. 2006 Jul-Aug;8(4):208-11. This article on PubMed
- Schatz SM, Lieber MM. Update on oncocytoma. Curr Urol Rep. 2003 Feb;4(1):30-5. This article on PubMed
- Licht MR, Novick AC, Tubbs RR, Klein EA, Levin HS, Streem SB. Renal oncocytoma: clinical and biological correlates. J Urol. 1993 Nov;150(5 Pt 1):1380-3. This article on PubMed
- Lieber MM. Renal oncocytoma: prognosis and treatment. Eur Urol. 1990; 18(2):17-21. This article on PubMed
- Zerban H, Nogueira E, Riedasch G, Bannasch P. Renal oncocytoma: origin from the collecting duc. Virchows Arch B Cell Pathol Incl Mol Pathol. 1987; 52(5):375-387. This article on PubMed
- Crotty TB, Farrow GM, Leiber MM. Chromophobe cell renal carcinoma: clinicopathologic feature of 50 cases. J Urol. 1995; 154(3):964-967. This article on PubMed
- Cochand-Priollet B, Molini� V, Bougaran J, Bouvier R, et al. Renal chromophobe cell carcinoma and oncocytoma. A comparative morphologic, histochemical, and immunohistochemical study of 124 cases. Arch Pathol Lab Med. 1997 Oct;121(10):1081-6. This article on PubMed
- Stopyra GA, Warhol MJ, Multhaupt HA. Cytokeratin 20 immunoreactivity in renal oncocytomas. J Histochem Cytochem. 2001 Jul;49(7):919-20. This article on PubMed
- Chu PG, Weiss LM. Cytokeratin 14 immunoreactivity distinguishes oncocytic tumour from its renal mimics: an immunohistochemical study of 63 cases. Histopathology. 2001 Nov;39(5):455-62. This article on PubMed
- Neuzillet Y, Lechevallier E, Andre M, Daniel L, Nahon O, Coulange C. Follow-up of renal oncocytoma diagnosed by percutaneous tumor biopsy. Urology. 2005 Dec;66(6):1181-5. This article on PubMed