Prevalence and factors associated with transmission of schistosomiasis in school-aged children in South Sudan: a cross-sectional study
Mutale Nsakashalo Senkwe, Kibebu Kinfu Berta, Samuel Makoy Yibi, Julia Sube, Alex Bidali, Abias Abe, Adiele Onyeze, Jane Pita Hilary Ajo, John Rumunu Pascale, Fabian Ndenzako, Olushayo Oluseun Olu
Corresponding author: Mutale Nsakashalo Senkwe, World Health Organization, Country Office, Juba, South Sudan 
Received: 24 Feb 2022 - Accepted: 25 May 2022 - Published: 07 Jun 2022
Domain: Epidemiology,Public health
Keywords: Schistosomiasis, S. haematobium, S. mansoni, bilharzia, circulating cathodic antigen, Kato Katz, urine filtration, haematuria, mapping surveys, South Sudan
This article is published as part of the supplement The public health situation in South Sudan, a humanitarian context: successes, challenges and key lessons for the future, commissioned by World Health Organization South Sudan, Juba, South Sudan.
©Mutale Nsakashalo Senkwe et al. Pan African Medical Journal (ISSN: 1937-8688). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Mutale Nsakashalo Senkwe et al. Prevalence and factors associated with transmission of schistosomiasis in school-aged children in South Sudan: a cross-sectional study. Pan African Medical Journal. 2022;42(1):2. [doi: 10.11604/pamj.supp.2022.42.1.34006]
Available online at: https://www.panafrican-med-journal.com//content/series/42/1/2/full
Research 
Prevalence and factors associated with transmission of schistosomiasis in school-aged children in South Sudan: a cross-sectional study
Prevalence and factors associated with transmission of schistosomiasis in school-aged children in South Sudan: a cross-sectional study
![]() Mutale Nsakashalo Senkwe1,&, Kibebu Kinfu Berta1, Samuel Makoy Yibi2, Julia Sube1, Alex Bidali1, Abias Abe3, Adiele Onyeze4, Jane Pita Hilary Ajo1, John Rumunu Pascale2, Fabian Ndenzako1,
Mutale Nsakashalo Senkwe1,&, Kibebu Kinfu Berta1, Samuel Makoy Yibi2, Julia Sube1, Alex Bidali1, Abias Abe3, Adiele Onyeze4, Jane Pita Hilary Ajo1, John Rumunu Pascale2, Fabian Ndenzako1, ![]() Olushayo Oluseun Olu1
Olushayo Oluseun Olu1
&Corresponding author
Introduction: South Sudan is affected by a high burden of Neglected Tropical Diseases (NTDs). The country is very vulnerable to NTDs due to its favourable tropical climate and multiple risk factors. However, the distribution of the diseases and the populations at risk for the various NTDs is unknown. This paper described the distribution of schistosomiasis in 58 counties and 261 schools in South Sudan.
Methods: a descriptive quantitative cross-sectional study of schistosomiasis in 58 counties in 8 states of South Sudan recruited school-aged children. Using different laboratory techniques, the children were tested for Schistosoma mansoni (S. mansoni) and Schistosoma haematobium (S. haematobium). A quantitative descriptive statistical was performed to determine the prevalence rates and the endemicity of schistosomiasis among 13,286 school-aged children.
Results: the overall prevalence of S. mansoni and S. haematobium were 6.1% and 3.7% using Kato Katz and urine filtration concentration testing techniques. The highest state prevalence was reported in Western Equatoria for both S. mansoni (14.7%) and S. haematobium (7.3%). The age of the participants varied from 4 to 18 years; of these, children 10 to 12 years old had the highest prevalence of S. mansoni (6.8%) and S. haematobium (3.7%). The prevalence of S. mansoni (7% male vs 5% female) and S. haematobium (3.6% male vs 3.1% female) were higher in males than females. The likelihood of the prevalence of S. mansoni in males was 1.42 (95% CI:1.23, 1.64) higher than in females, while for S. haematobium, 1.36 (95% CI:1.12, 1.65) higher than in females. The prevalence of S. mansoni and S. haematobium showed a statistically significant gender difference (P< 0.05).
Conclusion: the study had provided evidence of the distribution of schistosomiasis in South Sudan for policy direction and recommended annual preventive chemotherapy with praziquantel in all endemic areas.
Schistosomiasis, bilharzia, or snail fever is a common neglected tropical disease (NTD), highly endemic in the tropics and subtropical areas [1]. It is caused by trematode parasites of the genus Schistosoma which has five species known to infect and cause morbidity in humans, namely S. haematobium, S. intercalatum, S. japonicum, S. mekongi, and S. mansoni [2]. Sexual reproduction occurs in the human host, where fertilized eggs are produced and released through urine and stool. The eggs trapped in tissues induce inflammation and die [3]. In freshwater, free-living miracidia released by the eggs infect the snail and undergo asexual replication to release the cercariae, which penetrates the skin of humans who come into contact with the infested water [2]. The African region is predominantly affected by S. mansoni and S. haematobium due to the availability of freshwater where the intermediate hosts thrive well. S. japonicum and S. mekongi are confined to Asia, while the Caribbean and Latin America are affected mainly by S. mansoni [4]. Schistosomiasis is the commonest NTD affecting at least 230 million people globally. Sub-Saharan Africa contributes 90% of the disease burden [1-3].
Acute schistosomiasis, or “Katayama Syndrome” is a short-lived hypersensitivity reaction related to tissue migration of the larva. Acute infection presents as fever, myalgia, headache, cough, and rash. The symptoms appear 14-84 days after infection and may last for several weeks [5]. At the same time, chronic diseases include bladder cancer, blood in the stool, constipation, diarrhoea, bowel wall ulceration, fibrosis, hyperplasia, polyposis, and portal hypertension [5,6].
In South Sudan, hospital records have indicated ongoing schistosomiasis transmission in Upper Nile, Jonglei, Eastern and Central Equatoria states with two species, S. mansoni and S. haematobium. The eastern and central parts of South Sudan including Jongile, Unity, Upper Nile state, and Greater Pibor Administrative Area (GPAA) is prone to frequent flooding. Abnormally heavy rainfall causes the overflow of Lol, Nile, Pibor, Sobat and other rivers that lead to inland flooding where people reside and causes displacement. In addition, the flooding increases the chance of contact between people and the parasite carrying an intermediate host [7,8]. A survey conducted in 2009 in Northern Bahr el Ghazal showed that S. haematobium is endemic mainly in areas along the Lol River [9]. In a 2010 survey, both S. mansoni and S.haematobium were endemic throughout Unity and some foci in Central Equatoria and Eastern Equatoria States [10]. These observations suggested that schistosomiasis was present, and transmission was ongoing; however, the country's actual geographical distribution, extent, and prevalence remain unknown. South Sudan is yet to scale up and sustain control activities, ensure access to essential medicines, and complement public health interventions in response to the resolution of World Health Assembly 54 (WHA54) schistosomiasis and soil-transmitted helminth infections [11]. To adequately implement control and elimination measures, and monitor and assess the impact of interventions, accurate estimates of the disease burden and prevalence are crucial. The manuscript describes the national distribution of human schistosomiasis in 58 counties from 2016 through 2019.
Study design and area: we conducted a cross-sectional quantitative study to know the prevalence of schistosomiasis among school-aged children 10 to 14 years of age in South Sudan from 2016 to 2019. The study was based on the methods and procedures stated recommendation under WHO neglected tropical diseases transmission assessment survey guidelines [12].
Study site selection and sample size: South Sudan is administratively divided into 10 states which are further divided into 79 counties. Twenty-two counties in four states, with available recent schistosomiasis prevalence data, were identified and excluded from the current study. The states without prevalence and transmission risk data were included in the study. Besides, the selection was guided by previous knowledge of ecological factors (i.e., proximity to lakes, streams, and other water bodies) from different geographical locations [13]. It had helped the management of survey implementation including logistics constraints. All 52 counties in the six remaining states and six counties (Panyijar county from Unity and Kapoeta South, Kapoeta North, Torit, Magwi, and Lapon counties from Eastern Equatoria) were included in the study with a total of 58 counties. In these counties, the selection of schools was based on the WHO survey sample builder stated in the assessment guideline [12]. It is an excel based tool used to develop survey design and random samples. It had facilitated the random selection of schools and children from a list of randomized numbers. A cluster-based simple random sampling technique was applied to select five schools in each county. Before the section of the schools, all schools in the county were listed down for random sections. Fifty children aged between 10 and 14 years, balanced by gender, from each school were randomly enrolled on the study. WHO NTD transmission assessment survey guideline recommends 50 children per cluster [12]. When the target was not reached, children from an expanded age range were selected. Otherwise, the target age from the surrounding villages or the nearest school was sampled to complete the required samples. Each child submitted a stool and urine sample to test the presence of intestinal Schistosoma (S. mansoni) eggs and urinary Schistosoma (S. haematobium), respectively.
Data collection: the data collection was conducted by nine teams, each comprising a supervisor (i.e. laboratory technologist or an experienced laboratory technician), two laboratory technicians, a data clerk and a social mobilizer. A data manager coordinated the work of all data clerks and had access to the Open Data Kit (ODK) web-based cloud to monitor data as entries were made and uploaded using smartphones. A detailed fieldwork plan was developed based on the teams created and the counties included in the study. The local health personnel led the study teams to the selected schools. Geographical coordinates (i.e. longitude, latitude, and altitude) were taken at the survey sites using smartphones. Children aged between 10 to 14 years were randomly selected, and both urine and stool samples were collected from the selected children. The survey determined the prevalence of S. haematobium based on micro-hematuria or parasite eggs in urine using reagent dipstick and urine filtration procedures, respectively. A reagent dipstick for hematuria is a qualitative rapid diagnostic tool used to screen school-aged children for urinary schistosomiasis. The interpretation of the result was based on the presence or absence of a test strip line. Samples with red test lines were labelled as positive; however, further quantification of the test line was not performed. The urine filtration procedures determine the concentration of the egg per 10 millilitres (ml) of the urine sample. The testing result was reported as light: <50 ova per 10ml; heavy: ≥50 ova per 10ml.
The prevalence of S. mansoni was determined parasite eggs in stool using Kato Katz or circulating cathodic antigen (CCA) in urine. The Kato Katz technique is used for semi-quantitative and quantitative diagnosis of Schistosoma species (S. mansoni), soil-transmitted helminthiasis caused by Trichuris trichiura, hookworm, and ascaris lumbricoides. WHO had recommended the use of the technique in areas with moderate to high transmission of intestinal schistosomiasis (10-49.9% and ≥50%) [14]. A gram of faeces was collected and tested for the presence of an egg (egg per gram of faeces) using a microscope. The testing result was reported as light infection (0-99 e.p.g.), medium (100-399 e.p.g.), and heavy: (≥400 e.p.g.). A point-of-care Circulating Cathodic Antigen (POC-CCA) urine test is a simplified rapid diagnostic test (RDT) used for the qualitative detection of an active S. mansoni infection. The test detects Schistosoma specific antigen execrated in urine [15]. The result was labelled positive based on the appearance of a visible test line seen within 20 minutes of application of the urine. The prevalence estimate for each survey site was classified into non-endemic (0%), low (<10%), moderate (10-49.9%), or high-risk (≥50%). The prevalence estimate for each survey site was classified into non-endemic (0%), low (<10%), moderate (10-49.9%), or high-risk (≥50%) areas.
Data analyses: in 2016, data were captured on to a server hosted at the national level using Bold Like Us (BLU) Studio 5.5 smartphones running Android 4.2 (Jelly Bean) through a modified version of Open Data Kit (ODK), the LINKS application. Raw data on schistosoma infection were downloaded into Microsoft Excel, cleaned, and summarized from the server. In subsequent years, the Expanded Special Project for Elimination of Neglected Tropical Diseases (ESPEN) Collect application was used to capture, facilitate cleaning, and aggregation collected individual data into site data. The data entered was synchronized and hosted at the Standard Code Server. Only authorized officers had access to download individual data from the dashboard on the server. We conducted descriptive analyses on socio-demographic characteristics and epidemiological distribution of schistosomiasis. We have used International Business Machine (IBM) Statistical Package for Social Science Window Version 26.0 (SPSS V26) to conduct descriptive epidemiology. A prevalence map was produced using ArcGIS (ESRI, California, and the USA). A two-by-two table was used to determine the relationship between test types (urine filtration versus dipstick and CCA versus Kato Katz) and test result (positive versus negative). A two-by-two table was performed to compare whether the observed prevalence of S. haematobium and S. mansoni results for the two test types for each species were statistically different enough to reject the null hypothesis (there is no significant difference between the two test types) at a 95% confidence interval (CI) and a significant level of 0.05.
Ethical consideration: during the 2016, 2018, 2019 survey ethical approval was obtained from the Research and Ethics Committee of the national Ministry of Health. During the survey, consent was obtained from the headteachers and parents. All positive cases found during the study received a single dose of 20 mg/kg praziquantel. However, ethical approval was not obtained to use the secondary anonymized data in the analysis. Data protection measures were employed to ensure the security of the data.
Study population and schistosoma infection prevalence: the screening of the 13,286 samples collected from the children revealed the presence of both S. haematobium (n=441) and S. mansoni (n=804) Table 1. The age of the children tested varied from 4 to 18 years old. The age group 10 to 12 years had the highest prevalence rate for S. mansoni, S. haematobium, and Co-infection of the two species. Of the 6,982 males tested for S. mansoni, 7% tested positive. The prevalence of S. mansoni in males was 1.42 (95% CI: 1.23,1.64) times higher than in females. Regarding S. haematobium, 3.8% of males were tested positive compared to 2.8% positive females. The prevalence of S. haematobium in males was 1.36 (95% CI:1.12,1.65) times greater than in females.
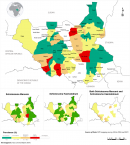
Prevalence of schistosoma infection by location: S. mansoni infection was found in all the eight states of South Sudan, with an overall prevalence rate of 6.1% as shown in Table 2. Western Equatoria had the highest prevalence rate of S. manosni (14.7%). The prevalence rate varied from 0 to 65.9% at the county level, with the highest in Bor South (65.9%). At the school level, 86% out of 261 schools were endemic to S. mansoni. The school prevalence rate of S. manosni varied from 0 to 93.3%; the highest was recorded at Bandala primary school in Nagero county (93.3%) of Western Equatoria state. S. haematobium, infection was found in all eight states with an overall prevalence rate of 3.7%. Unity state had the highest prevalence rate (17.2%) Table 2. At the primary school level, 72 (%) out of 261 schools were endemic to S. haematobium. The school prevalence rate varied from 0% to 84.4%, with the highest at Toch primary school in Old Fangak (84.4%) in Jonglei state. Overall, high endemicity for schistosomiasis combined is in Bor South, Old Fangak, Ezo and Ibba counties as indicated in Figure 1. Bor South county has highest the endemicity for S. mansoni species.
Prevalence of parasitic intensity: light intensity infection (1-99 egg per gram of faeces (e.p.g.) for S. mansoni by Kato Katz technique was observed in all the states that varied from 0.2 and 13.6% as indicated in Table 3. Western Equatoria recording the highest. Medium intensity infections (100-399 e.p.g. of faeces) were observed in five out of the eight states at 1.3% in Jonglei, 1.1% in Western Equatoria and 0.1% each in Eastern Equatoria, Lakes and Warrap states and heavy intensity infection (>400 e.p.g. of faeces) was observed in three states of Jonglei at 0.9%, Western Equatoria 0.3% and Lakes 0.1%. For S. haematobium using urine filtration, light intensity infection (<50 eggs/10mls of urine) was observed in the eight states with the highest in Unity (16.2%), Western Equatoria (4.4%) and Jonglei (3.8%), while heavy intensity infections (>50 eggs/10mls of urine) were observed in all except Western Bahr el Ghazal.
Overall, the S. mansoni prevalence rate for 2016 surveys using Circulating Cathodic Antigen (CCA) was 19.7%, while for the Kato Katz technique was 7.8%. The likelihood of the test being positive was 2.87 (95% CI: 2.54, 3.24) higher for CCA than Kato Katz. There was a statistically significant difference between the two tests (P<.001) (Table 4). As for S. haematobium, the overall prevalence rate using urine filtration was 5.0%, while for dipstick was 3.4%. The likelihood of the test being positive was 1.49 (95% CI: 1.27, 1.75) higher in urine filtration compared to the dipstick. There was a statistically significant difference between the two tests (P<.001) (Table 4).
The cross-sectional study provides the baseline data on schistosomiasis prevalence as the first sizeable school-based survey in the 58 unmapped counties of South Sudan. Our study revealed the presence of schistosomiasis infections in all the states and almost in every county with a heterogeneous prevalence rate of S. haematobium and S. mansoni across the country, indicating the latter to be highly endemic [16]. There is a high prevalence of S. mansoni in Jonglei, Western Equatoria and the Lakes States and S. haematobium in Jonglei, Unity and Western Equatoria States. Higher co-infection has been observed in Jonglei State. The parasitic intensity was light across the eight states, with heavy intensities observed more for S. haematobium than S. mansoni. Jonglei and the Western Equatoria States had co-infection of the two Schistosoma parasites [16]. Our findings showed the schistosomiasis burden in the central and southwestern regions of the country is high. The persistence of schistosomiasis's high prevalence rate over the years indicates that no control or elimination measures are being implemented [16]. In Africa, the prevalence of human schistosomiasis is dependent on the level of environmental sanitation, the suitability of the area for the intermediate snail hosts, and the type of the snails. In South Sudan, this has been exacerbated by political conflict negatively impacting the country's socio-economic status and funding for preventing and controlling infectious diseases such as schistosomiasis [17]. Previous surveys of 2010 showed a constantly lower prevalence of S. haematobium than that of S. mansoni, a similar finding in six of the eight states of our study a common feature in Madagascar due to the Biomphalaria snails in the Nile River and high positivity of S. mansoni [18]. One unexpected finding was the low prevalence of schistosomiasis in Eastern Equatoria (2.3%) compared to the previous survey findings of 14.2% 6 years prior. The conflicts in the region and mainly children (62% of children became refugees) and women fleeing insecurity, violence, and famine to Uganda, may elucidate the reduction [19,20]. Nonetheless, the rapid decline in prevalence rate requires further research to verify these findings and assess the contributing factors at length.
Previous schistosomiasis predictive maps for South Sudan suggested a high prevalence rate of S. haematobium in Greater Bahr El Ghazel between 10-25% and the Equatoria States at ≤5%, and findings of a non-endemic Western Bahr El Ghazal and low prevalence rate of 0.5% in Western Equatoria [21]. While the surveys of 2009 found a prevalence rate of 3% in Northern Bahr El Ghazal [9]. Deganello in 2007 reported the presence of S. mansoni mainly in Central Equatoria, with a national prevalence of both species between 10-25% again. This is inconsistent with our observed findings which are relatively lower with an overall prevalence rate of 7.9% (7.3-8.3% at 95% confidence interval) [22]. However, the conclusions of Jonglei were consistent with the predicted low prevalence of <5%. Overall, the current state-level data indicates an uneven distribution throughout the country, with a few previously thought-to-be highly endemic places found to be lowly or non-endemic. The prevalence of schistosomiasis from the initial prevalence rate surveys and our study is not surprising as no sustainable control measures were instituted.
An alarmingly (very high) prevalence rate of schistosomiasis was observed in Bor South, where schools recorded over 90% site prevalence rates. This high prevalence could be attributed to the long-running civil war that has devastated social and health services in the area and the presence of the schistosoma, intermediate host, and freshwater bodies. The high prevalence was seen in Bor South, Ibba, Ezo, Awerial, Nagero, and Old Fangak were comparable to other focal prevalence rate surveys in East African countries, Madagascar, Egypt, and the Democratic Republic of Congo, an indication of the high burden of schistosomiasis in South Sudan and many parts of the Africa Region [21,22]. Also, the observed high prevalence in rural areas compared to urban areas such as Wau may be attributed to inadequate sanitary facilities, contaminated water sources for domestic chores, bathing, and insufficient health education on the affected communities´ preventive measures. In some countries, urban infection is found in individuals due to urbanisation. This could be the assumption for the situation seen in Juba, the capital city of South Sudan in the state of Central Equatoria, where the prevalence is high [10]. Typically, if the intermediate host is not present in an area with schistosomiasis cases, it is easier to control through better chemotherapy, sanitation, and water access facilities [23].
Presently, chemotherapy is the most cost-effective approach to controlling schistosomiasis in the short term, but complimenting it with access to clean water and sanitation would help address the risk factors, reduce transmission, and have a broader impact on health. While the provision of water, adequate sanitation, and snail control are essential for schistosomiasis elimination, directing these interventions to selected high transmission areas would ensure a higher impact [4]. The study´s five sites per county is an innovative way to improve resource allocation for interventions to focal transmission. However, this is also a study limitation as some counties are enormous, and the five sites may not be represented precisely. The utilisation of WASH facilities through behavioural modification has been challenging in some countries despite being the best approach to managing health problems [3]. The communities require sufficient knowledge and attitude about schistosomiasis through health education in the endemic areas.
Preventive chemotherapy either school or community-based approaches have not shown significant differences in some countries following treatment [24]. Most children are exposed to similar conditions when they get home with inadequate WASH facilities. Therefore, most schistosomiasis treatment should be both community and school-based, with many children in communities. Treatment immediately after 12 months is vital because delayed repeat therapy may not suppress transmission due to persistent reinfections leading to reduced drug efficacy observed after one to three years [24]. Therefore, the state government should emphasise the need for additional non-drug control measures in highly endemic areas.
This study shows that schistosomiasis is endemic in South Sudan, with a moderate to high prevalence rate in most parts of the country. In these endemic communities we advise the national schistosomiasis control programme to implement the WHO recommendation of annual preventive chemotherapy with a single dose of praziquantel at 75% treatment coverage in all the age groups from 2 years. The treatment should include pregnant women in the second and third trimester, lactating women and adults. In the low endemic communities, the test and treat approach is recommended. The low parasitic intensity for both species in South Sudan is an indication that with suitable interventions, the prevalence of infection in the affected population can reduce to elimination levels. The study has generated crucial epidemiological baseline information and indicators which could guide policy formulation, monitoring, and evaluation of interventions. Heightened health education to reduce contact with contaminated water and access to safe water, sanitation, and hygiene plus environmental interventions including snail control with moluscicides should be complementary measures to reduce the prevalence and burden of schistosomiasis. The effective implementation and monitoring of these strategies will interrupt transmission and eliminate schistosomiasis in South Sudan. The verification for the interruption of transmission should use diagnostics with high sensitivity and specificity.
What is known about this topic
- Although schistosomiasis is endemic in South Sudan, the geographic distribution of the disease is unclear;
- The superior sensitivity of circulating cathodic antigen over KK in the detection of the S. mansoni circulating cathodic antigen.
What this study adds
- This study provides information on the prevalence and distribution of schistosoma infection in South Sudan;
- The younger children are at higher risk of S. mansoni infection;
- Key recommendations for scaling up effective and integrated public health measures for prevention and control of schistosomiasis including treatment of the pre-school age children.
The authors declare no competing interests.
MNS and KKB conceived and wrote the first draft of the manuscript. KKB and MNS conducted the data analyses. OOO, MNS and KKB provided insights into the study's conceptualisation and conducted an extensive review of all manuscript drafts. All authors read and provided significant inputs into all drafts of the manuscript, agreed to be accountable for all aspects of the work and approved the final draft of the manuscript for publication.
The authors would like to thank the WHO Country Office South Sudan, intercountry support team, Zimbabwe for providing technical support to conduct secondary analysis. We thank MoH for providing administrative approval to publish the result. Also, the authors thank the NTD programme of the Ministry of Health of South Sudan for providing unhindered access to the data used for this study. The authors alone are responsible for the views expressed in this article, which do not necessarily represent the institutions' views, decisions, or policies with which they are affiliated.
Table 1: social demographic characteristics of schistosomiasis in school-aged children, South Sudan (2016 to 2019)
Table 2: summary of Schistosoma mansoni laboratory test, South Sudan (2016 to 2019)
Table 3: summary of Schistosoma haematobium laboratory test, South Sudan (2016 to 2019)
Table 4: compare of the performance of the different tests of Schistosoma mansoni and Schistosoma haematobium, South Sudan, (2016 to 2019)
Figure 1: schistosomiasis endemicity map, South Sudan (2016 to 2019)
- Gabrielli AF, Montresor A, Chitsulo L, Engels D, Savioli L. Preventive chemotherapy in human helminthiasis: theoretical and operational aspects. Trans R Soc Trop Med Hyg. 2011 Dec;105(12):683-93. PubMed | Google Scholar
- World Health Organization. Schistosomiasis: progress report 2001 - 2011, strategic plan 2012 - 2020. 2013. Accessed July 23, 2021.
- Colley DG, Bustinduy AL, Secor WE, King CH. Human schistosomiasis. Lancet Lond Engl. 2014;383(9936):2253-2264. PubMed | Google Scholar
- Lu XT, Gu QY, Limpanont Y, Song LG, Wu ZD, Okanurak K et al. Snail-borne parasitic diseases: an update on global epidemiological distribution, transmission interruption and control methods. Infect Dis Poverty. 2018 Apr 9;7(1):28. PubMed | Google Scholar
- Engels D, Zhou XN. Neglected tropical diseases: an effective global response to local poverty-related disease priorities. Infect Dis Poverty. 2020 Jan 28;9(1):10. PubMed | Google Scholar
- Nelwan ML. Schistosomiasis: life cycle, diagnosis, and control. Curr Ther Res Clin Exp. 2019 Jun 22;91:5-9. PubMed | Google Scholar
- Guo SY, Li L, Zhang LJ, Li YL, Li SZ, Xu J. From the one health perspective: schistosomiasis japonica and flooding. Pathogens. 2021 Nov 25;10(12):1538. PubMed | Google Scholar
- Wu XH, Zhang SQ, Xu XJ, Huang YX, Steinmann P, Utzinger J et al. Effect of floods on the transmission of schistosomiasis in the Yangtze River valley, People´s Republic of China. Parasitol Int. 2008 Sep;57(3):271-6. PubMed | Google Scholar
- Sturrock HJW, Picon D, Sabasio A, Oguttu D, Robinson E, Lado M et al. Integrated mapping of neglected tropical diseases: epidemiological findings and control implications for Northern Bahr-el-Ghazal State, Southern Sudan. PLoS Negl Trop Dis. 2009 Oct 27;3(10):e537. PubMed | Google Scholar
- Finn TP, Stewart BT, Reid HL, Petty N, Sabasio A, Oguttu D et al. Integrated rapid mapping of neglected tropical diseases in three states of South Sudan: survey findings and treatment needs. PLoS One. 2012;7(12):e52789. PubMed | Google Scholar
- World Health Assembly. Elimination of lymphatic filariasis as a public health problem. 1997. Accessed July 26, 2021.
- World Health Organization. Control of neglected tropical diseases. Accessed December 7, 2021.
- Rebollo MP, Onyeze AN, Tiendrebeogo A, Senkwe MN, Impouma B, Ogoussan K et al. Baseline mapping of neglected tropical diseases in Africa: the accelerated WHO/AFRO mapping project. Am J Trop Med Hyg. 2021 Apr 26;104(6):2298-2304. PubMed | Google Scholar
- Bharti B, Bharti S, Khurana S. Worm infestation: diagnosis, treatment and prevention. Indian J Pediatr. 2018 Nov;85(11):1017-1024. PubMed | Google Scholar
- Casacuberta-Partal M, Hoekstra PT, Kornelis D, van Lieshout L, van Dam GJ. An innovative and user-friendly scoring system for standardised quantitative interpretation of the urine-based point-of-care strip test (POC-CCA) for the diagnosis of intestinal schistosomiasis: a proof-of-concept study. Acta Trop. 2019 Nov;199:105150. PubMed | Google Scholar
- Rumunu J, Brooker S, Hopkins A, Chane F, Emerson P, Kolaczinski J. Southern Sudan: an opportunity for NTD control and elimination? Trends Parasitol. 2009 Jul;25(7):301-7. PubMed | Google Scholar
- ISPI. Environmental vulnerability: South Sudan´s endgame. 2018. Accessed July 23, 2021.
- Spencer SA, Penney JMSJ, Russell HJ, Howe AP, Linder C, Rakotomampianina ALD et al. High burden of Schistosoma mansoni infection in school-aged children in Marolambo District, Madagascar. Parasit Vectors. 2017 Jun 24;10(1):307. PubMed | Google Scholar
- United Nations Security Council. Working group on children and armed conflict. Accessed July 23, 2021.
- UN HRC. Report of the commission on human rights in South Sudan (A/HRC/34/63) (advance edited version). ReliefWeb, South Sudan, 2017. Accessed July 23, 2021.
- Secor WE, Wiegand RE, Montgomery SP, Karanja DMS, Odiere MR. Comparison of school-based and community-wide mass drug administration for schistosomiasis control in an area of western kenya with high initial Schistosoma mansoni infection prevalence: a cluster randomized trial. Am J Trop Med Hyg. 2020 Feb;102(2):318-327. PubMed | Google Scholar
- Woodall PA, Kramer MR. Schistosomiasis and infertility in East Africa. Am J Trop Med Hyg. 2018 Apr;98(4):1137-1144. PubMed | Google Scholar
- Silva LK, Barbosa LM, Kovach JD, Teixeira RDS, Soares ES, Cardoso CW et al. The changing profile of schistosomiasis in a changing urban landscape. Int J Parasitol . 2020 Jan;50(1):27-34. PubMed | Google Scholar
- Onkanga IO, Mwinzi PNM, Muchiri G, Andiego K, Omedo M, Karanja DMS et al. Impact of two rounds of praziquantel mass drug administration on Schistosoma mansoni infection prevalence and intensity: a comparison between community-wide treatment and school-based treatment in western Kenya. Int J Parasitol. 2016 Jun;46(7):439-45. PubMed | Google Scholar
Search
This article authors
On Pubmed
On Google Scholar
Citation [Download]
Navigate this article
Similar articles in
Key words
Tables and figures
This supplement
- Assessment of infection prevention and control readiness for Ebola virus and other diseases outbreaks in a humanitarian crisis setting: a cross-sectional study of health facilities in six high-risk States of South Sudan (Accessed 1280 times)
- Integrated disease surveillance and response in humanitarian context: South Sudan experience (Accessed 1200 times)
- Prevalence and factors associated with transmission of lymphatic filariasis in South Sudan: a cross-sectional quantitative study (Accessed 1113 times)
- The sensitivity of acute flaccid paralysis surveillance - the case of South Sudan: retrospective secondary analysis of AFP surveillance data 2014-2019 (Accessed 1065 times)
- Acute flaccid paralysis surveillance performance from 2011 to 2020 in Jonglei State, South Sudan: progress and challenges encountered (Accessed 975 times)
- South Sudan's road to universal health coverage: a slow but steady journey (Accessed 896 times)
- Prevalence and factors associated with transmission of lymphatic filariasis in South Sudan: a cross-sectional quantitative study (Downloaded 482 times)
- South Sudan's road to universal health coverage: a slow but steady journey (Downloaded 139 times)
- Integrated disease surveillance and response in humanitarian context: South Sudan experience (Downloaded 84 times)
- Assessment of infection prevention and control readiness for Ebola virus and other diseases outbreaks in a humanitarian crisis setting: a cross-sectional study of health facilities in six high-risk States of South Sudan (Downloaded 81 times)
- Acute flaccid paralysis surveillance performance from 2011 to 2020 in Jonglei State, South Sudan: progress and challenges encountered (Downloaded 76 times)
- The sensitivity of acute flaccid paralysis surveillance - the case of South Sudan: retrospective secondary analysis of AFP surveillance data 2014-2019 (Downloaded 71 times)